Flammability Limits _MS thesis_
... analysis from obtained experimental data, which includes modification of Le Chatelier’s Law if necessary, or quantitatively relating the observed flammability limits to stoichiometric concentrations of fuel mixtures, or connecting combustion heat with the measured flammability limits, external condi ...
... analysis from obtained experimental data, which includes modification of Le Chatelier’s Law if necessary, or quantitatively relating the observed flammability limits to stoichiometric concentrations of fuel mixtures, or connecting combustion heat with the measured flammability limits, external condi ...
c00kieee - Ritter Illustration
... Uranium ore is typically mined or leached in situ. For the mined ore, the raw material is milled to produce a high surface area slurry, which is then treated with H2 SO4 . The sulfuric acid oxidizes the uranium to the soluble hexavalent state. The addition of base to the solution precipitates an oxi ...
... Uranium ore is typically mined or leached in situ. For the mined ore, the raw material is milled to produce a high surface area slurry, which is then treated with H2 SO4 . The sulfuric acid oxidizes the uranium to the soluble hexavalent state. The addition of base to the solution precipitates an oxi ...
Oxygen Carriers Materials for Chemical
... Fe2O3 and Mn2O3 showed signs of deactivation as function of cycle number, likely due to the formation of metallic silicates. Only NiO showed high selectivity toward H2. All the MgAl2O4-supported metal oxides exhibited high reactivity under reducing and oxidizing conditions. In contrast to the SiO2 b ...
... Fe2O3 and Mn2O3 showed signs of deactivation as function of cycle number, likely due to the formation of metallic silicates. Only NiO showed high selectivity toward H2. All the MgAl2O4-supported metal oxides exhibited high reactivity under reducing and oxidizing conditions. In contrast to the SiO2 b ...
Catalysts Containing Depleted Uranium Compounds
... The prevailing consumption of uranium in the form of uranium dioxide is made by the enterprises of atomic power engineering, thus oxygen-containing compounds of uranium were investigated most completely with the purpose of manufacturing nuclear fuel. At the same time, due to the aforementioned varie ...
... The prevailing consumption of uranium in the form of uranium dioxide is made by the enterprises of atomic power engineering, thus oxygen-containing compounds of uranium were investigated most completely with the purpose of manufacturing nuclear fuel. At the same time, due to the aforementioned varie ...
Cyanide Destruction with Chlorine Dioxide
... should be considered when levels of oxidizable cyanide are too high to be processed by a biological treatment system. Chlorine dioxide Chlorine dioxide is unique in its ability to be used at moderately basic pH's. All other oxidants require pH's greater than 12. In the case of chlorine this is neces ...
... should be considered when levels of oxidizable cyanide are too high to be processed by a biological treatment system. Chlorine dioxide Chlorine dioxide is unique in its ability to be used at moderately basic pH's. All other oxidants require pH's greater than 12. In the case of chlorine this is neces ...
Combustion characteristics
... allows the flame to go backwards and penetrate the porous media, which is being heated by the slowly moving flame front. The process is also known as filtration combustion. Notice that the flame-front temperature is lower than the adiabatic value when the front moves backwards, but, if the gas injec ...
... allows the flame to go backwards and penetrate the porous media, which is being heated by the slowly moving flame front. The process is also known as filtration combustion. Notice that the flame-front temperature is lower than the adiabatic value when the front moves backwards, but, if the gas injec ...
Reactions of Plutonium Dioxide with Water and Oxygen
... to 50, 100, 150, and 250°C over a period in excess of 110 hours. Water vapor was introduced into the evacuated balantxtchamber via a gas manifold system and was maintained at a constant pressure with a water reservoir held at a fixed temperature. A constant sample temperature was maintained with a p ...
... to 50, 100, 150, and 250°C over a period in excess of 110 hours. Water vapor was introduced into the evacuated balantxtchamber via a gas manifold system and was maintained at a constant pressure with a water reservoir held at a fixed temperature. A constant sample temperature was maintained with a p ...
1. dia
... Chemistry of sulfur oxide formation Burning of elemental sulfur: oxygen atom starts the reaction, transition compound: sulfur monoxide. O + S2 = SO + S O + SO = SO2 Combustion of sulfur containing materials (fuel-S) : thermal decay → char-S, (shrunken solid fuel) hydrogen sulfide (H2S) and carbonyl ...
... Chemistry of sulfur oxide formation Burning of elemental sulfur: oxygen atom starts the reaction, transition compound: sulfur monoxide. O + S2 = SO + S O + SO = SO2 Combustion of sulfur containing materials (fuel-S) : thermal decay → char-S, (shrunken solid fuel) hydrogen sulfide (H2S) and carbonyl ...
module-iv --- combustion thermodynamic applied termodynamics
... All conventional fossil fuels, whether, solid, liquid or gaseous, contain basically carbon and hydrogen which invariably react with the oxygen in the air forming carbon dioxide, carbon monoxide or water vapour. The heat energy released as a result of combustion can be utilized for heating purposes o ...
... All conventional fossil fuels, whether, solid, liquid or gaseous, contain basically carbon and hydrogen which invariably react with the oxygen in the air forming carbon dioxide, carbon monoxide or water vapour. The heat energy released as a result of combustion can be utilized for heating purposes o ...
REVIEW OF HYDROGEN CONVERSION TECHNOLOGIES Abstract: F. Barbir Clean Energy Research Institute
... salt, with carbonate ions providing ionic conduction. At such high operating temperatures, noble metal catalysts are typically not required [5,6]. Solid oxide fuel cells (SOFC) use a solid, nonporous metal oxide, usually Y2O3-stabilized ZrO2 as the electrolyte. The cell operates at 900-1000°C where ...
... salt, with carbonate ions providing ionic conduction. At such high operating temperatures, noble metal catalysts are typically not required [5,6]. Solid oxide fuel cells (SOFC) use a solid, nonporous metal oxide, usually Y2O3-stabilized ZrO2 as the electrolyte. The cell operates at 900-1000°C where ...
Chapter Three The hydrogen fuel cell power system
... suitably fast reaction kinetics, these are the two most often chosen. All further examples will use hydrogen and oxygen unless otherwise noted. ...
... suitably fast reaction kinetics, these are the two most often chosen. All further examples will use hydrogen and oxygen unless otherwise noted. ...
Series 9
... A check on both the accuracy of the given molar analyses and the calculations conducted to determine the unknown coefficients is obtained by applying conservation of mass to nitrogen. The amount of nitrogen in the reactants is 0.105 ⫹ 13.76212.8922 ⫽ 10.98 kmolⲐkmol of fuel The amount of nitrogen in ...
... A check on both the accuracy of the given molar analyses and the calculations conducted to determine the unknown coefficients is obtained by applying conservation of mass to nitrogen. The amount of nitrogen in the reactants is 0.105 ⫹ 13.76212.8922 ⫽ 10.98 kmolⲐkmol of fuel The amount of nitrogen in ...
Fuel Cells in Energy Production Xiaoyu Huang Bachelor’s Thesis
... A large power plant such as thermal power plant can obtain high efficiency. However, giant units of the power plant are affected by various limitations, so customers can achieve power from an electrical grid in which the power plant makes concentrated power. The big unit’s inflexible generation cann ...
... A large power plant such as thermal power plant can obtain high efficiency. However, giant units of the power plant are affected by various limitations, so customers can achieve power from an electrical grid in which the power plant makes concentrated power. The big unit’s inflexible generation cann ...
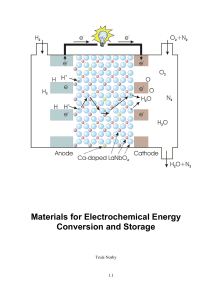
Materials for Electrochemical Energy Conversion and Storage
... overpotentials serve to increase the applied voltage. Obviously, the goal of materials research on electrochemical energy conversion and storage devices is to lower the ohmic and kinetic resistances as much as possible, until they become small compared with the external load. The resistance of an el ...
... overpotentials serve to increase the applied voltage. Obviously, the goal of materials research on electrochemical energy conversion and storage devices is to lower the ohmic and kinetic resistances as much as possible, until they become small compared with the external load. The resistance of an el ...
Chemical Reactions Definitions Air Fuel Ratio
... hydrocarbons and are denoted by the general formula of CnHm. For example octane is C8H18. Combustion: is a chemical reaction during which a fuel is oxidized and a large quantity of energy is released. The oxidizer is often air. Reactants: are the components that exist before the reaction. Products: ...
... hydrocarbons and are denoted by the general formula of CnHm. For example octane is C8H18. Combustion: is a chemical reaction during which a fuel is oxidized and a large quantity of energy is released. The oxidizer is often air. Reactants: are the components that exist before the reaction. Products: ...
New Dust Burner: Unique System Solution for Special Customer
... which we have parameterised permits an automatic know-how and the required equipment to construct fuel switch-over between pulverous and liquid fuels. and test the dust burners. In this combination, Hans “The decisive factor thereby is however, that with ...
... which we have parameterised permits an automatic know-how and the required equipment to construct fuel switch-over between pulverous and liquid fuels. and test the dust burners. In this combination, Hans “The decisive factor thereby is however, that with ...
the optimization of proton exchange membrane hydrogen fuel cells
... oxidation has only one possible mechanism and therefore the selectivity does not affect the anode [9]. There are two possible mechanisms at the cathode for the oxygen reduction and the pathway taken is determined by the selectivity of the catalyst [9]. One pathway involves a four-electron oxidation ...
... oxidation has only one possible mechanism and therefore the selectivity does not affect the anode [9]. There are two possible mechanisms at the cathode for the oxygen reduction and the pathway taken is determined by the selectivity of the catalyst [9]. One pathway involves a four-electron oxidation ...
possibilities and limitations of co-firing of biomass
... • Wastes can be utilized • Biofuels and wastes can be converted at a high efficiency and with efficient flue gas cleaning • There may be positive synergy effects • Seasonal variations in the supply of additional fuels can be handled, and the impact of quality variations can be Bo Leckner CTH ...
... • Wastes can be utilized • Biofuels and wastes can be converted at a high efficiency and with efficient flue gas cleaning • There may be positive synergy effects • Seasonal variations in the supply of additional fuels can be handled, and the impact of quality variations can be Bo Leckner CTH ...
CHAPTER 8: ENERGY FROM ELECTRON TRANSFER
... b. List two advantages fuel cells have over internal combustion engines. Answer: a. The electrolyte allows ions to move between the electrodes, thus completing the circuit. b. Fuel cells using hydrogen gas (H2) as the fuel produce only water as a product, whereas internal combustion engines produce ...
... b. List two advantages fuel cells have over internal combustion engines. Answer: a. The electrolyte allows ions to move between the electrodes, thus completing the circuit. b. Fuel cells using hydrogen gas (H2) as the fuel produce only water as a product, whereas internal combustion engines produce ...
Uranium
... uranium-238 is about 4.5 billion years, which means it is not very radioactive. In fact, its very long half-life (and thus low radioactivity) is the reason uranium still exists on the Earth. Three additional isotopes of uranium are not naturally present but can be produced by nuclear transformations ...
... uranium-238 is about 4.5 billion years, which means it is not very radioactive. In fact, its very long half-life (and thus low radioactivity) is the reason uranium still exists on the Earth. Three additional isotopes of uranium are not naturally present but can be produced by nuclear transformations ...
Auto-Ignition Characteristics of Different Fuels with IQT, and Pentane
... using the ignition quality tester (IQT), and auto-ignition characteristics of pentane isomers using the cooperative fuel research (CFR) engine. The ignition delay (ID) and derived cetane number (DCN) measured from IQT are important in diesel fuel formulation since the rate of burning is essential in ...
... using the ignition quality tester (IQT), and auto-ignition characteristics of pentane isomers using the cooperative fuel research (CFR) engine. The ignition delay (ID) and derived cetane number (DCN) measured from IQT are important in diesel fuel formulation since the rate of burning is essential in ...
LGray_MMEES_F2014_Project_SOFC-GT_Cycle_Perf_Mode+
... SOFC-GT can produce 500kW at nearly twice the overall efficiency as a stand-alone gas turbine power unit, (56 percent compared to 28.5percent). It was also shown that the carbon dioxide emissions produced by the SOFC-GT were 40 percent lower than the GT. ...
... SOFC-GT can produce 500kW at nearly twice the overall efficiency as a stand-alone gas turbine power unit, (56 percent compared to 28.5percent). It was also shown that the carbon dioxide emissions produced by the SOFC-GT were 40 percent lower than the GT. ...
- International Journal of Multidisciplinary Research and
... temperature should be 400-500 Cº but increasing the temperature would give more desirable results. Lowering the pressure has a positive impact on the process. Typically, The reaction performs preferably at lower pressure. The type of catalyst also influences the reaction. The better the catalyst pre ...
... temperature should be 400-500 Cº but increasing the temperature would give more desirable results. Lowering the pressure has a positive impact on the process. Typically, The reaction performs preferably at lower pressure. The type of catalyst also influences the reaction. The better the catalyst pre ...
- E3S Web of Conferences
... The test results of RDF fuel gasification containing formulation Bio-CONOx are shown in Fig. 2 and 3. Fig. 2 shows the dependence of the of generator gas composition CH4, CO, H2, CO2 from the amount of oxygen. Taking into account that the air flow during the gasification process was maintained at a ...
... The test results of RDF fuel gasification containing formulation Bio-CONOx are shown in Fig. 2 and 3. Fig. 2 shows the dependence of the of generator gas composition CH4, CO, H2, CO2 from the amount of oxygen. Taking into account that the air flow during the gasification process was maintained at a ...