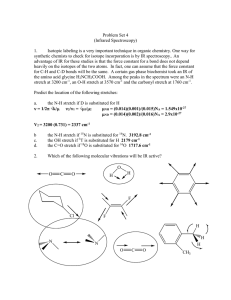
GQ2613291336
... distilled water at 298K, and the conductivity cell was fitted with a rubber stopper and the conductivity readings from conductivity meter (C) were recorded (the conductivity of distilled water as a blank was excluded). The other neck of the flask (A) connected by a rubber tube to the flask (I) which ...
... distilled water at 298K, and the conductivity cell was fitted with a rubber stopper and the conductivity readings from conductivity meter (C) were recorded (the conductivity of distilled water as a blank was excluded). The other neck of the flask (A) connected by a rubber tube to the flask (I) which ...
2840 GCC 16pp
... The Shielded CO Reactor For the determination of δ18O, the analyte must not contact the ceramic tube which is used to protect against air, and for stability. The pyrolysis takes place in an inert platinum inlay. Due to the catalytic properties of the platinum, the reaction can be performed at 1280 ° ...
... The Shielded CO Reactor For the determination of δ18O, the analyte must not contact the ceramic tube which is used to protect against air, and for stability. The pyrolysis takes place in an inert platinum inlay. Due to the catalytic properties of the platinum, the reaction can be performed at 1280 ° ...
Isotopes of Volatile Organic Compounds: An Emerging Approach for
... VOCs, and halogenated VOCs are ubiquitous trace gases that have important impacts on atmospheric chemistry in both the troposphere and the stratosphere. VOCs combine with oxides of nitrogen to produce O3, contribute to aerosol growth and thus impact atmospheric radiative processes and visibility, su ...
... VOCs, and halogenated VOCs are ubiquitous trace gases that have important impacts on atmospheric chemistry in both the troposphere and the stratosphere. VOCs combine with oxides of nitrogen to produce O3, contribute to aerosol growth and thus impact atmospheric radiative processes and visibility, su ...
W. M. White Geochemistry Chapter 9: Stable Isotopes Chapter 9
... (1) They have low atomic mass. (2) The relative mass difference between their isotopes is large. (3) They form bonds with a high degree of covalent character. (4) The elements exist in more than one oxidation state (C, N, and S), form a wide variety of compounds (O), or are important constituents of ...
... (1) They have low atomic mass. (2) The relative mass difference between their isotopes is large. (3) They form bonds with a high degree of covalent character. (4) The elements exist in more than one oxidation state (C, N, and S), form a wide variety of compounds (O), or are important constituents of ...
Get Reprint - McMaster Chemistry
... silicon, to yield a σ-bonded complex which collapses to (alkoxysilane) product by proton transfer from oxygen to carbon.5 Various product15,16b,17 and kinetic16,17 studies have lent support to the general features of this mechanism but have allowed it to be refined considerably. In particular, kinet ...
... silicon, to yield a σ-bonded complex which collapses to (alkoxysilane) product by proton transfer from oxygen to carbon.5 Various product15,16b,17 and kinetic16,17 studies have lent support to the general features of this mechanism but have allowed it to be refined considerably. In particular, kinet ...
The effect of confinement on chemical reactions
... significantly higher density than the bulk phase. The combination of these two factors naturally causes an enhancement of the equilibrium yield, as the increased density of the adsorbed phase displaces the equilibrium to the side with a lower number of moles. It would be interesting to consider now ...
... significantly higher density than the bulk phase. The combination of these two factors naturally causes an enhancement of the equilibrium yield, as the increased density of the adsorbed phase displaces the equilibrium to the side with a lower number of moles. It would be interesting to consider now ...
Even-Odd Effect of 35Cl Quadrupole Coupling
... This even-odd effect was also found along the c-axis (Fig. 3); as the carbon number increased and alternated between even and odd, the increments in the c-length were smaller than the length of the CH 2 group. The packings of CxHCl with odd values of x were tighter than those with even values of x. ...
... This even-odd effect was also found along the c-axis (Fig. 3); as the carbon number increased and alternated between even and odd, the increments in the c-length were smaller than the length of the CH 2 group. The packings of CxHCl with odd values of x were tighter than those with even values of x. ...
The Acid Hydrolysis Mechanism of Acetals Catalyzed
... formalism (including the mechanism for orthoformate hydrolysis in 1) in which a substrate binding pre-equilibrium occurs before the rate limiting step of the reaction. Having established the importance of the hydrophobic effect for substrate encapsulation in the catalysis, the kinetic order for [sub ...
... formalism (including the mechanism for orthoformate hydrolysis in 1) in which a substrate binding pre-equilibrium occurs before the rate limiting step of the reaction. Having established the importance of the hydrophobic effect for substrate encapsulation in the catalysis, the kinetic order for [sub ...
Physical Organic Chemistry
... Elimination reaction: to eliminate two atoms, two groups, or one atom and one group without substituted with another atom or group. The elimination of HX molecule from alkyl derivatives. While X is a halogen or ester… etc. the hydrogen atom on adjacent carbon with X Elimination reactions and n ...
... Elimination reaction: to eliminate two atoms, two groups, or one atom and one group without substituted with another atom or group. The elimination of HX molecule from alkyl derivatives. While X is a halogen or ester… etc. the hydrogen atom on adjacent carbon with X Elimination reactions and n ...
On the Importance of Prereactive Complexes in
... Ea ) E2 - E-1 ) (ETS - EP-R) - (ER - EP-R) ) ETS - ER ...
... Ea ) E2 - E-1 ) (ETS - EP-R) - (ER - EP-R) ) ETS - ER ...
Kinetics in the Study of Organic Reaction Mechanisms
... chemical transformations occur. That is, chemistry is concerned with the mechanisms of reactions as well as with a knowledge of their starting materials and products. There are a number of reasons for this preoccupation with reaction mechanisms. A knowledge of mechanisms aids the synthetic chemist i ...
... chemical transformations occur. That is, chemistry is concerned with the mechanisms of reactions as well as with a knowledge of their starting materials and products. There are a number of reasons for this preoccupation with reaction mechanisms. A knowledge of mechanisms aids the synthetic chemist i ...
Nucleophilic substitution at saturated carbon
... different in size, another factor comes into play—the polarizability of the atom. Because the electrons are farther away in the larger atom, they are not held as tightly and can, therefore, move more freely toward a positive charge. As a result, the electrons are able to overlap from farther away wi ...
... different in size, another factor comes into play—the polarizability of the atom. Because the electrons are farther away in the larger atom, they are not held as tightly and can, therefore, move more freely toward a positive charge. As a result, the electrons are able to overlap from farther away wi ...
Document
... Fractionation during a chemical reaction is created at the reactive center(s) of the reaction The primary isotope effect is bigger than the secundary isotopeo effect The apparent fractionation decreases with increasing molecule size since fractionation at the reactive center is „diluted“ by uninvolv ...
... Fractionation during a chemical reaction is created at the reactive center(s) of the reaction The primary isotope effect is bigger than the secundary isotopeo effect The apparent fractionation decreases with increasing molecule size since fractionation at the reactive center is „diluted“ by uninvolv ...
PowerPoint **
... α-Elimination: Generation of Carbene Defination: A carbene is a divalent carbon species link to two adjacent groups by covalent bonds, possessing two nonbonded electrons and six valence electrons. Preparation of carbenes a. ...
... α-Elimination: Generation of Carbene Defination: A carbene is a divalent carbon species link to two adjacent groups by covalent bonds, possessing two nonbonded electrons and six valence electrons. Preparation of carbenes a. ...
Stable Isotopes in Foraminiferal Carbonate
... have evolved from the original design by A.O. Nier, and for carbonate analysis, a dual inlet magnetic sector mass spectrometer (or isotope ratio mass spectrometer, IRMS) is most commonly used. The major components of the IRMS are: (1) the analyser consisting of ion source, flight tube and collector ...
... have evolved from the original design by A.O. Nier, and for carbonate analysis, a dual inlet magnetic sector mass spectrometer (or isotope ratio mass spectrometer, IRMS) is most commonly used. The major components of the IRMS are: (1) the analyser consisting of ion source, flight tube and collector ...
PDF w - ACS Publications - American Chemical Society
... Continuous Photolysis and KIE Measurements. Continuous photolysis was carried out in NMR tubes sealed with J. Young valves, and reaction progress was monitored by 1H NMR spectroscopy. NMR spectra were obtained on a Bruker Avance III 400 FT NMR spectrometer (400 MHz for 1H). Solutions of [(η5-iPr4C5H ...
... Continuous Photolysis and KIE Measurements. Continuous photolysis was carried out in NMR tubes sealed with J. Young valves, and reaction progress was monitored by 1H NMR spectroscopy. NMR spectra were obtained on a Bruker Avance III 400 FT NMR spectrometer (400 MHz for 1H). Solutions of [(η5-iPr4C5H ...
Imbalanced tunneling ready states in alcohol dehydrogenase
... and EIE has been well applied to the mechanistic understanding of the reactions involving heavy atom bond changes,3,4,6–8 its application in the study of the light H particle transfer reactions has been challenging.9,10 Since the early 1980’s, 21 deuterium or tritium (H/D or H/T) KIEs for many H-tra ...
... and EIE has been well applied to the mechanistic understanding of the reactions involving heavy atom bond changes,3,4,6–8 its application in the study of the light H particle transfer reactions has been challenging.9,10 Since the early 1980’s, 21 deuterium or tritium (H/D or H/T) KIEs for many H-tra ...
Chromatographic Enrichment of Lithium Isotopes by Hydrous
... The chemistry of the hydrous metal oxides has been studied in a large number of scientific fields, especially, ion exchange separation, scavenging by sorption and coprecipitation, separation of mineral, nuclear fuels, antiperspirants, radioactive contamination, and scavenging of trace metals.1 The h ...
... The chemistry of the hydrous metal oxides has been studied in a large number of scientific fields, especially, ion exchange separation, scavenging by sorption and coprecipitation, separation of mineral, nuclear fuels, antiperspirants, radioactive contamination, and scavenging of trace metals.1 The h ...
UN1001: Section 11: Hydrogen Effects
... Hydride-forming metals are susceptible to H- embrittlement . . .e.g., Zr-alloy pressure tubes (in CANDUs) and fuel sheathing (in all water- cooled reactors) pick up hydrogen (or deuterium in heavy water ) by general corrosion. The hydrogen (D) migrates through the metal lattice to cool regions and t ...
... Hydride-forming metals are susceptible to H- embrittlement . . .e.g., Zr-alloy pressure tubes (in CANDUs) and fuel sheathing (in all water- cooled reactors) pick up hydrogen (or deuterium in heavy water ) by general corrosion. The hydrogen (D) migrates through the metal lattice to cool regions and t ...
CH 3 Br + Nu
... B) it involves the formation of the carbocation from elimination of a good leaving group C) a common competing reaction is rearrangement of a less stable carbocation to a more stable carbocation D) the loss of a proton by the carbocation is a fast step E) all of the above ...
... B) it involves the formation of the carbocation from elimination of a good leaving group C) a common competing reaction is rearrangement of a less stable carbocation to a more stable carbocation D) the loss of a proton by the carbocation is a fast step E) all of the above ...
Nucleophilic Substitution Reactions
... ■ The polarity in halogenoalkanes is due to the fact that the halogen atom is more electronegative than carbon, and so exserts a stronger pull on the shared electrons in the carbon-halogen bond. ■ As a result, the halogen gains a partial negative charge and the carbon gains a partial positive charge ...
... ■ The polarity in halogenoalkanes is due to the fact that the halogen atom is more electronegative than carbon, and so exserts a stronger pull on the shared electrons in the carbon-halogen bond. ■ As a result, the halogen gains a partial negative charge and the carbon gains a partial positive charge ...
O–H hydrogen bonding promotes H-atom transfer from a C–H bonds
... metal catalyzed C(sp3)–H activation—including, among others, Hartwig’s rhodium-catalyzed borylation of terminal methyl groups (4) and White’s iron-catalyzed oxidation of both secondary (2°) and tertiary (3°) aliphatic C–H bonds (5)—highlight the importance of catalyst structure on site selectivity. ...
... metal catalyzed C(sp3)–H activation—including, among others, Hartwig’s rhodium-catalyzed borylation of terminal methyl groups (4) and White’s iron-catalyzed oxidation of both secondary (2°) and tertiary (3°) aliphatic C–H bonds (5)—highlight the importance of catalyst structure on site selectivity. ...
Tiny filters, big news: Novel process uses graphene and
... techniques developed in their previous paper . "The without which we would have gained significant insight into the proton transport process – but most important was the ability to produce a large number of fully suspended one-atom-thick crystal applications would still be a long way ahead. To do so ...
... techniques developed in their previous paper . "The without which we would have gained significant insight into the proton transport process – but most important was the ability to produce a large number of fully suspended one-atom-thick crystal applications would still be a long way ahead. To do so ...