Module P7.3 Internal energy, heat and energy transfer
... quantity of air trapped in a bicycle pump might be at a high temperature or a low one, its volume might be relatively large or relatively small, and its pressure might be quite high or quite low. By quoting values for these macroscopic properties (temperature, volume and pressure), or at least for a ...
... quantity of air trapped in a bicycle pump might be at a high temperature or a low one, its volume might be relatively large or relatively small, and its pressure might be quite high or quite low. By quoting values for these macroscopic properties (temperature, volume and pressure), or at least for a ...
the concept of exergy and energy quality
... fact, the mathematical representation of Kelvin-Planck ( Wcycle 0 ) indicates that no work can be produced, and equality ( Wcycle 0 ) only applies to the reversible case. ...
... fact, the mathematical representation of Kelvin-Planck ( Wcycle 0 ) indicates that no work can be produced, and equality ( Wcycle 0 ) only applies to the reversible case. ...
Statistical and Low Temperature Physics (PHYS393)
... magnetic moments coming from their electrons. At mK temperatures, the magnetic disorder entropy (about 1 J/mol) is large compared to all other entropies, such as lattice and coduction electron entropies, which may be neglected. We have previously looked at the properties of a paramagnetic salt. The ...
... magnetic moments coming from their electrons. At mK temperatures, the magnetic disorder entropy (about 1 J/mol) is large compared to all other entropies, such as lattice and coduction electron entropies, which may be neglected. We have previously looked at the properties of a paramagnetic salt. The ...
Wilson-Ch
... 11.2 Specific Heat and Calorimetry Calorimetry is the quantitative measurement of heat exchange; it is done using a calorimeter. A calorimeter is insulated from the environment, minimizing heat exchange. Therefore, heat lost by one object in the calorimeter must be gained by another. This is one wa ...
... 11.2 Specific Heat and Calorimetry Calorimetry is the quantitative measurement of heat exchange; it is done using a calorimeter. A calorimeter is insulated from the environment, minimizing heat exchange. Therefore, heat lost by one object in the calorimeter must be gained by another. This is one wa ...
T - UCSD Physics
... (b) What is the work W done per cycle? Express your answer in terms of the temperatures T1 and T2 , the ratios x and r, and the number of moles of gas ν. (c) Suppose VA = 1.00 L and VB = 10.0 L. Suppose further that T1 = 50◦ C and T2 = 400◦ C. At what value of VA′ would the work done per cycle vanis ...
... (b) What is the work W done per cycle? Express your answer in terms of the temperatures T1 and T2 , the ratios x and r, and the number of moles of gas ν. (c) Suppose VA = 1.00 L and VB = 10.0 L. Suppose further that T1 = 50◦ C and T2 = 400◦ C. At what value of VA′ would the work done per cycle vanis ...
as PDF
... isothermal processes, respectively, with T1 < TH and TL < T2. There is also a heat loss Q from the hot reservoir to the cold reservoir and there are other internal irreversibilities (such as dissipative processes inside the working fluid). This Carnot-like model was chosen because of its simplicity ...
... isothermal processes, respectively, with T1 < TH and TL < T2. There is also a heat loss Q from the hot reservoir to the cold reservoir and there are other internal irreversibilities (such as dissipative processes inside the working fluid). This Carnot-like model was chosen because of its simplicity ...
energy and power - Beck-Shop
... Heat is a form of energy. It has the capacity to do work directly as thermal warming or by conversion to other energy forms, mainly mechanical. The quantity of heat involved in a process can be measured by some effect which accompanies the process. Traditional units of measurement for the quantity o ...
... Heat is a form of energy. It has the capacity to do work directly as thermal warming or by conversion to other energy forms, mainly mechanical. The quantity of heat involved in a process can be measured by some effect which accompanies the process. Traditional units of measurement for the quantity o ...
Calorimetry Measurement
... there is a surplus of thermal energy (i.e., of fast molecules or electrons, or a higher density of phonons) in some area, some of it will flow toward areas with a lower thermal energy density until thermal equilibrium has been established. This flow of energy P (in W = J s–1) is called heat flow. He ...
... there is a surplus of thermal energy (i.e., of fast molecules or electrons, or a higher density of phonons) in some area, some of it will flow toward areas with a lower thermal energy density until thermal equilibrium has been established. This flow of energy P (in W = J s–1) is called heat flow. He ...
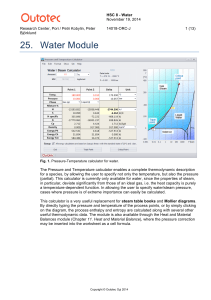
Water - HSC Chemistry 9
... for a species, by allowing the user to specify not only the temperature, but also the pressure (partial). This calculator is currently only available for water, since the properties of steam, in particular, deviate significantly from those of an ideal gas, i.e. the heat capacity is purely a temperat ...
... for a species, by allowing the user to specify not only the temperature, but also the pressure (partial). This calculator is currently only available for water, since the properties of steam, in particular, deviate significantly from those of an ideal gas, i.e. the heat capacity is purely a temperat ...
Phase changes
... As a closed system, i.e. keeping invariant the total mass, a two-phase system may be processed at constant volume (by heating or cooling), at constant pressure (and thus temperature; e.g. heating or cooling it in a cylinder-piston arrangement), adiabatically (by a quick compression or expansion), or ...
... As a closed system, i.e. keeping invariant the total mass, a two-phase system may be processed at constant volume (by heating or cooling), at constant pressure (and thus temperature; e.g. heating or cooling it in a cylinder-piston arrangement), adiabatically (by a quick compression or expansion), or ...












![[PDF]](http://s1.studyres.com/store/data/008813344_1-6b54197619c7ffbc0fcfc4fbb7e270fc-300x300.png)