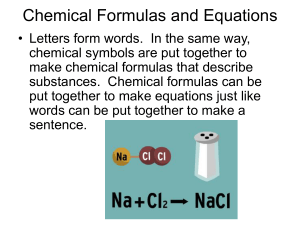
7.1 Describing Reactions
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
Chemical Formulas and Equations
... The Importance of Accuracy • CO2 is a colorless, odorless gas you exhale. • CO is a colorless, odorless, and poisonous gas. • Co is an element. ...
... The Importance of Accuracy • CO2 is a colorless, odorless gas you exhale. • CO is a colorless, odorless, and poisonous gas. • Co is an element. ...
Reactions Balancing Chemical Equations uses Law of conservation
... Solving problems involving Chemical reactions ...
... Solving problems involving Chemical reactions ...
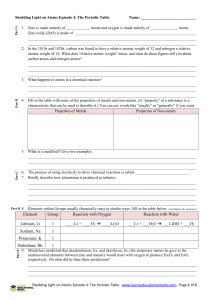
Element Group Reaction with Oxygen Reaction with Water Lithium
... atomic weight of 14. What does “relative atomic weight” mean, and what do these figures tell you about carbon atoms and nitrogen atoms? ______________________________________________________________________________________ _____________________________________________________________________________ ...
... atomic weight of 14. What does “relative atomic weight” mean, and what do these figures tell you about carbon atoms and nitrogen atoms? ______________________________________________________________________________________ _____________________________________________________________________________ ...
Chemistry STAAR Review File
... Atoms combine in new ways during a chemical change. When elements react, their atoms combine in simple, whole-number ratios. Two criteria are usually applied to any theory. First, does it agree with facts which are already known? Second, does it predict new relationships and stimulate additional obs ...
... Atoms combine in new ways during a chemical change. When elements react, their atoms combine in simple, whole-number ratios. Two criteria are usually applied to any theory. First, does it agree with facts which are already known? Second, does it predict new relationships and stimulate additional obs ...
Naming Binary Molecular Compounds
... Oxidation # for Ba = +2 Which elements have specific rules? Oxygen has a rule....-2 in most compounds Oxidation # for O = -2 Which element does not have a specific rule? N does not have a specific rule. Use rule 8 to find the oxidation # of N Let N = Oxidation # for nitrogen (# Ba) (Oxid. # of Ba) + ...
... Oxidation # for Ba = +2 Which elements have specific rules? Oxygen has a rule....-2 in most compounds Oxidation # for O = -2 Which element does not have a specific rule? N does not have a specific rule. Use rule 8 to find the oxidation # of N Let N = Oxidation # for nitrogen (# Ba) (Oxid. # of Ba) + ...
Sample 112 Final
... b) ammonium sulfate, NH4SO4 c) iron(III) chloride, Fe2Cl3 d) carbon tetrachloride, CCl4 e) magnesium nitrite, Mg(NO3)2 ...
... b) ammonium sulfate, NH4SO4 c) iron(III) chloride, Fe2Cl3 d) carbon tetrachloride, CCl4 e) magnesium nitrite, Mg(NO3)2 ...
Chemistry Fall Final Study Guide Concepts
... Electronegativity is the attraction of bonded electrons by an atom. The most electronegative elements (polar bears) are located on the top, right (nonmetals) of the periodic table. The least electronegative elements (metals) are located at the bottom left of the periodic table. 15. How does the elec ...
... Electronegativity is the attraction of bonded electrons by an atom. The most electronegative elements (polar bears) are located on the top, right (nonmetals) of the periodic table. The least electronegative elements (metals) are located at the bottom left of the periodic table. 15. How does the elec ...
College Chemistry I PHS 1025 Fall 2012 Practice Exam 3A
... C) Mn2+(aq) + 4 OH-(aq) → MnO2(s) + 2 H2O(l) + 2eD) Mn2+(aq) + 2 H2O(l) → MnO2(s) + 4 H+(aq) + 2e77) The reaction Cu(s) + 2 AgNO3(aq) → Cu(NO3)2(aq) + 2 Ag(s) is best classified as a(n) A) oxidation-reduction reaction. C) double replacement reaction. ...
... C) Mn2+(aq) + 4 OH-(aq) → MnO2(s) + 2 H2O(l) + 2eD) Mn2+(aq) + 2 H2O(l) → MnO2(s) + 4 H+(aq) + 2e77) The reaction Cu(s) + 2 AgNO3(aq) → Cu(NO3)2(aq) + 2 Ag(s) is best classified as a(n) A) oxidation-reduction reaction. C) double replacement reaction. ...
Energy Level Models - Middle School Chemistry
... electrons is intended to suggest information about the substructure within energy levels. This substructure is made up of regions called orbitals which comprise each energy level. The shape and size of the orbital is defined by the space around the nucleus where there is a high probability of findin ...
... electrons is intended to suggest information about the substructure within energy levels. This substructure is made up of regions called orbitals which comprise each energy level. The shape and size of the orbital is defined by the space around the nucleus where there is a high probability of findin ...
Knox Chem Prelim 2009
... with dilute acid. Choose a metal which reacts more slowly than magnesium, but more rapidly than lead to answer the following questions. (a) Describe your procedure and the observations you made. ...
... with dilute acid. Choose a metal which reacts more slowly than magnesium, but more rapidly than lead to answer the following questions. (a) Describe your procedure and the observations you made. ...
Multivalent Ionic Compounds
... 4.2 PRACTICE: Names and Formulas of Compounds 1. Complete Names and Formulas of Compounds challenge, 4.2 crossword, 4.2 Quiz, and 4.2 Check Your Understanding. 2. Determine the formula of each of the following monovalent ionic compounds. Use your periodic table to look up the charge on each ion. If ...
... 4.2 PRACTICE: Names and Formulas of Compounds 1. Complete Names and Formulas of Compounds challenge, 4.2 crossword, 4.2 Quiz, and 4.2 Check Your Understanding. 2. Determine the formula of each of the following monovalent ionic compounds. Use your periodic table to look up the charge on each ion. If ...
Unit D: Quantitative Relationships in Chemical Change
... Every chemical reaction involves the rearrangement of atoms into different combinations. However, during these reactions, the total number of atoms of each type of element is the same after the reaction as it was before the reaction. ...
... Every chemical reaction involves the rearrangement of atoms into different combinations. However, during these reactions, the total number of atoms of each type of element is the same after the reaction as it was before the reaction. ...
L22 - Supplementary Student Notes Package
... Every chemical reaction involves the rearrangement of atoms into different combinations. However, during these reactions, the total number of atoms of each type of element is the same after the reaction as it was before the reaction. ...
... Every chemical reaction involves the rearrangement of atoms into different combinations. However, during these reactions, the total number of atoms of each type of element is the same after the reaction as it was before the reaction. ...
Chemistry Final Exam Review 2006-2007
... 2. Know how to calculate the mole ratio between reactants and products in a chemical formula. a) What is the mole ratio for calcium and oxygen in 2Ca + O2 → 2CaO 3. Know how to solve mole to mole, mole to mass, mass to mole, mass to mass problems. a) How many moles of lithium hydroxide are required ...
... 2. Know how to calculate the mole ratio between reactants and products in a chemical formula. a) What is the mole ratio for calcium and oxygen in 2Ca + O2 → 2CaO 3. Know how to solve mole to mole, mole to mass, mass to mole, mass to mass problems. a) How many moles of lithium hydroxide are required ...
File
... atomic emission spectra; Bohr model of the hydrogen atom including explanation of H line spectrum and orbits, electron cloud and the probability model, wave/particle duality of electrons revisited, relate electron configurations of atoms to the Bohr and electron cloud models, describe the concepts o ...
... atomic emission spectra; Bohr model of the hydrogen atom including explanation of H line spectrum and orbits, electron cloud and the probability model, wave/particle duality of electrons revisited, relate electron configurations of atoms to the Bohr and electron cloud models, describe the concepts o ...
The Free High School Science Texts: A Textbook for High School
... Aside: Probabilities describe the chance of something happening or of being true. They usually have a value between 0 and 1 or 0% and 100% where 0 means no chance at all and 1 means definite. Probabilities are used when the state of something is uncertain. For example, probabilities are often used ...
... Aside: Probabilities describe the chance of something happening or of being true. They usually have a value between 0 and 1 or 0% and 100% where 0 means no chance at all and 1 means definite. Probabilities are used when the state of something is uncertain. For example, probabilities are often used ...
Section 3.6
... 16. (a) Dimes were shipped out of the country because it is illegal to deface or alter Canadian currency in Canada. (b) These metals have very different magnetic properties, which could be used to separate them. (c) A magnet should separate these coins easily, because nickel is ferromagnetic (strong ...
... 16. (a) Dimes were shipped out of the country because it is illegal to deface or alter Canadian currency in Canada. (b) These metals have very different magnetic properties, which could be used to separate them. (c) A magnet should separate these coins easily, because nickel is ferromagnetic (strong ...
Spring 2014
... 13. The equilibrium constant is equal to 5.00 at 1300 K for the reaction 2 SO2 (g) + O2 (g) W 2 SO3 (g). If initial concentrations are [SO2] = 4.0 M, [O2] = 4.0 M, and [SO3] = 4.0 M, the system is a) not at equilibrium and will shift to the right (products) to achieve an equilibrium state. b) not at ...
... 13. The equilibrium constant is equal to 5.00 at 1300 K for the reaction 2 SO2 (g) + O2 (g) W 2 SO3 (g). If initial concentrations are [SO2] = 4.0 M, [O2] = 4.0 M, and [SO3] = 4.0 M, the system is a) not at equilibrium and will shift to the right (products) to achieve an equilibrium state. b) not at ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... Analyze: Our task is to write a net ionic equation for a precipitation reaction, given the names of the reactants present in solution. Plan: We first need to write the chemical formulas of the reactants and products and to determine which product is insoluble. Then we write and balance the molecular ...
... Analyze: Our task is to write a net ionic equation for a precipitation reaction, given the names of the reactants present in solution. Plan: We first need to write the chemical formulas of the reactants and products and to determine which product is insoluble. Then we write and balance the molecular ...
Chapter 2 Expanded Notes
... The Atomic Number: This is more important for chemistry. Note as mentioned earlier that atoms are classified into elements based on certain characteristics, which we call identity, and they retain this identity through chemical reactions. Atoms of one element are different from atoms of another ele ...
... The Atomic Number: This is more important for chemistry. Note as mentioned earlier that atoms are classified into elements based on certain characteristics, which we call identity, and they retain this identity through chemical reactions. Atoms of one element are different from atoms of another ele ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.