
The science of chemistry is concerned
... a mixture of two or more products containing the same element is formed. For example, when octane (or gasoline in general) burns in an excess of air, the reaction is 2C8H18 + 25O2 → 16CO2 + 18H2O If oxygen is the limiting reagent, however, the reaction does not necessarily stop short of consuming al ...
... a mixture of two or more products containing the same element is formed. For example, when octane (or gasoline in general) burns in an excess of air, the reaction is 2C8H18 + 25O2 → 16CO2 + 18H2O If oxygen is the limiting reagent, however, the reaction does not necessarily stop short of consuming al ...
Transition Metal-Modified Zirconium Phosphate Electrocatalysts for
... in relation to the two types of catalyst systems, resulting in overpotentials for metal-adsorbed ZrP catalysts that were less than, or equal to, their metal-intercalated counterparts. Keywords: zirconium phosphate; intercalation; ion exchange; catalyst support; oxygen evolution reaction; electrocata ...
... in relation to the two types of catalyst systems, resulting in overpotentials for metal-adsorbed ZrP catalysts that were less than, or equal to, their metal-intercalated counterparts. Keywords: zirconium phosphate; intercalation; ion exchange; catalyst support; oxygen evolution reaction; electrocata ...
Polyhedral Oligomeric Silsesquioxane
... phase-transfer agent is usually required to cover the water-soluble metal salts, and then they are transferred into the organic phase. Different from hydrophilic metal salts, the palladium acetate [Pd(OAc)2] is a source of Pd salts that can be dissolved in many organic solvents (e.g., toluene, tetrah ...
... phase-transfer agent is usually required to cover the water-soluble metal salts, and then they are transferred into the organic phase. Different from hydrophilic metal salts, the palladium acetate [Pd(OAc)2] is a source of Pd salts that can be dissolved in many organic solvents (e.g., toluene, tetrah ...
2 - cloudfront.net
... 6.78 g of copper is produced when 3.92 g of Al are reacted with excess copper (II) sulfate. Write balanced equation: 2Al + 3 CuSO4 Al2(SO4)3 + 3Cu What is the actual yield? What is the theoretical yield? What is the percent yield? ...
... 6.78 g of copper is produced when 3.92 g of Al are reacted with excess copper (II) sulfate. Write balanced equation: 2Al + 3 CuSO4 Al2(SO4)3 + 3Cu What is the actual yield? What is the theoretical yield? What is the percent yield? ...
Chapter 4 - Chemistry
... H2 PO4 can either accept a proton, H, to become H3PO4 and thus behaves as a Brønsted base, or can ...
... H2 PO4 can either accept a proton, H, to become H3PO4 and thus behaves as a Brønsted base, or can ...
EVS - RSC - Developments in Microwave Chemistry
... Evolution of Microwave Chemistry ............................................................7 Microwave Chemistry Apparatus ..............................................................9 Benefits of Microwave Chemistry ........................................................... 11 Limitations of M ...
... Evolution of Microwave Chemistry ............................................................7 Microwave Chemistry Apparatus ..............................................................9 Benefits of Microwave Chemistry ........................................................... 11 Limitations of M ...
Ozone decomposition
... When the ozone concentration exceeds 15–20% it has a blue color. At atmospheric pressure and temperature of 161.3 K, the ozone becomes fluid and is of deep blue color. It cures at 80.6 K by acquiring a dark purple color (Lunin et al., 1998). Ozone is explosive in all three physical conditions. Work ...
... When the ozone concentration exceeds 15–20% it has a blue color. At atmospheric pressure and temperature of 161.3 K, the ozone becomes fluid and is of deep blue color. It cures at 80.6 K by acquiring a dark purple color (Lunin et al., 1998). Ozone is explosive in all three physical conditions. Work ...
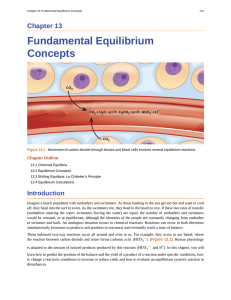
Fundamental Equilibrium Concepts
... Imagine a beach populated with sunbathers and swimmers. As those basking in the sun get too hot and want to cool off, they head into the surf to swim. As the swimmers tire, they head to the beach to rest. If these two rates of transfer (sunbathers entering the water, swimmers leaving the water) are ...
... Imagine a beach populated with sunbathers and swimmers. As those basking in the sun get too hot and want to cool off, they head into the surf to swim. As the swimmers tire, they head to the beach to rest. If these two rates of transfer (sunbathers entering the water, swimmers leaving the water) are ...
Stoichiometry - Social Circle City Schools
... The mole (abbreviated mol) is the unit chemists use when counting numbers of atoms or molecules in a sample. The number of particles (atoms, molecules, or other objects) in one mole is equal to the number of atoms in exactly 12 g of carbon-12. This number of particles is called Avogadro’s number (NA ...
... The mole (abbreviated mol) is the unit chemists use when counting numbers of atoms or molecules in a sample. The number of particles (atoms, molecules, or other objects) in one mole is equal to the number of atoms in exactly 12 g of carbon-12. This number of particles is called Avogadro’s number (NA ...
Stoichiometry Chapter 3 CHEMA1301 [Compatibility Mode]
... Symbols indicating the physical state of each reactant and product are often shown in chemical equations. We use the symbols (g), (l), (s), and (aq) for gas, liquid, solid, and aqueous (water) solution, respectively. Thus, Sometimes the conditions under which the reaction proceeds appear above or be ...
... Symbols indicating the physical state of each reactant and product are often shown in chemical equations. We use the symbols (g), (l), (s), and (aq) for gas, liquid, solid, and aqueous (water) solution, respectively. Thus, Sometimes the conditions under which the reaction proceeds appear above or be ...
Preparation and reactions of some lower tungsten halides and
... At present, the chief ores are wolframite (iron, manganese tungstate), scheelite (calcium tungstate), and stolzite (lead tungstate). The purification of tungsten from its ores is relatively simple. ...
... At present, the chief ores are wolframite (iron, manganese tungstate), scheelite (calcium tungstate), and stolzite (lead tungstate). The purification of tungsten from its ores is relatively simple. ...
PART 6-ICHO-26-30
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
Determination of Equilibrium Constants for Reactions between Nitric
... supply for ozone generation. A data logger was used to collect data every 2 s. The operation terminated when the outlet concentration reached 605 ppm or so and remained invariable, which indicated that the reaction reached equilibrium. Experiments for determination of the equilibrium constants were ...
... supply for ozone generation. A data logger was used to collect data every 2 s. The operation terminated when the outlet concentration reached 605 ppm or so and remained invariable, which indicated that the reaction reached equilibrium. Experiments for determination of the equilibrium constants were ...
Part 2-ICHO-26-30
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
Document
... then convert to favor spontaneity. For an endothermic process such as this, degreeschange Celsius. which requires the input of heat, it should make sense that adding Solution more heat by increasing the temperature will shift the equilibrium to 476 J 1 kJ the right, thusΔS making it “more spontaneou ...
... then convert to favor spontaneity. For an endothermic process such as this, degreeschange Celsius. which requires the input of heat, it should make sense that adding Solution more heat by increasing the temperature will shift the equilibrium to 476 J 1 kJ the right, thusΔS making it “more spontaneou ...
Chapter 3 Stoichiometry
... present in either one molecule of the compound or one mole of the compound. For example, one molecule of acetic acid, CH3CO2H, contains two atoms of oxygen and one mole of acetic acid contains 2 mol of oxygen atoms. When working with ionic and other types of nonmolecular compounds, the compound f ...
... present in either one molecule of the compound or one mole of the compound. For example, one molecule of acetic acid, CH3CO2H, contains two atoms of oxygen and one mole of acetic acid contains 2 mol of oxygen atoms. When working with ionic and other types of nonmolecular compounds, the compound f ...
Balancing Chemical Equations Using Models
... 6. Then add any coefficients if they are needed, and count how many atoms we now have. ___HCl + ____NaOH ____NaCl + ____H2O For our example we have the same number of each atom for both products and reactants, we do not need to add any coefficients to balance the equation. Reactants Before Balancing ...
... 6. Then add any coefficients if they are needed, and count how many atoms we now have. ___HCl + ____NaOH ____NaCl + ____H2O For our example we have the same number of each atom for both products and reactants, we do not need to add any coefficients to balance the equation. Reactants Before Balancing ...
CHEMICAL AND PROCESS DESIGN HANDBOOK
... as in the reaction of aniline with methyl alcohol in the presence of sulfuric acid catalyst: C6H5NH2 + 2CH3OH → C6H5N(CH3)2 + 2H2O Thus, aniline, with a considerable excess of methyl alcohol and a catalytic amount of sulfuric acid, is heated in an autoclave at about 200o C for 5 or 6 hours at a high ...
... as in the reaction of aniline with methyl alcohol in the presence of sulfuric acid catalyst: C6H5NH2 + 2CH3OH → C6H5N(CH3)2 + 2H2O Thus, aniline, with a considerable excess of methyl alcohol and a catalytic amount of sulfuric acid, is heated in an autoclave at about 200o C for 5 or 6 hours at a high ...
free sample
... 13) Identify the balanced equation to show the reaction of aqueous aluminum acetate with aqueous ammonium phosphate to form solid aluminum phosphate and aqueous ammonium acetate. A) Al(C2H3O2)2(aq) + (NH4)2PO4(aq) → AlPO4(s) + 2NH4C2H3O2(aq) B) Al(C2H3O2)2(aq) + (NH3)2PO4(aq) → AlPO4(s) + 2NH3C2H3O2 ...
... 13) Identify the balanced equation to show the reaction of aqueous aluminum acetate with aqueous ammonium phosphate to form solid aluminum phosphate and aqueous ammonium acetate. A) Al(C2H3O2)2(aq) + (NH4)2PO4(aq) → AlPO4(s) + 2NH4C2H3O2(aq) B) Al(C2H3O2)2(aq) + (NH3)2PO4(aq) → AlPO4(s) + 2NH3C2H3O2 ...
b - Gordon State College
... What is the mass of O2 in grams required by NH3? Which reactant is the limiting reagent? How many grams of NO will be produced in theory? How many grams of H2O will be produced in theory? How many grams of the excess reagent remain unreacted? If only 1.80 g of NO are produced, what is the percent yi ...
... What is the mass of O2 in grams required by NH3? Which reactant is the limiting reagent? How many grams of NO will be produced in theory? How many grams of H2O will be produced in theory? How many grams of the excess reagent remain unreacted? If only 1.80 g of NO are produced, what is the percent yi ...
Section 1.3 - The Student Room
... a Standard enthalpy change of combustion is the enthalpy change when 1 mole of the compound is burnt completely in oxygen, under standard conditions (ie the compound and the products in their most stable states at 1 atmosphere pressure and at a stated temperature, often 298 K). b Standard enthalpy c ...
... a Standard enthalpy change of combustion is the enthalpy change when 1 mole of the compound is burnt completely in oxygen, under standard conditions (ie the compound and the products in their most stable states at 1 atmosphere pressure and at a stated temperature, often 298 K). b Standard enthalpy c ...
Workshop materials for Class XII
... What new technology can help you? Learn more about computers—use them not only for power points but also for video recording. Do you get good contacts because of your nature that helps you benefit the school What trend do you see in your school how you can take advantage of these— eg: A circular ...
... What new technology can help you? Learn more about computers—use them not only for power points but also for video recording. Do you get good contacts because of your nature that helps you benefit the school What trend do you see in your school how you can take advantage of these— eg: A circular ...
CHAPTER 4 REACTIONS IN AQUEOUS SOLUTIONS
... In order to work this problem, you need to assign the oxidation numbers to all the elements in the compounds. In each case oxygen has an oxidation number of −2 (rule 3). These oxidation numbers should then be compared to the range of possible oxidation numbers that each element can have. Molecular o ...
... In order to work this problem, you need to assign the oxidation numbers to all the elements in the compounds. In each case oxygen has an oxidation number of −2 (rule 3). These oxidation numbers should then be compared to the range of possible oxidation numbers that each element can have. Molecular o ...
Experiments in General Chemistry: Featuring MeasureNet
... Chemists observe matter by determining, measuring, and monitoring physical and chemical properties of matter. A property is any characteristic that can be used to describe matter (e.g., size, color, mass, density, solubility, etc.). In this experiment, we will determine the density of liquids and so ...
... Chemists observe matter by determining, measuring, and monitoring physical and chemical properties of matter. A property is any characteristic that can be used to describe matter (e.g., size, color, mass, density, solubility, etc.). In this experiment, we will determine the density of liquids and so ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.







![Stoichiometry Chapter 3 CHEMA1301 [Compatibility Mode]](http://s1.studyres.com/store/data/014247793_1-84b4b6fe6fa37d77afbf7eb657ee347a-300x300.png)














