Chemical Reactions - Waukee Community School District Blogs
... 1. H.O.F.Br.I.N.Cl. (Dr. HOFINBrCl) 2. These elements need a subscript 2 after them if ...
... 1. H.O.F.Br.I.N.Cl. (Dr. HOFINBrCl) 2. These elements need a subscript 2 after them if ...
Slide 1
... Step 1: Obtain the mass of each element present (in grams) Step 2: Determine the number of moles of each type of atom present Step 3: Divide the number of mole of each element by the smallest number of moles to convert the smallest number to 1. If all of the numbers so obtained are integers, these a ...
... Step 1: Obtain the mass of each element present (in grams) Step 2: Determine the number of moles of each type of atom present Step 3: Divide the number of mole of each element by the smallest number of moles to convert the smallest number to 1. If all of the numbers so obtained are integers, these a ...
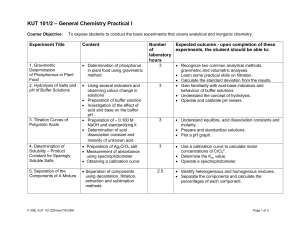
KUT 101/2 – General Chemistry Practical I
... • Recognise coordination compounds, wherein the metal is a Lewis acid and the atoms or molecules joined to the metal are Lewis base or ligands. • Calculate the percentage yield. ...
... • Recognise coordination compounds, wherein the metal is a Lewis acid and the atoms or molecules joined to the metal are Lewis base or ligands. • Calculate the percentage yield. ...
Practice problem chap3 1. The atomic mass of 35Cl (75.53%) and
... 1. The atomic mass of Cl (75.53%) and 37Cl (24.47%) are 34.968amu and 36.956amu.Calculate the average atomic mass in amu. 2. What is the mass percent (%) for O in SO2? (a) 38.09 (b) 45.41 (c) 50.00 (d) 53.86 (e) 56.43 3. How many molecules of ethane (C2H6) are present in 0.334g of C2H6?How many H at ...
... 1. The atomic mass of Cl (75.53%) and 37Cl (24.47%) are 34.968amu and 36.956amu.Calculate the average atomic mass in amu. 2. What is the mass percent (%) for O in SO2? (a) 38.09 (b) 45.41 (c) 50.00 (d) 53.86 (e) 56.43 3. How many molecules of ethane (C2H6) are present in 0.334g of C2H6?How many H at ...
Unit 7 Packet
... make, there are patterns in the way chemical bonds are rearranged in a reaction. These patterns make it easier to predict the outcome of a chemical reaction. Below are 5 useful reaction patterns. Look at the examples given for each type. Clues for identifying these patterns include the kind of subst ...
... make, there are patterns in the way chemical bonds are rearranged in a reaction. These patterns make it easier to predict the outcome of a chemical reaction. Below are 5 useful reaction patterns. Look at the examples given for each type. Clues for identifying these patterns include the kind of subst ...
Chemical reactions alter arrangements of atoms.
... faster. The result is that more collisions take place per second and occur with greater force. The most common way to make the particles move faster is to add energy to the reactants, which will raise their temperature. ...
... faster. The result is that more collisions take place per second and occur with greater force. The most common way to make the particles move faster is to add energy to the reactants, which will raise their temperature. ...
120CH05 - Louisiana Tech University
... Theoretical and percent yield • How good an experimentalist are you? • What if 100% of reactants are not converted to desired products? • Frequently happens because of “side reactions” (other products), handling, etc. – 100% amount is theoretical yield – Amount obtained is actual yield ...
... Theoretical and percent yield • How good an experimentalist are you? • What if 100% of reactants are not converted to desired products? • Frequently happens because of “side reactions” (other products), handling, etc. – 100% amount is theoretical yield – Amount obtained is actual yield ...
Project Advance Chemistry 106 Sample Questions
... an increase in the frequency of the molecular collisions. an increase in the fraction of molecules that have enough energy to react. an increase in the rate constant. an increase in the kinetic energy of the molecules. an increase in the average potential energy of the molecules. ...
... an increase in the frequency of the molecular collisions. an increase in the fraction of molecules that have enough energy to react. an increase in the rate constant. an increase in the kinetic energy of the molecules. an increase in the average potential energy of the molecules. ...
Smith Reaction- HW PSI Chemistry
... 37) Which of the following is NOT true concerning the decomposition of a simple binary compound? A) The products are unpredictable. B) The products are the constituent elements. C) The reactant is a single substance. D) The reactant could be an ionic or a molecular compound. E) Energy is usually r ...
... 37) Which of the following is NOT true concerning the decomposition of a simple binary compound? A) The products are unpredictable. B) The products are the constituent elements. C) The reactant is a single substance. D) The reactant could be an ionic or a molecular compound. E) Energy is usually r ...
chm 434f/1206f solid state materials chemistry
... • Form or morphology and physical size of product controls synthesis method of choice and potential utility • Single crystal, phase pure, defect free solids - do not exist and if they did not likely of much interest! • Single crystal (SC) that has been defect modified with dopants - intrinsic vs ext ...
... • Form or morphology and physical size of product controls synthesis method of choice and potential utility • Single crystal, phase pure, defect free solids - do not exist and if they did not likely of much interest! • Single crystal (SC) that has been defect modified with dopants - intrinsic vs ext ...
- Gondwana University, Gadchiroli
... isothermal & adiabatic conditions for reversible process, carnot’s cycle & its efficiency, thermodynamic scale of temperature. [5L] (C) Thermochemistry: Heat of reaction, standard states, relation between heat of reaction at constant volume & at constant pressure, Hess’s law of constant heat of summ ...
... isothermal & adiabatic conditions for reversible process, carnot’s cycle & its efficiency, thermodynamic scale of temperature. [5L] (C) Thermochemistry: Heat of reaction, standard states, relation between heat of reaction at constant volume & at constant pressure, Hess’s law of constant heat of summ ...
Click here for the Reaction NOTES Handout
... Redox: Oxidation is loss of electrons. Reduction is gain of electrons. LEO the lion goes GER For example, in the extraction of iron from its ore. Because both reduction and oxidation are going on side-byside, this is known as a redox reaction. ...
... Redox: Oxidation is loss of electrons. Reduction is gain of electrons. LEO the lion goes GER For example, in the extraction of iron from its ore. Because both reduction and oxidation are going on side-byside, this is known as a redox reaction. ...
New Title
... 2. Is the following sentence true or false? Chemical equations use symbols instead of words to summarize chemical reactions. 3. If a molecule of carbon dioxide is involved in a chemical reaction, how is it represented in the chemical equation for the reaction? 4. The substances you have at the begin ...
... 2. Is the following sentence true or false? Chemical equations use symbols instead of words to summarize chemical reactions. 3. If a molecule of carbon dioxide is involved in a chemical reaction, how is it represented in the chemical equation for the reaction? 4. The substances you have at the begin ...
FINAL EXAM Spring 2012
... The last page of this examination is a periodic table [Gas constant = 8.314 J/mol K; 0.08206 L*atm/mole*K,1 faraday = 96500 J/V mol e-; at 25oC] 1) The reaction has the rate law, Rate = k[A][B]2. Which will cause the rate to increase the most? A) doubling [A] B) doubling [B] C) tripling [B] D) quadr ...
... The last page of this examination is a periodic table [Gas constant = 8.314 J/mol K; 0.08206 L*atm/mole*K,1 faraday = 96500 J/V mol e-; at 25oC] 1) The reaction has the rate law, Rate = k[A][B]2. Which will cause the rate to increase the most? A) doubling [A] B) doubling [B] C) tripling [B] D) quadr ...
AP Chemistry Summer Assignment
... 1. Write the most common guidelines to determine significant figures (digits) with an example? 2. Use factor labeling method to convert the following: a. 200 meters = ___ miles. b. 650 in = ____ meters c. 4 years= _____ seconds. d. 200 liters = _____ ml 3. Classify each of the following as units of ...
... 1. Write the most common guidelines to determine significant figures (digits) with an example? 2. Use factor labeling method to convert the following: a. 200 meters = ___ miles. b. 650 in = ____ meters c. 4 years= _____ seconds. d. 200 liters = _____ ml 3. Classify each of the following as units of ...
AP Chemistry Summer Assignment
... summer packet during the first week of school. You can expect a quiz in naming compounds, identifying ions the first day of school. Based on prior knowledge I can tell you that kids are not coming prepared with the material even though the assignment is completed. Make sure just completing assignmen ...
... summer packet during the first week of school. You can expect a quiz in naming compounds, identifying ions the first day of school. Based on prior knowledge I can tell you that kids are not coming prepared with the material even though the assignment is completed. Make sure just completing assignmen ...
Chemical Reactions
... (BrINClHOF) For example, Oxygen is O2 as an element. In a compound, it can’t be a diatomic element because it’s not an element anymore, it’s a compound! ...
... (BrINClHOF) For example, Oxygen is O2 as an element. In a compound, it can’t be a diatomic element because it’s not an element anymore, it’s a compound! ...
Unit 3 - Salina USD 305
... a reaction card. Complete the Find Someone Who Activity in your notebooklet on p. 11 by finding students who match up with the different reaction ...
... a reaction card. Complete the Find Someone Who Activity in your notebooklet on p. 11 by finding students who match up with the different reaction ...
Section 4.6: Double Displacement Reactions
... 7. Silver ions are the only metal ions that can be precipitated from a solution containing the C2H3O2− ions. Therefore, a solution such as NaC2H3O2(aq) can be used to precipitate silver ions from a mixture of dissolved metal ions. 8. Answers may vary. Sample answer: Most of the limescale that forms ...
... 7. Silver ions are the only metal ions that can be precipitated from a solution containing the C2H3O2− ions. Therefore, a solution such as NaC2H3O2(aq) can be used to precipitate silver ions from a mixture of dissolved metal ions. 8. Answers may vary. Sample answer: Most of the limescale that forms ...
Types of Chemical Reactions
... in the fume hoods. These chemicals are toxic and will harm the environment if not disposed of properly. • Do not eat, drink, or apply the chemicals to skin. Many of these chemicals are highly corrosive and in addition to being toxic, they will burn your skin and muscle tissue. Ouch! • If any of the ...
... in the fume hoods. These chemicals are toxic and will harm the environment if not disposed of properly. • Do not eat, drink, or apply the chemicals to skin. Many of these chemicals are highly corrosive and in addition to being toxic, they will burn your skin and muscle tissue. Ouch! • If any of the ...