Nuclear Chemistry PowerPoint
... reactions. Although many people think of the sun as a large fireball, the sun (and all stars) are actually enormous fusion reactors. Stars are primarily gigantic balls of hydrogen gas under tremendous pressure due to gravitational forces. Hydrogen molecules are fused into helium and heavier elements ...
... reactions. Although many people think of the sun as a large fireball, the sun (and all stars) are actually enormous fusion reactors. Stars are primarily gigantic balls of hydrogen gas under tremendous pressure due to gravitational forces. Hydrogen molecules are fused into helium and heavier elements ...
Beryllium isotopes in geochronology Cosmogenic Be and Be
... processes. In some situations, it is possible to estimate “exposure ages” for rocks in eroding terrains. By comparing measured 10Be concentrations with estimated rates of in place cosmogenic 10Be production, the rate of rock erosion and formation of canyons and other geologic features can be determi ...
... processes. In some situations, it is possible to estimate “exposure ages” for rocks in eroding terrains. By comparing measured 10Be concentrations with estimated rates of in place cosmogenic 10Be production, the rate of rock erosion and formation of canyons and other geologic features can be determi ...
Lecture
... This is the story of a mysterious science which entered history at the onset of major transitions in science, and itself triggered these changes… From its modest beginnings as a minor phenomenon, radioactivity quickly developed into a major research field… The radioactive decays of naturally occur ...
... This is the story of a mysterious science which entered history at the onset of major transitions in science, and itself triggered these changes… From its modest beginnings as a minor phenomenon, radioactivity quickly developed into a major research field… The radioactive decays of naturally occur ...
Phys 282 EXP 8
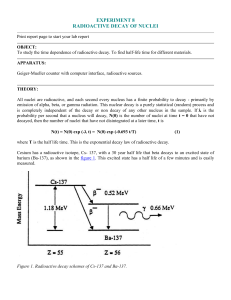
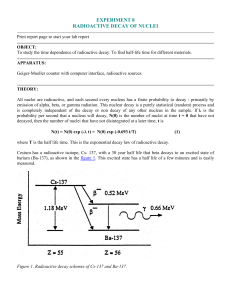
... To study the time dependence of radioactive decay. To find half-life time for different materials. APPARATUS: Geiger-Mueller counter with computer interface, radioactive sources. ...
... To study the time dependence of radioactive decay. To find half-life time for different materials. APPARATUS: Geiger-Mueller counter with computer interface, radioactive sources. ...
experiment 8 radioactive decay of nuclei
... To study the time dependence of radioactive decay. To find half-life time for different materials. APPARATUS: Geiger-Mueller counter with computer interface, radioactive sources. THEORY: All nuclei are radioactive, and each second every nucleus has a finite probability to decay - primarily by emissi ...
... To study the time dependence of radioactive decay. To find half-life time for different materials. APPARATUS: Geiger-Mueller counter with computer interface, radioactive sources. THEORY: All nuclei are radioactive, and each second every nucleus has a finite probability to decay - primarily by emissi ...
Chapter 12 –Radioactivity
... stable. • A few naturally occurring isotopes and all of the man-made isotopes are unstable. • Unstable isotopes can become stable by releasing different types of particles. • This process is called radioactive decay and the elements which undergo this process are called radioisotopes/radionuclides. ...
... stable. • A few naturally occurring isotopes and all of the man-made isotopes are unstable. • Unstable isotopes can become stable by releasing different types of particles. • This process is called radioactive decay and the elements which undergo this process are called radioisotopes/radionuclides. ...
12 · Nuclear Chemistry
... 11. True/False: The rate of radioactivity slows down over time. True 12. In the video, 16 atoms of Radon-212 are shown. The half-life is 24 minutes. Fill in this data chart. ...
... 11. True/False: The rate of radioactivity slows down over time. True 12. In the video, 16 atoms of Radon-212 are shown. The half-life is 24 minutes. Fill in this data chart. ...
Acrobat - chemmybear.com
... 1. Nuclear radiation has been part of the human experience since ________________________. 2. In the mid-19th century, the scientist __________ ______________, experimented with a glass tube in which the air was excited by electricity. 3. Cathode rays are deflected by both _____________ fields and _ ...
... 1. Nuclear radiation has been part of the human experience since ________________________. 2. In the mid-19th century, the scientist __________ ______________, experimented with a glass tube in which the air was excited by electricity. 3. Cathode rays are deflected by both _____________ fields and _ ...
Word - chemmybear.com
... 1. Nuclear radiation has been part of the human experience since ________________________. 2. In the mid-19th century, the scientist __________ ______________, experimented with a glass tube in which the air was excited by electricity. 3. Cathode rays are deflected by both _____________ fields and _ ...
... 1. Nuclear radiation has been part of the human experience since ________________________. 2. In the mid-19th century, the scientist __________ ______________, experimented with a glass tube in which the air was excited by electricity. 3. Cathode rays are deflected by both _____________ fields and _ ...
Word - chemmybear.com
... 1. Nuclear radiation has been part of the human experience since ________________________. 2. In the mid-19th century, the scientist __________ ______________, experimented with a glass tube in which the air was excited by electricity. 3. Cathode rays are deflected by both _____________ fields and _ ...
... 1. Nuclear radiation has been part of the human experience since ________________________. 2. In the mid-19th century, the scientist __________ ______________, experimented with a glass tube in which the air was excited by electricity. 3. Cathode rays are deflected by both _____________ fields and _ ...
Chapter 21 Powerpoint: Nuclear Chemistry
... start with a sample of 5.00 g, how much of it will still be left after 93 hours? ...
... start with a sample of 5.00 g, how much of it will still be left after 93 hours? ...
Types of Radiation
... Students know how to relate the position of an element in the periodic table to its atomic number and atomic mass. (1a) Students know the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions. The change in mass (calculated by E = mc ...
... Students know how to relate the position of an element in the periodic table to its atomic number and atomic mass. (1a) Students know the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions. The change in mass (calculated by E = mc ...
RaLa Experiment

The RaLa Experiment, or RaLa, was a series of tests during and after the Manhattan Project designed to study the behavior of converging shock waves to achieve the spherical implosion necessary for compression of the plutonium pit of the nuclear weapon. The experiment used significant amounts of a short-lived radioisotope lanthanum-140, a potent source of gamma radiation; the RaLa is a contraction of Radioactive Lanthanum. The method was proposed by Robert Serber and developed by a team led by the Italian experimental physicist Bruno Rossi.The tests were performed with 1/8 inch (3.2 mm) spheres of radioactive lanthanum, equal to about 100 curies (3.7 TBq) and later 1,000 Ci (37 TBq), located in the center of a simulated nuclear device. The explosive lenses were designed primarily using this series of tests. Some 254 tests were conducted between September 1944 and March 1962. In his history of the Los Alamos project, David Hawkins wrote: “RaLa became the most important single experiment affecting the final bomb design”.