Angewandte Chemie : International Edition Highly
... The predictions can be further assessed with reference to previous works. Reports of clusters containing multiple carbon and platinum or palladium atoms are scarce. Studies of the adsorption and reactions of CH4 and CO on Ptn clusters[1e] and the structures of PtnOm clusters[1d] have been reported. ...
... The predictions can be further assessed with reference to previous works. Reports of clusters containing multiple carbon and platinum or palladium atoms are scarce. Studies of the adsorption and reactions of CH4 and CO on Ptn clusters[1e] and the structures of PtnOm clusters[1d] have been reported. ...
Comparison for Bonding Situation between Tetrylone and Tetrylene
... W4-SiP2 and therefore, they were not shown in Figure 2. Note that the green/red colors in the figures for Ψk/Ψ-k indicated the sign of the orbitals, and the yellow/blue colors in the deformation density designated charge depletion, and the blue areas pointed to charge accumulation. The charge flo ...
... W4-SiP2 and therefore, they were not shown in Figure 2. Note that the green/red colors in the figures for Ψk/Ψ-k indicated the sign of the orbitals, and the yellow/blue colors in the deformation density designated charge depletion, and the blue areas pointed to charge accumulation. The charge flo ...
molybdenum(O)
... due to the nitrile carbon atoms. This unequivocally indicates that the FN ligand is coordinated to the M (CO)5 moiety through one nitrile nitrogen atom. A symmetric coordination of FN would give only two 13C NMR signals, one for the olefinic and one for the nitrile carbon atoms. It is to be noted th ...
... due to the nitrile carbon atoms. This unequivocally indicates that the FN ligand is coordinated to the M (CO)5 moiety through one nitrile nitrogen atom. A symmetric coordination of FN would give only two 13C NMR signals, one for the olefinic and one for the nitrile carbon atoms. It is to be noted th ...
Tuning PCP-Ir Complexes: The impact of an N
... multiplets that correspond to the diastereotopic protons of the two NCH2 groups appear at δ 4.41–4.24 and 4.69–4.54 ppm for 2, and δ 5.00–4.84 and 4.64–4.50 ppm for 3. The 13C NMR spectra show triplets at δ 160.6 ppm for 2 and 161.2 ppm for 3, both with a JC-P of 3.4 Hz, which can be attributed to t ...
... multiplets that correspond to the diastereotopic protons of the two NCH2 groups appear at δ 4.41–4.24 and 4.69–4.54 ppm for 2, and δ 5.00–4.84 and 4.64–4.50 ppm for 3. The 13C NMR spectra show triplets at δ 160.6 ppm for 2 and 161.2 ppm for 3, both with a JC-P of 3.4 Hz, which can be attributed to t ...
Organometallics - X-Ray - University of Kentucky
... and DFT computational studies. It does not appear that 4 possesses agostic interactions in solution. High oxidation state transition metal complexes containing double metal-carbon bonds (usually termed Schrock1 carbenes or alkylidenes) have attracted significant attention,2 in part because of their ...
... and DFT computational studies. It does not appear that 4 possesses agostic interactions in solution. High oxidation state transition metal complexes containing double metal-carbon bonds (usually termed Schrock1 carbenes or alkylidenes) have attracted significant attention,2 in part because of their ...
M - Chemistry
... can be extremely dramatic. There are cases known where the presence of a chelate will change the equilibrium constant by a factor of 1010 favoring the coordinated form of the ligand to the metal over the dissociated form. Naturally, the longer and more flexible the bridging group in a chelating liga ...
... can be extremely dramatic. There are cases known where the presence of a chelate will change the equilibrium constant by a factor of 1010 favoring the coordinated form of the ligand to the metal over the dissociated form. Naturally, the longer and more flexible the bridging group in a chelating liga ...
Pi-Acid handout - U of L Class Index
... Unlike CO, metal coordination can either raise or lower n(CN) (2000-2200 cm-1) because the HOMO of CNR is much more antibonding than that of CO. ...
... Unlike CO, metal coordination can either raise or lower n(CN) (2000-2200 cm-1) because the HOMO of CNR is much more antibonding than that of CO. ...
Drawing Organic Structures Functional Groups Constitutional Isomers
... • Benzene will reduce at high pressure and temperature or ...
... • Benzene will reduce at high pressure and temperature or ...
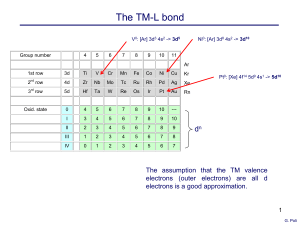
Organometallic compounds of the d-block elements
... Explain the term "ligand cone angle". Which of the following have the largest and smallest ligand cone angles, P(Ph)3, P(o-tolyl)3 and PH3. Give metal valence electron counts for the following systems and indicate those which are likely to be stable and those which are not; [Mn(5-C5H5)(CO)3], [Co( ...
... Explain the term "ligand cone angle". Which of the following have the largest and smallest ligand cone angles, P(Ph)3, P(o-tolyl)3 and PH3. Give metal valence electron counts for the following systems and indicate those which are likely to be stable and those which are not; [Mn(5-C5H5)(CO)3], [Co( ...
CHEM 494 Lecture 10b - UIC Department of Chemistry
... Quaternary Ammonium Salts (R4N+ X-) Nitrogen atoms are positively charged when bonded to four substituents: when all of these substituents are carbon, the molecule is referred to as a quaternary ammonium ion. ...
... Quaternary Ammonium Salts (R4N+ X-) Nitrogen atoms are positively charged when bonded to four substituents: when all of these substituents are carbon, the molecule is referred to as a quaternary ammonium ion. ...
UJDIPa - The Vital Chemist
... All metals form complexes, although the extent of formation and nature of these depend very largely on the electronic structure of the metal. The complexes contain: ...
... All metals form complexes, although the extent of formation and nature of these depend very largely on the electronic structure of the metal. The complexes contain: ...
HYPERVALENT IODINE IN CARBON-CARBON BOND
... Hypervalent iodine compounds have been known since the late 19th century, and their use in organic synthesis as mild oxidizing agents, particularly IBX and its derivatives such as Dess-Martin periodinane, has become widespread. In recent years there has been an intensified interest in the use of hyp ...
... Hypervalent iodine compounds have been known since the late 19th century, and their use in organic synthesis as mild oxidizing agents, particularly IBX and its derivatives such as Dess-Martin periodinane, has become widespread. In recent years there has been an intensified interest in the use of hyp ...
Activation of C–F bonds using Cp*2ZrH2: a
... Examination of a different olefin helps differentiate these pathways. Reaction of 1 with 1,1-difluoromethylenecyclohexane only occurs slowly upon heating to 80 ⬚C. One observes both the monofluoro and the difluoro reduction products, as shown in eqn. (1). This observation is inconsistent with the H/F meta ...
... Examination of a different olefin helps differentiate these pathways. Reaction of 1 with 1,1-difluoromethylenecyclohexane only occurs slowly upon heating to 80 ⬚C. One observes both the monofluoro and the difluoro reduction products, as shown in eqn. (1). This observation is inconsistent with the H/F meta ...
High-spin Cyclopentadienyl Complexes, Part 6. σ/π
... 180 ppm [17], whereas the signal of the mesityl ipso-C atom in the dimethylaminoethylcyclopentadienyl complex [(H4 C5 CH2 CH2 NMe2 )Ni(C6 H2 Me3 -2,4,6)] appears at 125.4 ppm [18]. The crystal structure of 5 shows that the sandwich part of the molecule is very similar to the corresponding part of th ...
... 180 ppm [17], whereas the signal of the mesityl ipso-C atom in the dimethylaminoethylcyclopentadienyl complex [(H4 C5 CH2 CH2 NMe2 )Ni(C6 H2 Me3 -2,4,6)] appears at 125.4 ppm [18]. The crystal structure of 5 shows that the sandwich part of the molecule is very similar to the corresponding part of th ...
4 Investigation of substituent effect on
... Besides the above properties, quite a few theoretical investigations have focused on the effect of substituents on the carbene ligand. Several studies have shown that the chemical reactivity of the Fischer-type Cr-carbene complexes [Cr(CO)5{C(X)R}] depends mainly on the electronic characteristics of ...
... Besides the above properties, quite a few theoretical investigations have focused on the effect of substituents on the carbene ligand. Several studies have shown that the chemical reactivity of the Fischer-type Cr-carbene complexes [Cr(CO)5{C(X)R}] depends mainly on the electronic characteristics of ...
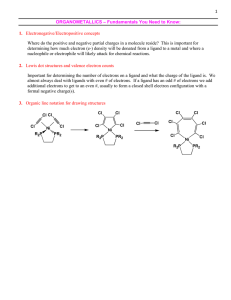
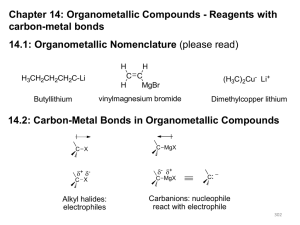
Organometallic Compounds - Reagents
... the hypothetical cleavage of a bond back to precursors of the target molecule. Functional Group Interconversion (FGI): the process of converting one functional group into another by substitution, addition, elimination, reduction, or oxidation ...
... the hypothetical cleavage of a bond back to precursors of the target molecule. Functional Group Interconversion (FGI): the process of converting one functional group into another by substitution, addition, elimination, reduction, or oxidation ...
Grignard Reagents
... the hypothetical cleavage of a bond back to precursors of the target molecule. Functional Group Interconversion (FGI): the process of converting one functional group into another by substitution, addition, elimination, reduction, or oxidation ...
... the hypothetical cleavage of a bond back to precursors of the target molecule. Functional Group Interconversion (FGI): the process of converting one functional group into another by substitution, addition, elimination, reduction, or oxidation ...
Chapter 4: Structural features of rhenium carbene complexes
... for the equatorial carbonyl ligands of the two rhenium fragments are the same within experimental error (for [Re2(CO)10] the average eq–CO metal–carbon distance is 1.987 (6) Å). On the {Re(CO)4} fragment, that bears the carbene ligand, there are two carbonyl ligands that are trans to non–carbonyl li ...
... for the equatorial carbonyl ligands of the two rhenium fragments are the same within experimental error (for [Re2(CO)10] the average eq–CO metal–carbon distance is 1.987 (6) Å). On the {Re(CO)4} fragment, that bears the carbene ligand, there are two carbonyl ligands that are trans to non–carbonyl li ...
Versatile Cooperative Ligand Effects in Group 9 Transition Metal
... transfer processes could potentially have a profound impact on catalysis, the initial aim of the research described in this thesis was to develop rhodium and iridium complexes containing such cooperative ligand systems and to study their application in (de)hydrogenation-related catalysis. Secondary ...
... transfer processes could potentially have a profound impact on catalysis, the initial aim of the research described in this thesis was to develop rhodium and iridium complexes containing such cooperative ligand systems and to study their application in (de)hydrogenation-related catalysis. Secondary ...
Chapter 1 - University of Amsterdam
... In certain complexes with redox non-innocent ligands the relative metal and ligand orbital energies can be reversed (Figure 4, bottom). For complexes containing oxidizable ligands, the metal d-orbitals can end-up lower in energy than the ligand orbitals, thus effectively leading to reduction of the ...
... In certain complexes with redox non-innocent ligands the relative metal and ligand orbital energies can be reversed (Figure 4, bottom). For complexes containing oxidizable ligands, the metal d-orbitals can end-up lower in energy than the ligand orbitals, thus effectively leading to reduction of the ...
Palladation of diimidazolium salts at the C4 position
... The organometallic chemistry of N-heterocyclic carbenes (NHCs)1 has provided an entirely new twist on the development of active catalysts2 and materials for optical and electronic applications.3 The success of NHCs in these areas is quite generally ascribed to the relative4 stability of the M–CNHC b ...
... The organometallic chemistry of N-heterocyclic carbenes (NHCs)1 has provided an entirely new twist on the development of active catalysts2 and materials for optical and electronic applications.3 The success of NHCs in these areas is quite generally ascribed to the relative4 stability of the M–CNHC b ...
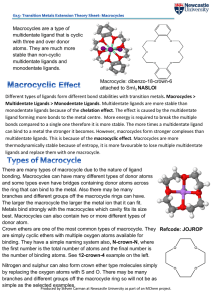
Different types of ligands form different bond stabilities with transition
... There are many types of macrocycle due to the nature of ligand bonding. Macrocycles can have many different types of donor atoms and some types even have bridges containing donor atoms across the ring that can bind to the metal. Also there may be many branches and different groups off the macrocycle ...
... There are many types of macrocycle due to the nature of ligand bonding. Macrocycles can have many different types of donor atoms and some types even have bridges containing donor atoms across the ring that can bind to the metal. Also there may be many branches and different groups off the macrocycle ...
Document
... Pd(0) and Pd(II) are both capable of interacting with unsaturated systems such as alkenes or alkynes via p-bonding. The two types of complexes are however different in nature. Pd(0) is highly electron-rich and back-donates to the ligand (Pd L), whereas Pd(II) is electrophilic, and its main interac ...
... Pd(0) and Pd(II) are both capable of interacting with unsaturated systems such as alkenes or alkynes via p-bonding. The two types of complexes are however different in nature. Pd(0) is highly electron-rich and back-donates to the ligand (Pd L), whereas Pd(II) is electrophilic, and its main interac ...
Organic Chemistry
... • A useful alkene methathesis catalyst consists of ruthenium, Ru, complexed with nucleophilic carbenes and another carbenoid ligand. • In this example, the other carbenoid ligand is a ...
... • A useful alkene methathesis catalyst consists of ruthenium, Ru, complexed with nucleophilic carbenes and another carbenoid ligand. • In this example, the other carbenoid ligand is a ...
Persistent carbene

A persistent carbene (also known as stable carbene) is a type of carbene demonstrating particular stability. The best-known examples and by far largest subgroup are the N-heterocyclic carbenes (sometimes called Arduengo carbenes), for example diaminocarbenes with the general formula (R2N)2C:, where the 'R's are typically alkyl and aryl groups. The groups can be linked to give heterocyclic carbenes, such as those derived from imidazole, imidazoline, thiazole or triazole.Traditionally carbenes are viewed as so reactive that were only studied indirectly, e.g. by trapping reactions. This situation has changed dramatically with the emergence of persistent carbenes. Although they are fairly reactive substances, i.e., undergoing dimerization, many can be isolated as pure substances.Persistent carbenes can exist in the singlet or the triplet states with the singlet state carbenes being more stable. The relative stability of these compounds is only partly due to steric hindrance by bulky groups. Some singlet carbenes are thermodynamically stable in the absence of moisture and (in most cases) oxygen, and can be isolated and indefinitely stored. Others are not thermodynamically stable and will dimerise slowly over days. The less stable triplet state carbenes have half-lives measured in seconds, and therefore can be observed but not stored.