Ch.9cellrespiration
... chain) is a series of reduction/oxidation reactions Enzymes embedded in mitochondrial membranes ...
... chain) is a series of reduction/oxidation reactions Enzymes embedded in mitochondrial membranes ...
Cellular Respiration NOTES
... Cellular respiration converts the energy stored in the bonds of the glucose into energy in ATP. Why is it important? – all living organisms need to convert the energy in the food they eat (or in the case of plants in the food they produce through photosynthesis) into a form of energy that is easy to ...
... Cellular respiration converts the energy stored in the bonds of the glucose into energy in ATP. Why is it important? – all living organisms need to convert the energy in the food they eat (or in the case of plants in the food they produce through photosynthesis) into a form of energy that is easy to ...
115 things you should know for the living environment
... 3. Enzymes are protein molecules that catalyze (help) chemical reactions. 4. The 3-dimensional shape of a molecule it important to its proper functioning. 5. Bacteria are prokaryotic cells, which lack a nucleus and,other membrane bound orgenelles. 6. The nucleus contains DNA in eukaryotic cells. 7. ...
... 3. Enzymes are protein molecules that catalyze (help) chemical reactions. 4. The 3-dimensional shape of a molecule it important to its proper functioning. 5. Bacteria are prokaryotic cells, which lack a nucleus and,other membrane bound orgenelles. 6. The nucleus contains DNA in eukaryotic cells. 7. ...
Biology Top 101 - Magnolia High School
... • An organism with a gene from another source • used to improve food supply, research, and healthcare ...
... • An organism with a gene from another source • used to improve food supply, research, and healthcare ...
Midterm Exam: 2000-2001
... first within the observed organelle? A. Light to chemical C. Heat to electrical B. ATP to light D. Chemical to chemical 26. A protein in the cell membrane changed its shape to move sodium and potassium ions against their concentration gradients. Which molecule was most likely used by the protein as ...
... first within the observed organelle? A. Light to chemical C. Heat to electrical B. ATP to light D. Chemical to chemical 26. A protein in the cell membrane changed its shape to move sodium and potassium ions against their concentration gradients. Which molecule was most likely used by the protein as ...
Cell Standards
... the reaction equilibrium and the activities of enzymes depend on the temperature, ionic conditions, and the pH of the surroundings. Almost all enzymes are protein catalysts made by living organisms. Enzymes speed up favorable (spontaneous) reactions by reducing the activation energy required for the ...
... the reaction equilibrium and the activities of enzymes depend on the temperature, ionic conditions, and the pH of the surroundings. Almost all enzymes are protein catalysts made by living organisms. Enzymes speed up favorable (spontaneous) reactions by reducing the activation energy required for the ...
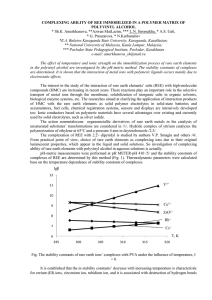
COMPLEXING ABILITY OF REE IMMOBILIZED IN A POLYMER
... according to /7/ the tendency to complexes’ formation is reduced in this line. However, the line changed may happen at low temperatures due to the influence of the solvent. It is seen on the example of trivalent yttrium, as well which at 298 K, more susceptible to hydration, and hence forms outer-sp ...
... according to /7/ the tendency to complexes’ formation is reduced in this line. However, the line changed may happen at low temperatures due to the influence of the solvent. It is seen on the example of trivalent yttrium, as well which at 298 K, more susceptible to hydration, and hence forms outer-sp ...
6O2 + C6H12O6 ------------------------
... b. _____________________ is broken down in to 2 molecules of pyruvate c. Uses ________ ATP to start, then makes ______ ATP, so net gain= ____________ d. Uses NAD+ and FAD which are coenzymes made from Vitamin C III. Aerobic Respiration: Occurs when oxygen ______ available. a. Occurs in _____________ ...
... b. _____________________ is broken down in to 2 molecules of pyruvate c. Uses ________ ATP to start, then makes ______ ATP, so net gain= ____________ d. Uses NAD+ and FAD which are coenzymes made from Vitamin C III. Aerobic Respiration: Occurs when oxygen ______ available. a. Occurs in _____________ ...
The Proton Motive Force
... Energy-requiring metabolic reactions Nutrients Supply of monomers (or precursors of) required by cells for growth Macronutrients Nutrients required in large amounts Carbon Required by all cells Typical bacterial cell ~50% carbon (by dry weight) Major element in all classes of macromolecules Nitrogen ...
... Energy-requiring metabolic reactions Nutrients Supply of monomers (or precursors of) required by cells for growth Macronutrients Nutrients required in large amounts Carbon Required by all cells Typical bacterial cell ~50% carbon (by dry weight) Major element in all classes of macromolecules Nitrogen ...
BIO 101 Chapter 1 Lecture Notes * WHAT IS LIFE?
... BIO 101 Chapter 1 Lecture Notes – WHAT IS LIFE? I. ...
... BIO 101 Chapter 1 Lecture Notes – WHAT IS LIFE? I. ...
ANAEROBIC NITROGEN FIXERS ON MARS. B. G. Lewis, Dept. of
... mass % [1]; molybdenum and vanadium are possibly present, estimated to be about 1.7 ppm and 162 ppm, respectively [6], within the range of their occurrence in terrestrial soils. Sulfur is present at about 5 mass % (expressed as SO3) in the “soil” from Pathfinder data [1]. Sulfite reduction is more t ...
... mass % [1]; molybdenum and vanadium are possibly present, estimated to be about 1.7 ppm and 162 ppm, respectively [6], within the range of their occurrence in terrestrial soils. Sulfur is present at about 5 mass % (expressed as SO3) in the “soil” from Pathfinder data [1]. Sulfite reduction is more t ...
2008 CELL BIOLOGY – TRAINING HANDOUT
... Introduction - Types of chemical reactions Endergonic: require energy Exergoinc: give off energy Catabolism: reactions breakdown large molecules and produce small molecules; Ex. Digestion of protein into amino acids Anabolism: reactions requiring energy to make large molecules from small mol ...
... Introduction - Types of chemical reactions Endergonic: require energy Exergoinc: give off energy Catabolism: reactions breakdown large molecules and produce small molecules; Ex. Digestion of protein into amino acids Anabolism: reactions requiring energy to make large molecules from small mol ...
+ 2 HCL(aq) CaCl2(aq) + H2O(l) + CO2(g)
... atoms of these elements. Oxidation Number: positive or negative number on the periodic table that indicates how many electrons an element has gained, lost or shared when bonding with another element. Polyatomic Atom: A compound with two or more elements. Law of Conservation of Mass: states that matt ...
... atoms of these elements. Oxidation Number: positive or negative number on the periodic table that indicates how many electrons an element has gained, lost or shared when bonding with another element. Polyatomic Atom: A compound with two or more elements. Law of Conservation of Mass: states that matt ...
Ch. 27 - ltcconline.net
... photoheterotrophs - use light for energy but obtain carbon in organic form (found in a number of marine prokaryotes), chemoheterotrophs - must consume energy and nutrients from surroundings -which of these groups do most eukaryotes fall into? A. Metabolic Relationships to oxygen 1. Obligate aerobes ...
... photoheterotrophs - use light for energy but obtain carbon in organic form (found in a number of marine prokaryotes), chemoheterotrophs - must consume energy and nutrients from surroundings -which of these groups do most eukaryotes fall into? A. Metabolic Relationships to oxygen 1. Obligate aerobes ...
LIST203 2008 spring Microbiology Page 1 Page 1
... 28 What is episome? (2 points) ( a plasmid able to integrate into chromosome and replicated with chromosome ) 29 What is the function of Lipopolysaccharides (LPSs)? What are three parts consisting of LPS? (5 points) (giving negative charge on surface, stabilizing outer membrane, eliciting immune res ...
... 28 What is episome? (2 points) ( a plasmid able to integrate into chromosome and replicated with chromosome ) 29 What is the function of Lipopolysaccharides (LPSs)? What are three parts consisting of LPS? (5 points) (giving negative charge on surface, stabilizing outer membrane, eliciting immune res ...
Plasma membrane
... The plasma membrane or bacterial cytoplasmic membrane is composed of a phospholipid bilayer and thus has all of the general functions of a cell membrane such as acting as a permeability barrier for most molecules and serving as the location for the transport of molecules into the cell. In addition t ...
... The plasma membrane or bacterial cytoplasmic membrane is composed of a phospholipid bilayer and thus has all of the general functions of a cell membrane such as acting as a permeability barrier for most molecules and serving as the location for the transport of molecules into the cell. In addition t ...
Carbon Cycle
... 2. Animals and plants cannot directly use all the nitrogen found in our ________________. 3. Only special bacteria can directly use nitrogen in our atmosphere and “fix” it so other organisms can benefit. These bacteria are called ____________-_________ bacteria. 4. Higher organisms use nitrogen to m ...
... 2. Animals and plants cannot directly use all the nitrogen found in our ________________. 3. Only special bacteria can directly use nitrogen in our atmosphere and “fix” it so other organisms can benefit. These bacteria are called ____________-_________ bacteria. 4. Higher organisms use nitrogen to m ...
Lecture 17.The d-Block Elements.General properties
... • Fourth-period d-block elements form ionic bonds with somewhat less ionic character than do the metals of the s-block. • Lower oxidation states (+2, +3) usually correspond to ionic character. • For Co through Zn, relative energies of the 4s and 3d subshells are such that few (or no) 3d electrons ar ...
... • Fourth-period d-block elements form ionic bonds with somewhat less ionic character than do the metals of the s-block. • Lower oxidation states (+2, +3) usually correspond to ionic character. • For Co through Zn, relative energies of the 4s and 3d subshells are such that few (or no) 3d electrons ar ...
Biosynthesis of Essential Amino Acids
... Written by Harold B. White 1. It has been shown in many instances that enzymes catalyzing chemically similar reactions (e.g. malate and lactate dehydrogenases) have amazingly similar tertiary structures which suggests they evolved from a common ancestral protein. Likewise, chemical similarity in seq ...
... Written by Harold B. White 1. It has been shown in many instances that enzymes catalyzing chemically similar reactions (e.g. malate and lactate dehydrogenases) have amazingly similar tertiary structures which suggests they evolved from a common ancestral protein. Likewise, chemical similarity in seq ...
Evolution of metal ions in biological systems
Evolution of Metal Ions in Biological Systems refers to the incorporation of metallic ions into living organisms and how it has changed over time. Metal ions have been associated with biological systems for billions of years, but only in the last century have scientists began to truly appreciate the scale of their influence. Major (iron, manganese, magnesium and zinc) and minor (copper, cobalt, nickel, molybdenum, tungsten) metal ions have symbiotically aligned with living organisms and the associated complexes have evolved over time.