Durham Research Online
... 2-dimensional projection of the original 3-dimensional velocity distribution of the NO molecules onto the (vx , vz ) plane. Such a set-up allows the direct measurement of the absolute velocity of both the molecular beam and of the NO fragment molecules, which is vital in maximizing the efficiency of ...
... 2-dimensional projection of the original 3-dimensional velocity distribution of the NO molecules onto the (vx , vz ) plane. Such a set-up allows the direct measurement of the absolute velocity of both the molecular beam and of the NO fragment molecules, which is vital in maximizing the efficiency of ...
Bonding and Structure - Lesmahagow High School
... A covalent bond is the electrostatic force of attraction between positively charged nuclei and negatively charged outer electrons. In non-metal elements, e.g the diatomic elements, there is an equal sharing of electrons between atoms as they have the same electronegativity. ...
... A covalent bond is the electrostatic force of attraction between positively charged nuclei and negatively charged outer electrons. In non-metal elements, e.g the diatomic elements, there is an equal sharing of electrons between atoms as they have the same electronegativity. ...
PES Topography - Materials Computation Center
... This simple requirement is not met by truncated CI. • E should be additive for noninteracting systems • should be a product Exponential maps products to sums… ...
... This simple requirement is not met by truncated CI. • E should be additive for noninteracting systems • should be a product Exponential maps products to sums… ...
The Future is Noisy: The Role of Spatial Fluctuations
... E. coli is of the order of 1 mm, and the free volume in which the messenger molecules diffuse within the cell is ⬃1 mm3 [14]. Comparing to the vdW radius, we obtain p ⬃ 5 3 1025 . Note that for these numbers, the probability that none out of 100 molecules is within the IV, 共1 2 p兲100 艐 99.5%, is sti ...
... E. coli is of the order of 1 mm, and the free volume in which the messenger molecules diffuse within the cell is ⬃1 mm3 [14]. Comparing to the vdW radius, we obtain p ⬃ 5 3 1025 . Note that for these numbers, the probability that none out of 100 molecules is within the IV, 共1 2 p兲100 艐 99.5%, is sti ...
Principles of Spectroscopy
... anharmonic oscillator. The potential energy is then calculated by the Morse equation, and is asymmetric. The energy levels are no longer equally spaced, and are given by: Ev=(v + ½) h - (v + ½)2 xGl h where xGl is the anharmonicity constant. The anharmonic oscillator model allows for two imp ...
... anharmonic oscillator. The potential energy is then calculated by the Morse equation, and is asymmetric. The energy levels are no longer equally spaced, and are given by: Ev=(v + ½) h - (v + ½)2 xGl h where xGl is the anharmonicity constant. The anharmonic oscillator model allows for two imp ...
Low-energy scattering of molecules and ions in a magnetic field
... The essential difference between the current mechanism and sympathetic cooling from atoms (e.g., Ref. [27]) is that the collisions take place at longer range, which vastly increases the elastic collision rate while decreasing the rate for changing the internal state of the molecule. If ions and cold ...
... The essential difference between the current mechanism and sympathetic cooling from atoms (e.g., Ref. [27]) is that the collisions take place at longer range, which vastly increases the elastic collision rate while decreasing the rate for changing the internal state of the molecule. If ions and cold ...
Packing and Molecular Orientation of Alkanethiol Monolayers on
... not possible for the "racheting effect" to establish this special interlocking. Figure 7 shows how the best interaction energy (at the best tilt for this spacing) varies as a function of the lattice spacing. The minima near 4.24 A requires a nearly vertical orientation, and that near 4.95 A requires ...
... not possible for the "racheting effect" to establish this special interlocking. Figure 7 shows how the best interaction energy (at the best tilt for this spacing) varies as a function of the lattice spacing. The minima near 4.24 A requires a nearly vertical orientation, and that near 4.95 A requires ...
Photofragmentation-laser induced fluorescence: a
... A laboratory PF-LIF system consists of three major components: (1) one or two high energy pulsed dye laser systems; (2) a fluorescence chamber with associated detection optics; and (3) sampling electronics and data processing hardware. The use of either one or two dye lasers to provide the photolysi ...
... A laboratory PF-LIF system consists of three major components: (1) one or two high energy pulsed dye laser systems; (2) a fluorescence chamber with associated detection optics; and (3) sampling electronics and data processing hardware. The use of either one or two dye lasers to provide the photolysi ...
Molecular orbital methods in organic chemistry
... approximate wave functions used must be amenable to detailed interpretation and comparison among related molecules. For example, it is helpful if the calculated electron charge distribution can be easily and realistically divided into contributions of individual atoms which may then be compared with ...
... approximate wave functions used must be amenable to detailed interpretation and comparison among related molecules. For example, it is helpful if the calculated electron charge distribution can be easily and realistically divided into contributions of individual atoms which may then be compared with ...
Experiment 4 - Macalester College
... The three-dimensionality of a molecule is critically linked to its chemical and physical properties. For example, the bent geometry of water causes its polarity. The polarity of a molecule (or lack thereof) strongly influences its boiling point, freezing point, reactivity, and countless other proper ...
... The three-dimensionality of a molecule is critically linked to its chemical and physical properties. For example, the bent geometry of water causes its polarity. The polarity of a molecule (or lack thereof) strongly influences its boiling point, freezing point, reactivity, and countless other proper ...
Implementing the Theory of Sum Frequency Generation Vibrational
... Sum frequency generation vibrational spectroscopy (SFS) is a surface-specific technique that provides vibrational spectra of molecules at interfaces (1, 2). SFS relies on the non-linear optical phenomenon of sum frequency generation (SFG). SFG occurs when two pulsed laser beams, one of fixed visible ...
... Sum frequency generation vibrational spectroscopy (SFS) is a surface-specific technique that provides vibrational spectra of molecules at interfaces (1, 2). SFS relies on the non-linear optical phenomenon of sum frequency generation (SFG). SFG occurs when two pulsed laser beams, one of fixed visible ...
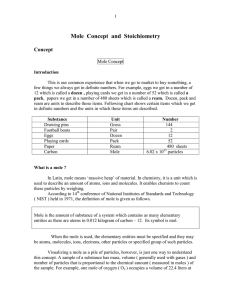
Mole Concept and Stoichiometry
... constructed that could determine if the sample was one atom over or under exactly 12 grams. If the first two requirements were met, it would take one million machines counting one million atoms each second more than 19,000 years to complete the task. So, practically it can be treated as impossible t ...
... constructed that could determine if the sample was one atom over or under exactly 12 grams. If the first two requirements were met, it would take one million machines counting one million atoms each second more than 19,000 years to complete the task. So, practically it can be treated as impossible t ...
Chapter 8 - Power Point Presentation
... bond, easier to break, making alkenes much more reactive than alkanes. This reaction occurs more readily ...
... bond, easier to break, making alkenes much more reactive than alkanes. This reaction occurs more readily ...
Intermolecular forces and molecules
... There are many topics included in this discussion and subtleties to the reasoning: o All of the molecules have van der Waal interactions, but it was only used to explain differences between the non-polar molecules. o The electronegativity for fluorine is much greater than hydrogen, but both CH4 and ...
... There are many topics included in this discussion and subtleties to the reasoning: o All of the molecules have van der Waal interactions, but it was only used to explain differences between the non-polar molecules. o The electronegativity for fluorine is much greater than hydrogen, but both CH4 and ...
Electronic Structure - Chemistry Teaching Resources
... the shape of an orbital. l = 0, 1, 2, and 3 (4 shapes) but we use letters for l (s, p, d and f). Usually we refer to the s, p, d and f-orbitals Magnetic Quantum Number, ml. This quantum number describes the orientation of orbitals of the same shape. The magnetic quantum number has integral values be ...
... the shape of an orbital. l = 0, 1, 2, and 3 (4 shapes) but we use letters for l (s, p, d and f). Usually we refer to the s, p, d and f-orbitals Magnetic Quantum Number, ml. This quantum number describes the orientation of orbitals of the same shape. The magnetic quantum number has integral values be ...