Chemistry 20H
... A chemical change results when the atoms of one or more substances are rearranged to produce new substances with new properties. In a chemical reaction the atoms remain the same, but their arrangement changes. For instance: H2O H2 + O2 In this reaction water is broken down into two substances, oxy ...
... A chemical change results when the atoms of one or more substances are rearranged to produce new substances with new properties. In a chemical reaction the atoms remain the same, but their arrangement changes. For instance: H2O H2 + O2 In this reaction water is broken down into two substances, oxy ...
A Gas-phase Electrochemical Reactor for Carbon Dioxide
... the main products obtained are C1 (or at most C2) hydrocarbons or oxygenates. The target is in fact the formation of liquid fuels with C≥2 because they are easier to transport and store, preserving the large investments made in the current energy and chemical infrastructures (Ampelli et al., 2011b). ...
... the main products obtained are C1 (or at most C2) hydrocarbons or oxygenates. The target is in fact the formation of liquid fuels with C≥2 because they are easier to transport and store, preserving the large investments made in the current energy and chemical infrastructures (Ampelli et al., 2011b). ...
Chapter 3 Reading
... 3.6 Quantitative Information from Balanced Equations The coefficients in a balanced chemical equation give the ratios of molecules (or formula units) involved in the reaction. •The stoichiometric coefficients in the balanced equation may be interpreted as: •the relative numbers of molecules or formu ...
... 3.6 Quantitative Information from Balanced Equations The coefficients in a balanced chemical equation give the ratios of molecules (or formula units) involved in the reaction. •The stoichiometric coefficients in the balanced equation may be interpreted as: •the relative numbers of molecules or formu ...
H 2 O
... Schuler formula • The Schuler formula gives the yield of secondary radicals formed from the reaction of hydroxyl radicals and the scavenger S at concentration [S] G N 2O ( OH) 5.2 3.0 ...
... Schuler formula • The Schuler formula gives the yield of secondary radicals formed from the reaction of hydroxyl radicals and the scavenger S at concentration [S] G N 2O ( OH) 5.2 3.0 ...
Oxidation Numbers and Ionic Compounds
... Directions: Classify the following compounds as (I) ionic (metal + nonmetal) (C) covalent (nonmetal + nonmetal) (D) diatomic molecules (two of the same atom bound together) (P) polyatomic ion (groups containing more than two elements covalently bonded together that carry an overall charge. ) ...
... Directions: Classify the following compounds as (I) ionic (metal + nonmetal) (C) covalent (nonmetal + nonmetal) (D) diatomic molecules (two of the same atom bound together) (P) polyatomic ion (groups containing more than two elements covalently bonded together that carry an overall charge. ) ...
Synthesis Reaction
... I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replacement, double replacement, and combustion) ...
... I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replacement, double replacement, and combustion) ...
Document
... I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replacement, double replacement, and combustion) ...
... I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replacement, double replacement, and combustion) ...
Chapter 2 (Hill/Petrucci/McCreary/Perry This chapter deals with
... This chapter deals with matter, in its various forms, on the nanoscale. While we believe that all matter is composed of protons, electrons and, usually, neutrons, we encounter matter as elements in the form of atoms (and, occasionally, molecules), as compounds in the form of molecules and crystal la ...
... This chapter deals with matter, in its various forms, on the nanoscale. While we believe that all matter is composed of protons, electrons and, usually, neutrons, we encounter matter as elements in the form of atoms (and, occasionally, molecules), as compounds in the form of molecules and crystal la ...
Chapter 4: Aqueous Solutions (Chs 4 and 5 in Jespersen, Ch4 in
... 5) Displacement reactions (a reaction where an ion in solution is displaced (or replaced) through oxidation of an element). Common types are metal, hydrogen and halogen displacements. 5a) Metal displacement - Oxidation of metals (metal → metal cation) by acids and salts: ...
... 5) Displacement reactions (a reaction where an ion in solution is displaced (or replaced) through oxidation of an element). Common types are metal, hydrogen and halogen displacements. 5a) Metal displacement - Oxidation of metals (metal → metal cation) by acids and salts: ...
chemistry 11 exam review
... Solubilty Chart for double displacement reactions Neutralization Reactions Chemical Reactions for the formation of acids and bases from metal oxides and non-metal oxides Practice: 1. Predict the products and write a balanced chemical equation for the following chemical reaction: Hydrochloric a ...
... Solubilty Chart for double displacement reactions Neutralization Reactions Chemical Reactions for the formation of acids and bases from metal oxides and non-metal oxides Practice: 1. Predict the products and write a balanced chemical equation for the following chemical reaction: Hydrochloric a ...
Formula and The Mole
... 18. Ionic compounds are usually soluble in water and covalent compounds do not usually dissolve in water, but will dissolve in other solvents like hexane. 19. An electric current is a flow of ions in an electrolyte and a flow of electrons in a metal or graphite. 20. Coloured ions can be seen migrati ...
... 18. Ionic compounds are usually soluble in water and covalent compounds do not usually dissolve in water, but will dissolve in other solvents like hexane. 19. An electric current is a flow of ions in an electrolyte and a flow of electrons in a metal or graphite. 20. Coloured ions can be seen migrati ...
Chapters 1-4 Numbers and Measurements in Chemistry Units SI
... that have the same chemical formula but are connected differently. – Three isomers of pentane, C5H12. ...
... that have the same chemical formula but are connected differently. – Three isomers of pentane, C5H12. ...
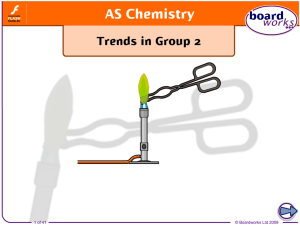
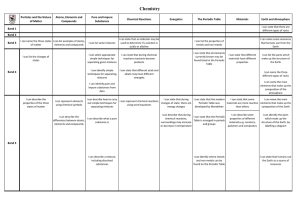
Groups 2 and 7
... William Fox Talbot (1800–1877) was a British scientist and mathematician. He was one of the key figures in the development of the use of silver halides in photography. A French scientist called Louis Daguerre developed the use of silver halides on copper plates. These were effective at producing pri ...
... William Fox Talbot (1800–1877) was a British scientist and mathematician. He was one of the key figures in the development of the use of silver halides in photography. A French scientist called Louis Daguerre developed the use of silver halides on copper plates. These were effective at producing pri ...
Atoms, Molecules and Ions
... 2. The atoms of one element are different from the atoms of another element. 3. Atoms combine in definite ratios to make compounds. 4 Combinations of atoms in compounds can change ...
... 2. The atoms of one element are different from the atoms of another element. 3. Atoms combine in definite ratios to make compounds. 4 Combinations of atoms in compounds can change ...
Lecture 4 Oxidation (applies to Si and SiC only) Reading: Chapter 4
... decreasing device dimensions. Electrons tend to be accelerated into the Hydrogen, breaking the H-Si Bond. These same broken bonds can then trap electrons, preventing or slowing their conduction. •Since the Si/SiO2 interface never sees the ambient, it is extremely pure (impurities must be adsorbed on ...
... decreasing device dimensions. Electrons tend to be accelerated into the Hydrogen, breaking the H-Si Bond. These same broken bonds can then trap electrons, preventing or slowing their conduction. •Since the Si/SiO2 interface never sees the ambient, it is extremely pure (impurities must be adsorbed on ...
MOLES, MASS, and VOLUME OF A GAS
... Insects of a particular species are able to identify their mate by using special chemicals called pheromones that transmit chemical messages. The pheromone that serves as a sex attractant for gypsy moths is called disparlure, and contains thew elements C, H, and O. Analysis of disparlure shows that ...
... Insects of a particular species are able to identify their mate by using special chemicals called pheromones that transmit chemical messages. The pheromone that serves as a sex attractant for gypsy moths is called disparlure, and contains thew elements C, H, and O. Analysis of disparlure shows that ...
Lecture 11 - U of L Class Index
... smallest reduction potential (in magnitude). This difference is due to solvation (hydration). As discussed in the section on acid/base properties, the electrostatic parameter for Li is high (ξ = Z2/r) because of its very small radius. This gives it a high energy of hydration, and it is this term in ...
... smallest reduction potential (in magnitude). This difference is due to solvation (hydration). As discussed in the section on acid/base properties, the electrostatic parameter for Li is high (ξ = Z2/r) because of its very small radius. This gives it a high energy of hydration, and it is this term in ...
CHEMISTRY SEMESTER ONE LAB 1 Lab 1: Stoichiometry and
... In this experiment you will observe the reaction between metallic iron and a solution of copper(II) sulfate. This reaction produces metallic copper, which is seen precipitating as a finely divided red powder. The reaction in which one metal replaces another metal from a solution of one of its salts ...
... In this experiment you will observe the reaction between metallic iron and a solution of copper(II) sulfate. This reaction produces metallic copper, which is seen precipitating as a finely divided red powder. The reaction in which one metal replaces another metal from a solution of one of its salts ...
Document
... H2(g) + O2(g) H2O(g) What do we do to avoid violating the law of conservation of matter? (As written we’ve lost ...
... H2(g) + O2(g) H2O(g) What do we do to avoid violating the law of conservation of matter? (As written we’ve lost ...
Midterm 1 Spring 2004
... 7. (4 points) Write the products of the following reactions. If no reaction takes place, indicate “N.R.” You must write all the products, but you do not need to balance the reaction. A. AgNO3(aq) + NaCl(aq) ? B. Cu(s) + AgNO3(aq) ? C. Au(s) + Zn(NO3)2(aq) ? D. CH4(g) + O2(g) ? 8. (6 points) How many ...
... 7. (4 points) Write the products of the following reactions. If no reaction takes place, indicate “N.R.” You must write all the products, but you do not need to balance the reaction. A. AgNO3(aq) + NaCl(aq) ? B. Cu(s) + AgNO3(aq) ? C. Au(s) + Zn(NO3)2(aq) ? D. CH4(g) + O2(g) ? 8. (6 points) How many ...
Chemistry of the Non
... We divide the periodic table into metals, nonmetals and metalloids. Nonmetals occupy the upper right portion of the periodic table. • H is a special case. Electronegativity is important when determining whether an element is a metal. Nonmetals tend to have higher electronegativities than metals. • T ...
... We divide the periodic table into metals, nonmetals and metalloids. Nonmetals occupy the upper right portion of the periodic table. • H is a special case. Electronegativity is important when determining whether an element is a metal. Nonmetals tend to have higher electronegativities than metals. • T ...
Chemistry - Edgbarrow School
... materials are more reactive elements that make up the using word equations ...
... materials are more reactive elements that make up the using word equations ...
Semester 2 Review
... 23. 1 mole of any gas (CH4, CO2, H2, etc…) = ______________ L (if at STP) 24. At an amusement park, a balloon is filled with 5.00 L of helium at a temperature of 308 K. What will be the volume of the balloon when it is brought into an air-conditioned house at 295 K? ...
... 23. 1 mole of any gas (CH4, CO2, H2, etc…) = ______________ L (if at STP) 24. At an amusement park, a balloon is filled with 5.00 L of helium at a temperature of 308 K. What will be the volume of the balloon when it is brought into an air-conditioned house at 295 K? ...
Hints for Names and Formulas (Ch. 4 in Zumdahl Chemistry)
... (1) in this course, molecules are usually defined as two or more nonmetallic atoms bonded covalently (2) molecules are finite groupings of nonmetallic atoms that may or may not be different elements ● only 10 elements exist as molecules when free in nature: H2, N2, O2, F2, Cl2, Br2, I2, At2, P4, S8 ...
... (1) in this course, molecules are usually defined as two or more nonmetallic atoms bonded covalently (2) molecules are finite groupings of nonmetallic atoms that may or may not be different elements ● only 10 elements exist as molecules when free in nature: H2, N2, O2, F2, Cl2, Br2, I2, At2, P4, S8 ...
Bonding Nomenclature Notes
... Single Covalent Bond -two atoms share 1 pair of electrons Double Covalent Bond -two atoms share 2 pair of electrons Triple Covalent Bond -two atoms share 3 pair of electrons ...
... Single Covalent Bond -two atoms share 1 pair of electrons Double Covalent Bond -two atoms share 2 pair of electrons Triple Covalent Bond -two atoms share 3 pair of electrons ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.