mobile protons on silica and alumina surfaces protones moviles
... The optical density of the continuous absorption increases with increasing basicity of the pyridine and decreases on substitution of bulky substituents in ortho position. This continuous absorption is strong evidence for the existence of mobile protons which may be formed by a transfer of protons fr ...
... The optical density of the continuous absorption increases with increasing basicity of the pyridine and decreases on substitution of bulky substituents in ortho position. This continuous absorption is strong evidence for the existence of mobile protons which may be formed by a transfer of protons fr ...
Molecular Beam Epitaxy
... (2 x 4) reconstructed terraces on vicinal GaAs(001) with straight As dimer and missing dimer rows, holes and islands. B-type steps are more ragged ...
... (2 x 4) reconstructed terraces on vicinal GaAs(001) with straight As dimer and missing dimer rows, holes and islands. B-type steps are more ragged ...
Tracing the Movements of Single Atoms and Molecules on Solid
... was very difficult to achieve, but it was concluded that a precision of about f0.5 8, could be achieved. The power of single atom tracking achieved its potential only when PC image digitizers become generally available. With a modern PC image digitizer and improved vacuum technology, not only the ad ...
... was very difficult to achieve, but it was concluded that a precision of about f0.5 8, could be achieved. The power of single atom tracking achieved its potential only when PC image digitizers become generally available. With a modern PC image digitizer and improved vacuum technology, not only the ad ...
Surface Tension
... (which is equivalent to N/m). For example, the amount of work required to create 1 m2 surface is about 72.8 103 J for water. Surface tension is defined as the force at right angle to any line of unit length in the surface. Therefore, surface tension = force/distance. It is expressed in N/m. T ...
... (which is equivalent to N/m). For example, the amount of work required to create 1 m2 surface is about 72.8 103 J for water. Surface tension is defined as the force at right angle to any line of unit length in the surface. Therefore, surface tension = force/distance. It is expressed in N/m. T ...
Lecture 35 (Slides) November 7
... • 1. A total of 3.00 g of liquid water was placed in an evacuated (initially) 12.0L container maintained at a temperature of 80.0 0C. Will a liquid/gas equilibrium be established? What piece of data is required to solve this problem? If no liquid/gas equilibrium is established, determine how much ad ...
... • 1. A total of 3.00 g of liquid water was placed in an evacuated (initially) 12.0L container maintained at a temperature of 80.0 0C. Will a liquid/gas equilibrium be established? What piece of data is required to solve this problem? If no liquid/gas equilibrium is established, determine how much ad ...
Tip Streaming from a Drop in the Presence of Surfactants
... flow conditions is of practical significance when immiscible fluids must be dispersed in each other to create emulsions, and in understanding the stability and rheology of emulsions once formed. Surfactants are added to the fluid mixtures to reduce the surface tension, reducing the work required to ...
... flow conditions is of practical significance when immiscible fluids must be dispersed in each other to create emulsions, and in understanding the stability and rheology of emulsions once formed. Surfactants are added to the fluid mixtures to reduce the surface tension, reducing the work required to ...
The Layer Model What is a Model? Empirical Models
... S(1− α ) = σ Tglass 4 both space and Boundary ...
... S(1− α ) = σ Tglass 4 both space and Boundary ...
Structure and stability of CaH2 surfaces
... [110], and [111] crystallographic orientations, respectively. The shaded area indicate (001), (110), and (111) lattice planes, respectively. Sticks connecting a Ca to its nearest neighbor H atoms. Two sets of symmetrically inequivalent hydrogens are denoted by Ha and Hb . CaHa bond lengths: δ1 = 2.2 ...
... [110], and [111] crystallographic orientations, respectively. The shaded area indicate (001), (110), and (111) lattice planes, respectively. Sticks connecting a Ca to its nearest neighbor H atoms. Two sets of symmetrically inequivalent hydrogens are denoted by Ha and Hb . CaHa bond lengths: δ1 = 2.2 ...
- Free Documents
... Lower Surface Energy Larger Contact Angle Poorer Wetting Low surface energy functional group Polymer chain molecules Surface reactive group Region of Lower Monomer Density Higher Surface Energy Smaller Contact Angle Better Wetting Fig. A wick structure was also used in the test performed against gra ...
... Lower Surface Energy Larger Contact Angle Poorer Wetting Low surface energy functional group Polymer chain molecules Surface reactive group Region of Lower Monomer Density Higher Surface Energy Smaller Contact Angle Better Wetting Fig. A wick structure was also used in the test performed against gra ...
Derivation of the BET and Langmuir Isotherms
... The BET isotherm is accurate when P/Po is between ∼0.05 and ∼0.3, pressures at which only a few complete layers have formed. At low pressures the BET isotherm does not do a good job at describing adsorption because in deriving the isotherm it was assumed that all the sites on the alumina surface are ...
... The BET isotherm is accurate when P/Po is between ∼0.05 and ∼0.3, pressures at which only a few complete layers have formed. At low pressures the BET isotherm does not do a good job at describing adsorption because in deriving the isotherm it was assumed that all the sites on the alumina surface are ...
Surface chemistry Surface chemistry deals with phenomena that
... (v) Diffusion of reaction products away from the catalyst’s surface. Thus, formed molecules may evaporate leaving the surface for the fresh reactant molecules. Important features of solid catalysts (a) Activity The activity of a catalyst depends upon the strength of chemisorption to a large extent. ...
... (v) Diffusion of reaction products away from the catalyst’s surface. Thus, formed molecules may evaporate leaving the surface for the fresh reactant molecules. Important features of solid catalysts (a) Activity The activity of a catalyst depends upon the strength of chemisorption to a large extent. ...
Title of paper - Utrecht University Repository
... simulation methods have been used to calculate and compare the surface structure and energies of various surfaces of calcite (Titiloye et al. 1998). The calcium and carbonate-terminated {1014} surface of calcite is the most stable crystal plane and will dominate the predicted equilibrium morphology. ...
... simulation methods have been used to calculate and compare the surface structure and energies of various surfaces of calcite (Titiloye et al. 1998). The calcium and carbonate-terminated {1014} surface of calcite is the most stable crystal plane and will dominate the predicted equilibrium morphology. ...
Physics, Chapter 13: Properties of Matter
... composing it always occur in a definite ratio of their weights. More complex substances consist of several or many compounds. The smallest constituent of a chemical compound is the molecule; it is the fundamental structural unit of the compound. A molecule is composed of one or more atoms held toget ...
... composing it always occur in a definite ratio of their weights. More complex substances consist of several or many compounds. The smallest constituent of a chemical compound is the molecule; it is the fundamental structural unit of the compound. A molecule is composed of one or more atoms held toget ...
Sahand University of Technology
... • Basically an adhesive must do two things: 1. It must wet the surfaces, that is it must spread and make a contact angle approaching zero. Intimate contact is required between the molecules of the adhesive and the atoms and molecules in the surface. When applied the adhesive will be a liquid of rela ...
... • Basically an adhesive must do two things: 1. It must wet the surfaces, that is it must spread and make a contact angle approaching zero. Intimate contact is required between the molecules of the adhesive and the atoms and molecules in the surface. When applied the adhesive will be a liquid of rela ...
The Sabatier Principle Illustrated by Catalytic H2
... leveling off of the data (observed in the inset of Figure 2). A reaction solution of 250 mL of 0.1 M KOH and the metal foil to be tested were placed in a B€uchner flask fitted with a rubber stopper and containing a magnetic stir bar. One end of the hose, attached to the side arm of the flask, was placed ...
... leveling off of the data (observed in the inset of Figure 2). A reaction solution of 250 mL of 0.1 M KOH and the metal foil to be tested were placed in a B€uchner flask fitted with a rubber stopper and containing a magnetic stir bar. One end of the hose, attached to the side arm of the flask, was placed ...
Fabrication of superhydrophobic surface of stearic acid grafted zinc
... focus to this area due to its importance in industry [1-8]. In general, a surface with a water contact angle (CA) more than 150o and a sliding angle (SA) less than 10o is considered to be a super-hydrophobic surface. According to the Cassie-Baxter equation [9], both rough surface and low surface fre ...
... focus to this area due to its importance in industry [1-8]. In general, a surface with a water contact angle (CA) more than 150o and a sliding angle (SA) less than 10o is considered to be a super-hydrophobic surface. According to the Cassie-Baxter equation [9], both rough surface and low surface fre ...
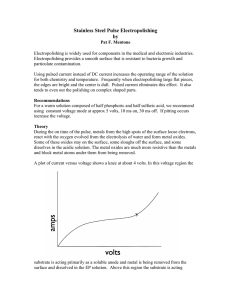
This is an overview of what can happen during the
... If the substrate functioned as a 100% soluble anode, the following would occur during the anodic portion of the pulse: 1. Metal atoms become free from their crystal structure and are mobile on substrate surface 2. Ions adjacent to the substrate align themselves into layers of alternating charge (cap ...
... If the substrate functioned as a 100% soluble anode, the following would occur during the anodic portion of the pulse: 1. Metal atoms become free from their crystal structure and are mobile on substrate surface 2. Ions adjacent to the substrate align themselves into layers of alternating charge (cap ...
The study on sensitivity analysis method of slope stability based on
... In this study, an attempt has been made to analyze the impacts of different parameters (such as the bulk density, the cohesive force, the internal angle of friction, the angle of slip surface and the pore water pressure) on the safety factor of a given slope. The analysis was done by using the Swedi ...
... In this study, an attempt has been made to analyze the impacts of different parameters (such as the bulk density, the cohesive force, the internal angle of friction, the angle of slip surface and the pore water pressure) on the safety factor of a given slope. The analysis was done by using the Swedi ...
blah
... The actual surface area was estimated by a utility in WSxM software [1] by considering the surface as a collection of 3D triangles (each of them formed by a point and its first neighbours). Then, we calculate the area of those singles triangles and obtain the real area of the sample by adding them u ...
... The actual surface area was estimated by a utility in WSxM software [1] by considering the surface as a collection of 3D triangles (each of them formed by a point and its first neighbours). Then, we calculate the area of those singles triangles and obtain the real area of the sample by adding them u ...
Colligative Properties of an Cyclohexane/1
... the meter and recorded a measurement. We found that our correction factor was 1.42. This was found by using the equation mg/R; m=mass of paper, R=measurement reading, and g=acceleration of gravity. Then we placed our various concentrations into the dish and recorded the different measurements. We th ...
... the meter and recorded a measurement. We found that our correction factor was 1.42. This was found by using the equation mg/R; m=mass of paper, R=measurement reading, and g=acceleration of gravity. Then we placed our various concentrations into the dish and recorded the different measurements. We th ...
NUCLEATION PRIMARY SECONDARY (induced by crystals
... breakage or abrasion, and pressure changes [Mul01]. There are different kinds of nucleation processes: If a solution contains no foreign particles or crystals of it’s own type, nucleus can only be formed by homogeneous nucleation. If foreign surfaces (particles) are present in the system, it is poss ...
... breakage or abrasion, and pressure changes [Mul01]. There are different kinds of nucleation processes: If a solution contains no foreign particles or crystals of it’s own type, nucleus can only be formed by homogeneous nucleation. If foreign surfaces (particles) are present in the system, it is poss ...
Catalyst Notes - University of Idaho
... are not consumed in the reaction concentration cancels out in the calculation of the equilibrium constant a small amount of catalyst affects the rate of reaction for a large amount of reactant (because the catalyst is not consumed, it can participate many times over) are classified as homogeneous or ...
... are not consumed in the reaction concentration cancels out in the calculation of the equilibrium constant a small amount of catalyst affects the rate of reaction for a large amount of reactant (because the catalyst is not consumed, it can participate many times over) are classified as homogeneous or ...
TVS_Au_poly_thiol_Au_junction_supplement_JCP
... the Au(111) surface directly. The HOMO-1, HOMO and LUMO of the biphenyl thiol molecule in the gas phase are also given for comparison. ...
... the Au(111) surface directly. The HOMO-1, HOMO and LUMO of the biphenyl thiol molecule in the gas phase are also given for comparison. ...
Modeling impacts of surface electromigration on stability and
... Thin films are deposited on thick substrates acting as heat sinks, thus they can sustain huge current densities (up to several MA/cm2 ) without melting Mikhail Khenner ...
... Thin films are deposited on thick substrates acting as heat sinks, thus they can sustain huge current densities (up to several MA/cm2 ) without melting Mikhail Khenner ...
Wetting

Wetting is the ability of a liquid to maintain contact with a solid surface, resulting from intermolecular interactions when the two are brought together. The degree of wetting (wettability) is determined by a force balance between adhesive and cohesive forces. Wetting deals with the three phases of materials: gas, liquid, and solid. It is now a center of attention in nanotechnology and nanoscience studies due to the advent of many nanomaterials in the past two decades (e.g. graphene, carbon nanotube).Wetting is important in the bonding or adherence of two materials. Wetting and the surface forces that control wetting are also responsible for other related effects, including so-called capillary effects. Regardless of the amount of wetting, the shape of a liquid drop on a rigid surface is roughly a truncated sphere.There are two types of wetting: non-reactive wetting and active wetting.