Time-resolved coherent anti-Stokes Raman scattering: imaging based on Raman free induction decay
... torily reproducing the characteristic features of the observed decay curve. The temporal resolution of the T-CARS microscope is given by the instrumental response function 共IRF兲 that has been independently measured by detecting the solely nonresonant coherent radiation originating from the glass sub ...
... torily reproducing the characteristic features of the observed decay curve. The temporal resolution of the T-CARS microscope is given by the instrumental response function 共IRF兲 that has been independently measured by detecting the solely nonresonant coherent radiation originating from the glass sub ...
Excited States, Lasers and Non-Linear Optics
... the CO2 symmetric stretch. • The lasing transition occurs between different rotational states of the anti-symmetric stretch at 2400 cm-1 and the other vibrational modes symmetric stretch at 1400 cm -1 and bend at 1300 cm -1. ...
... the CO2 symmetric stretch. • The lasing transition occurs between different rotational states of the anti-symmetric stretch at 2400 cm-1 and the other vibrational modes symmetric stretch at 1400 cm -1 and bend at 1300 cm -1. ...
Influence of Complex Exciton-Phonon Coupling on Optical
... (for organic dyes ∆ is in the order of 0.1 eV). The vibrational progression due to the high-energy modes with energies around 1.5 ∆ in the spectral density (see Fig. 1) is clearly visible. The considerable broadening of this progression mainly stems from the low-energy vibrations below 0.5 ∆ in the ...
... (for organic dyes ∆ is in the order of 0.1 eV). The vibrational progression due to the high-energy modes with energies around 1.5 ∆ in the spectral density (see Fig. 1) is clearly visible. The considerable broadening of this progression mainly stems from the low-energy vibrations below 0.5 ∆ in the ...
Week 8 – Intermolecular Forces
... (E) Cl2 has a greater electronegativity than Br2, therefore they have stronger dipole to dipole attractions. 15. What is the best reason for H2O having a higher boiling point as compared to H2S? (A) H2O is polar whereas H2S is nonpolar (B) H2O exhibits hydrogen bonding whereas H2S exhibits London (d ...
... (E) Cl2 has a greater electronegativity than Br2, therefore they have stronger dipole to dipole attractions. 15. What is the best reason for H2O having a higher boiling point as compared to H2S? (A) H2O is polar whereas H2S is nonpolar (B) H2O exhibits hydrogen bonding whereas H2S exhibits London (d ...
Physical Chemistry
... I wish to thank the editorial staff of Elsevier/Academic Press for their guidance and help during a rather long and complicated project, and also wish to thank Erica Ellison, who was a valuable consultant. I thank my wife, Ann, for her patience, love, and support during this project. ...
... I wish to thank the editorial staff of Elsevier/Academic Press for their guidance and help during a rather long and complicated project, and also wish to thank Erica Ellison, who was a valuable consultant. I thank my wife, Ann, for her patience, love, and support during this project. ...
Optics and Interferometry with Na2 Molecules
... distance between the gratings (Fig. 3). In addition to the interfering paths shown in Fig. 3, there are also interfering paths symmetric to the collimation axis as well as (small) contributions at larger offsets from the third grating involving the first and second order diffractions from the first ...
... distance between the gratings (Fig. 3). In addition to the interfering paths shown in Fig. 3, there are also interfering paths symmetric to the collimation axis as well as (small) contributions at larger offsets from the third grating involving the first and second order diffractions from the first ...
Relation between s-Polarized and p-Polarized Internal Reflection
... to deviate only slightly from that of a nonabsorbing medium.2 First, the film must be very thin. Second, the absorption coefficient R < 104 cm-1. The relation between the incident electric field components and the evanescent wave components at the surface of the IRE is given by eqs 1 and 2.1,2 ...
... to deviate only slightly from that of a nonabsorbing medium.2 First, the film must be very thin. Second, the absorption coefficient R < 104 cm-1. The relation between the incident electric field components and the evanescent wave components at the surface of the IRE is given by eqs 1 and 2.1,2 ...
A study of the structure and bonding of small aluminum oxide
... binding energy spectra are obtained by subtracting the kinetic energy spectrum from the photon energy. The energy resolution of our apparatus is better than 30 meV at 1 eV electron energy. Due to the dependence of resolution on electron energies for TOF type electron analyzers, various photon energi ...
... binding energy spectra are obtained by subtracting the kinetic energy spectrum from the photon energy. The energy resolution of our apparatus is better than 30 meV at 1 eV electron energy. Due to the dependence of resolution on electron energies for TOF type electron analyzers, various photon energi ...
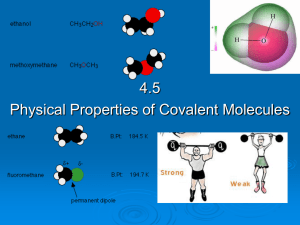
PowerPoint Chapter 14 - Preparatory Chemistry
... • whether a chemical bond is nonpolar covalent, polar covalent, or ionic. • which atom in a polar covalent bond is partial negative and which is partial positive. • which atom in an ionic bond forms the cation and which forms the anion. • which of two covalent bonds are more polar. ...
... • whether a chemical bond is nonpolar covalent, polar covalent, or ionic. • which atom in a polar covalent bond is partial negative and which is partial positive. • which atom in an ionic bond forms the cation and which forms the anion. • which of two covalent bonds are more polar. ...
Stoichiometry 2
... Step 3. Fill in the ladder grid with as much information as possible – this is typically supplied gram weights and molar mass data. Step 4. Convert g moles by ‘climbing’ down ladder(s) (g / M = moles). Remember: Molar masses are calculated for ONE molecular formula only. I.E. ignore any balancing ...
... Step 3. Fill in the ladder grid with as much information as possible – this is typically supplied gram weights and molar mass data. Step 4. Convert g moles by ‘climbing’ down ladder(s) (g / M = moles). Remember: Molar masses are calculated for ONE molecular formula only. I.E. ignore any balancing ...
Electron±electron correlations in carbon nanotubes
... electrons by 1, and M a magnetic internal transition where n stays constant. We note that at an external transition the spin is always increased, but between Coulomb peaks a hidden internal magnetic transition occurs which lowers the total spin of the tube. The spins of all incoming electrons can th ...
... electrons by 1, and M a magnetic internal transition where n stays constant. We note that at an external transition the spin is always increased, but between Coulomb peaks a hidden internal magnetic transition occurs which lowers the total spin of the tube. The spins of all incoming electrons can th ...
A unified picture of protein hydration: prediction of hydrodynamic
... tailed structures of proteins have been developed to calculate hydrodynamic properties w5᎐8x. It is time to re-examine the issue of hydrodynamic hydration. A number of techniques such as X-ray diffraction, nuclear magnetic resonance ŽNMR. spectroscopy, calorimety, infrared spectroscopy, and molecula ...
... tailed structures of proteins have been developed to calculate hydrodynamic properties w5᎐8x. It is time to re-examine the issue of hydrodynamic hydration. A number of techniques such as X-ray diffraction, nuclear magnetic resonance ŽNMR. spectroscopy, calorimety, infrared spectroscopy, and molecula ...
72KB
... bonded to three other C atoms in a 2-D or layered arrangement with weak intermolecular forces of attraction between the layers or sheets. In diamond, the covalent bonds between the carbon atoms are very strong and hold the atoms in place, making it difficult to break the bonds. Therefore diamond is ...
... bonded to three other C atoms in a 2-D or layered arrangement with weak intermolecular forces of attraction between the layers or sheets. In diamond, the covalent bonds between the carbon atoms are very strong and hold the atoms in place, making it difficult to break the bonds. Therefore diamond is ...
1 Janaky Narayanan PC 5213 AY 2004
... CD over the same wavelength range used to record an absorption spectrum. The resulting optical activity spectra are called ORD and CD spectra. If the sample contains only strongly allowed electronic transitions (such as *), the shape of the CD spectrum (often called a Cotton effect) is related ...
... CD over the same wavelength range used to record an absorption spectrum. The resulting optical activity spectra are called ORD and CD spectra. If the sample contains only strongly allowed electronic transitions (such as *), the shape of the CD spectrum (often called a Cotton effect) is related ...
Ultracold Triplet Molecules in the Rovibrational
... manner described in [24], we can map out the Bloch bands in momentum space. After 13 ms of ballistic expansion, the corresponding atomic distribution is recorded with standard absorption imaging (see left inset in Fig. 2). For our signals, we only count atoms in the central square zone, correspondin ...
... manner described in [24], we can map out the Bloch bands in momentum space. After 13 ms of ballistic expansion, the corresponding atomic distribution is recorded with standard absorption imaging (see left inset in Fig. 2). For our signals, we only count atoms in the central square zone, correspondin ...
24 Interferometry with Macromolecules: Quantum Paradigms Tested
... laser with a wavelength of below ∼ 100 nm. Then scattering of a laser photon by a fullerene would give us sufficient information about which way the molecule took through the diffraction grating. Therefore the environmental states would be orthogonal and the interference pattern should disappear. As a ...
... laser with a wavelength of below ∼ 100 nm. Then scattering of a laser photon by a fullerene would give us sufficient information about which way the molecule took through the diffraction grating. Therefore the environmental states would be orthogonal and the interference pattern should disappear. As a ...
Worksheet 3A on Molecules
... Of the species listed, only O3 and CO are polar. CO is polar due to the difference in electronegativity between O and C; O3 is polar because it has 3 RHED and one lone pair on the central atom. This lone pair is an area where negative charge is concentrated, so this results in the molecule having an ...
... Of the species listed, only O3 and CO are polar. CO is polar due to the difference in electronegativity between O and C; O3 is polar because it has 3 RHED and one lone pair on the central atom. This lone pair is an area where negative charge is concentrated, so this results in the molecule having an ...
nλ = dsinθ
... 1. Set the meter sticks on the end table at the base of the lamp. Be sure the two are exactly perpendicular to each other and that the stick is balanced at its 50 cm mark. 2. Put the grating on its support and place it on the optical bench so that it is about 100 cm from the mercury discharge tube. ...
... 1. Set the meter sticks on the end table at the base of the lamp. Be sure the two are exactly perpendicular to each other and that the stick is balanced at its 50 cm mark. 2. Put the grating on its support and place it on the optical bench so that it is about 100 cm from the mercury discharge tube. ...
Hyperfine Structure in the Rotational Spectrum of GaF: A
... The high-resolution pure rotational spectrum of GaF has been measured using a Balle–Flygare-type Fourier transform spectrometer. Improved nuclear quadrupolar coupling constants and rotational constants have been obtained along with the first reported fluorine spin–rotation constant for gallium fluor ...
... The high-resolution pure rotational spectrum of GaF has been measured using a Balle–Flygare-type Fourier transform spectrometer. Improved nuclear quadrupolar coupling constants and rotational constants have been obtained along with the first reported fluorine spin–rotation constant for gallium fluor ...