MC84 - Southchemistry.com
... If the equilibrium constant for the reaction above is 3.7 x 1015, which of the following correctly describes the standard voltage, E°, and the standard free energy change, G°, for this reaction? (A) E° is positive and G° is negative. (B) E° is negative and G° is positive. (C) E° and G° are both ...
... If the equilibrium constant for the reaction above is 3.7 x 1015, which of the following correctly describes the standard voltage, E°, and the standard free energy change, G°, for this reaction? (A) E° is positive and G° is negative. (B) E° is negative and G° is positive. (C) E° and G° are both ...
CH 5 CHEM1A GASES - Santa Rosa Junior College
... amount (mol,n) of gaseous reactant or product and the gas variables pressure (P), volume (V), and temperature (T). ...
... amount (mol,n) of gaseous reactant or product and the gas variables pressure (P), volume (V), and temperature (T). ...
Name __KEY____________ Per. ______ Polarity and
... from a table. In our textbook there is a table on page 177. As the difference in electronegativity becomes __larger__ (larger/ smaller) bonds will go from being nonpolar covalent to polar covalent to ionic. If a bond is polar that means that the atoms share their electrons __ unevenly__ (unevenly / ...
... from a table. In our textbook there is a table on page 177. As the difference in electronegativity becomes __larger__ (larger/ smaller) bonds will go from being nonpolar covalent to polar covalent to ionic. If a bond is polar that means that the atoms share their electrons __ unevenly__ (unevenly / ...
apch19 Introduction to Entropy POGIL
... We can use the signs of the H and S to predict whether a reaction will happen “spontaneously”. “Spontaneous” means that the reaction will occur on its own without any outside influence. It DOES NOT necessarily mean that it happens immediately or fast. Instead of “spontaneous”, the College Board us ...
... We can use the signs of the H and S to predict whether a reaction will happen “spontaneously”. “Spontaneous” means that the reaction will occur on its own without any outside influence. It DOES NOT necessarily mean that it happens immediately or fast. Instead of “spontaneous”, the College Board us ...
Name AP Chemistry Take Home Quiz – Due Thursday, 1/9/2014
... b. The average kinetic energy of the O2 molecules is greater than that of the He molecules. c. The pressure of the He(g) would be greater than that of the O2(g). d. There are equal numbers of He molecules and O2 molecules. e. The average kinetic energy of the He molecules is greater than that of the ...
... b. The average kinetic energy of the O2 molecules is greater than that of the He molecules. c. The pressure of the He(g) would be greater than that of the O2(g). d. There are equal numbers of He molecules and O2 molecules. e. The average kinetic energy of the He molecules is greater than that of the ...
2004 NEACS Ashdown Exam 1. The allotrope of carbon shown to
... (C) A sample contains many molecules, each of which has one C O bond shorter than the other two. Averaging all of these molecules produces an average distance that is somewhat shorter that a single C O bond. (D) One C O bond is more reactive than the other two because the double bond is less stable. ...
... (C) A sample contains many molecules, each of which has one C O bond shorter than the other two. Averaging all of these molecules produces an average distance that is somewhat shorter that a single C O bond. (D) One C O bond is more reactive than the other two because the double bond is less stable. ...
Year End Review
... c) an increase in the pressure of a gas could be due to a decrease in the number of molecules d) in a mixture of gases, the one with the most molecules exerts the most pressure. 41. Which one of the following is a basic assumption of the kinetic molecular theory? a) particles are in constant random ...
... c) an increase in the pressure of a gas could be due to a decrease in the number of molecules d) in a mixture of gases, the one with the most molecules exerts the most pressure. 41. Which one of the following is a basic assumption of the kinetic molecular theory? a) particles are in constant random ...
Chapter 1 Matter and Change
... States of matter 1) Solid- matter that can not flow (definite shape) and has definite volume. 2) Liquid- definite volume but takes the shape of its container (flows). 3) Gas- a substance without definite volume or shape and can flow. – Vapor- a substance that is currently a gas, but normally is a l ...
... States of matter 1) Solid- matter that can not flow (definite shape) and has definite volume. 2) Liquid- definite volume but takes the shape of its container (flows). 3) Gas- a substance without definite volume or shape and can flow. – Vapor- a substance that is currently a gas, but normally is a l ...
1) A clear glass bottle contains white sand, some nails, salt water
... 1) A clear glass bottle contains white sand, some nails, salt water with some dye dissolved in it, and a layer of gasoline on top. How many phases are present in this system (excluding the bottle and lid)? Four phases are mentioned: (1) white sand, (2) nails, (3) salt water with some dye dissolved i ...
... 1) A clear glass bottle contains white sand, some nails, salt water with some dye dissolved in it, and a layer of gasoline on top. How many phases are present in this system (excluding the bottle and lid)? Four phases are mentioned: (1) white sand, (2) nails, (3) salt water with some dye dissolved i ...
2008 local exam - American Chemical Society
... on that sheet, not written in the booklet. Each student should be provided with an answer sheet and scratch paper, both of which must be turned in with the test booklet at the end of the examination. Local Sections may use an answer sheet of their own choice. The full examination consists of 60 mult ...
... on that sheet, not written in the booklet. Each student should be provided with an answer sheet and scratch paper, both of which must be turned in with the test booklet at the end of the examination. Local Sections may use an answer sheet of their own choice. The full examination consists of 60 mult ...
Chemistry Final Exam Review
... 3. If you have a 2.75dm3 balloon under 455kPa of pressure at 100oC, what will the volume be at STP? ...
... 3. If you have a 2.75dm3 balloon under 455kPa of pressure at 100oC, what will the volume be at STP? ...
Symposium Papers - Respiratory Care
... British chemist, Ramsay. Later that year, Swedish chemists determined its atomic weight.9 –11 In 1903 the gas from a geyser in Kansas was found to contain 12% of an unidentified gas. In 1907, Cady and McFarland of the University of Kansas published a paper that identified the gas as helium and showe ...
... British chemist, Ramsay. Later that year, Swedish chemists determined its atomic weight.9 –11 In 1903 the gas from a geyser in Kansas was found to contain 12% of an unidentified gas. In 1907, Cady and McFarland of the University of Kansas published a paper that identified the gas as helium and showe ...
Advanced Placement Chemistry
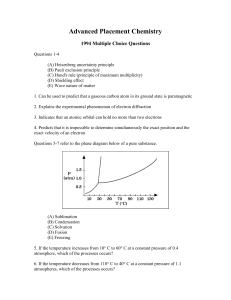
... (E) Wave nature of matter 1. Can be used to predict that a gaseous carbon atom in its ground state is paramagnetic 2. Explains the experimental phenomenon of electron diffraction 3. Indicates that an atomic orbital can hold no more than two electrons 4. Predicts that it is impossible to determine si ...
... (E) Wave nature of matter 1. Can be used to predict that a gaseous carbon atom in its ground state is paramagnetic 2. Explains the experimental phenomenon of electron diffraction 3. Indicates that an atomic orbital can hold no more than two electrons 4. Predicts that it is impossible to determine si ...
Ch06 BalancingChemRxns
... Electrical current through water. Electrolysis of water into its elements. 1. Write the skeleton equation ...
... Electrical current through water. Electrolysis of water into its elements. 1. Write the skeleton equation ...
makeup6
... 66. When propanol, CH3CH2CH2OH, is oxidized to propanoic acid, CH3CH2COOH, what is the number of electrons on the right side of the half-reaction? (A) 2 (B) 3 (C) 4 (D) 5 67. What is the [Cu2+] in the cell Zn / Zn2+ (0.05 M) // Cu2+ (X M) / Cu if the cell voltage is 1.03 V? (A) 0.12 M (B) 0.0002 M ( ...
... 66. When propanol, CH3CH2CH2OH, is oxidized to propanoic acid, CH3CH2COOH, what is the number of electrons on the right side of the half-reaction? (A) 2 (B) 3 (C) 4 (D) 5 67. What is the [Cu2+] in the cell Zn / Zn2+ (0.05 M) // Cu2+ (X M) / Cu if the cell voltage is 1.03 V? (A) 0.12 M (B) 0.0002 M ( ...
1. Explain electrophile and nucleophile. 2. Explain
... 37. When an alkali metal dissolves in liquid ammonia the solutions can acquire different colours.Explain the reasons for this type of colour change. 38. The solubility product of AgCl is 1.5*10-10.Predict whether there will be any precipitation by mixing some of 0.01M NaCl and 50 ml of 0.01 M AgNO3 ...
... 37. When an alkali metal dissolves in liquid ammonia the solutions can acquire different colours.Explain the reasons for this type of colour change. 38. The solubility product of AgCl is 1.5*10-10.Predict whether there will be any precipitation by mixing some of 0.01M NaCl and 50 ml of 0.01 M AgNO3 ...
Compulsory textbook Recommended textbooks Topics of the first
... Terms used in titrimetry Titration curves – plot the reagent volume on the horizontal axis and some function of the analyte on the vertical axis; the equivalence point can be read off the titration curve; it can either be sigmoidal or linear segment curve. Indicators – they are added to the analyte ...
... Terms used in titrimetry Titration curves – plot the reagent volume on the horizontal axis and some function of the analyte on the vertical axis; the equivalence point can be read off the titration curve; it can either be sigmoidal or linear segment curve. Indicators – they are added to the analyte ...
Slides for lecture 7 - Aleksey Kocherzhenko
... The first law of thermodynamics requires that if a forward reaction is exothermic, the reverse reaction must be endothermic:" ...
... The first law of thermodynamics requires that if a forward reaction is exothermic, the reverse reaction must be endothermic:" ...
MC94 - Southchemistry.com
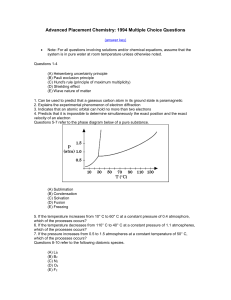
... (E) Wave nature of matter 1. Can be used to predict that a gaseous carbon atom in its ground state is paramagnetic 2. Explains the experimental phenomenon of electron diffraction 3. Indicates that an atomic orbital can hold no more than two electrons 4. Predicts that it is impossible to determine si ...
... (E) Wave nature of matter 1. Can be used to predict that a gaseous carbon atom in its ground state is paramagnetic 2. Explains the experimental phenomenon of electron diffraction 3. Indicates that an atomic orbital can hold no more than two electrons 4. Predicts that it is impossible to determine si ...
solutions - UMass Chemistry
... 14. (10 pts) Ammonia can break down into its elemental components as shown. If 0.25 mol ammonia contained in a flask at T = 25 ºC and P = 0.5 atm is completely converted to N2 and H2, what will the pressure in the flask be if the volume of the flask and T do not change? 2 NH3 (g) → ...
... 14. (10 pts) Ammonia can break down into its elemental components as shown. If 0.25 mol ammonia contained in a flask at T = 25 ºC and P = 0.5 atm is completely converted to N2 and H2, what will the pressure in the flask be if the volume of the flask and T do not change? 2 NH3 (g) → ...
Advanced Placement Chemistry
... (A) 1 (B) 2 (C) 3 (D) 4 (E) 6 43. A sample of 61.8 g of H3BO3, a weak acid is dissolvedin 1,000 g of water to make a 1.0molal solution. Which of the following would be the best procedure to determine to molarity of the solution? (Assume no additional information is available.) (A) Titration of the s ...
... (A) 1 (B) 2 (C) 3 (D) 4 (E) 6 43. A sample of 61.8 g of H3BO3, a weak acid is dissolvedin 1,000 g of water to make a 1.0molal solution. Which of the following would be the best procedure to determine to molarity of the solution? (Assume no additional information is available.) (A) Titration of the s ...
AP `99 Multiple Choice
... (C) Determination of the boiling point of the solution (D) Measurement of the total volume of the solution (E) Measurement of the specific heat of the solution 44. A rigid metal tank contains oxygen gas. Which of the following applies to the gas in the tank when additional oxygen is added at constan ...
... (C) Determination of the boiling point of the solution (D) Measurement of the total volume of the solution (E) Measurement of the specific heat of the solution 44. A rigid metal tank contains oxygen gas. Which of the following applies to the gas in the tank when additional oxygen is added at constan ...
1999 Advanced Placement Chemistry Exam
... (C) Determination of the boiling point of the solution (D) Measurement of the total volume of the solution (E) Measurement of the specific heat of the solution 44. A rigid metal tank contains oxygen gas. Which of the following applies to the gas in the tank when additional oxygen is added at constan ...
... (C) Determination of the boiling point of the solution (D) Measurement of the total volume of the solution (E) Measurement of the specific heat of the solution 44. A rigid metal tank contains oxygen gas. Which of the following applies to the gas in the tank when additional oxygen is added at constan ...
Elements compounds and mixtures
... always same. The properties of new compound are very different from those of the element in it. Cannot easily be separated into its elements. ...
... always same. The properties of new compound are very different from those of the element in it. Cannot easily be separated into its elements. ...
Chapter 1
... U = q + w = (-82) + (-622) = -704 kJ We see that the person ‘s internal energy falls by 704 kJ. 2). An electric battery is charged by supplying 250 kJ of energy to it as electrical work, but in the process it loses 25 kJ of energy as heat to the surroundings. What is the change in internal energy o ...
... U = q + w = (-82) + (-622) = -704 kJ We see that the person ‘s internal energy falls by 704 kJ. 2). An electric battery is charged by supplying 250 kJ of energy to it as electrical work, but in the process it loses 25 kJ of energy as heat to the surroundings. What is the change in internal energy o ...
Gas chromatography

Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture (the relative amounts of such components can also be determined). In some situations, GC may help in identifying a compound. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.In gas chromatography, the mobile phase (or ""moving phase"") is a carrier gas, usually an inert gas such as helium or an unreactive gas such as nitrogen. The stationary phase is a microscopic layer of liquid or polymer on an inert solid support, inside a piece of glass or metal tubing called a column (a homage to the fractionating column used in distillation). The instrument used to perform gas chromatography is called a gas chromatograph (or ""aerograph"", ""gas separator"").The gaseous compounds being analyzed interact with the walls of the column, which is coated with a stationary phase. This causes each compound to elute at a different time, known as the retention time of the compound. The comparison of retention times is what gives GC its analytical usefulness.Gas chromatography is in principle similar to column chromatography (as well as other forms of chromatography, such as HPLC, TLC), but has several notable differences. First, the process of separating the compounds in a mixture is carried out between a liquid stationary phase and a gas mobile phase, whereas in column chromatography the stationary phase is a solid and the mobile phase is a liquid. (Hence the full name of the procedure is ""Gas–liquid chromatography"", referring to the mobile and stationary phases, respectively.) Second, the column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled, whereas column chromatography (typically) has no such temperature control. Finally, the concentration of a compound in the gas phase is solely a function of the vapor pressure of the gas.Gas chromatography is also similar to fractional distillation, since both processes separate the components of a mixture primarily based on boiling point (or vapor pressure) differences. However, fractional distillation is typically used to separate components of a mixture on a large scale, whereas GC can be used on a much smaller scale (i.e. microscale).Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Strictly speaking, GLPC is the most correct terminology, and is thus preferred by many authors.