Lecture 11 - Laws of Thermodynamics
... It is easy to produce thermal energy using work, but how does one produce work using thermal energy? This is a heat engine; mechanical energy can be obtained from thermal energy only when heat can flow from a higher temperature to a lower temperature. ...
... It is easy to produce thermal energy using work, but how does one produce work using thermal energy? This is a heat engine; mechanical energy can be obtained from thermal energy only when heat can flow from a higher temperature to a lower temperature. ...
Thermodynamics and Irreversibility
... • For irreversible paths, we will get a difference in the calculation of δ W . This difference of work calculation will change the “predicted” reached point in the Clapeyron diagram; for example, shall we reach the temperature T1 or T2? See Figure 8. • Then, to calculate the difference of internal e ...
... • For irreversible paths, we will get a difference in the calculation of δ W . This difference of work calculation will change the “predicted” reached point in the Clapeyron diagram; for example, shall we reach the temperature T1 or T2? See Figure 8. • Then, to calculate the difference of internal e ...
A System and Its Surroundings
... saucepan. A thermos is used to keep things either cold or hot. Thus, a thermos does not allow for energy transfer. Additionally, the thermos, like any other closed container, does not allow matter transfer because it has a lid that does not allow anything to enter or leave the container. As a result ...
... saucepan. A thermos is used to keep things either cold or hot. Thus, a thermos does not allow for energy transfer. Additionally, the thermos, like any other closed container, does not allow matter transfer because it has a lid that does not allow anything to enter or leave the container. As a result ...
Second Order Phase Transitions
... The Ising ferromagnet shows a second order transition. Features are 1. A new state grows continuously out of the previous one: for T → Tc the two states become quantitatively the same. 2. As a consequence of (1) the thermodynamic potentials F, U, S . . . are continuous at Tc but not necessarily smoo ...
... The Ising ferromagnet shows a second order transition. Features are 1. A new state grows continuously out of the previous one: for T → Tc the two states become quantitatively the same. 2. As a consequence of (1) the thermodynamic potentials F, U, S . . . are continuous at Tc but not necessarily smoo ...
Thermochemistry
... q = msT where m = mass in grams of the body. Or sometimes H = msT. There is also an energy change when a substance changes phase. These energies are typically reported as molar heat of fusion, Hfus and molar heat of vaporization, Hvap and are reported in kJ/mole of heat needed to melt or vapori ...
... q = msT where m = mass in grams of the body. Or sometimes H = msT. There is also an energy change when a substance changes phase. These energies are typically reported as molar heat of fusion, Hfus and molar heat of vaporization, Hvap and are reported in kJ/mole of heat needed to melt or vapori ...
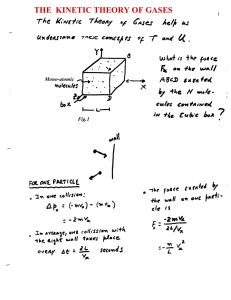
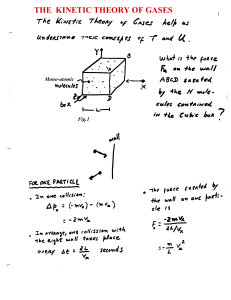
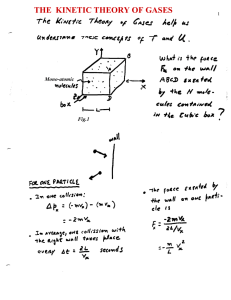
The kinetic theory of the gases
... Once the equation of state of a system is known, different thermodynamic processes can be studies in detail. Here let’s consider an adiabatic process ...
... Once the equation of state of a system is known, different thermodynamic processes can be studies in detail. Here let’s consider an adiabatic process ...
Chapter 22 Problems
... installed backward. It extracts energy from colder air outside and deposits it in a warmer room. Suppose that the ratio of the actual energy entering the room to the work done by the device’s motor is 10.0% of the theoretical maximum ratio. Determine the energy entering the room per joule of work do ...
... installed backward. It extracts energy from colder air outside and deposits it in a warmer room. Suppose that the ratio of the actual energy entering the room to the work done by the device’s motor is 10.0% of the theoretical maximum ratio. Determine the energy entering the room per joule of work do ...
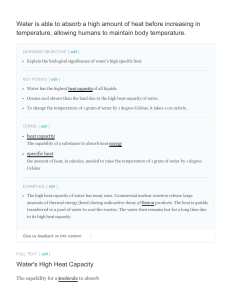
Water is able to absorb a high amount of heat before
... heat energy is called heat capacity, which can be calculated by the equation shown in the figure . Water's high heat capacity is a property caused by hydrogen bonding among water molecules. When heat is absorbed, hydrogen bonds are broken and water molecules can move freely. When the temperature of ...
... heat energy is called heat capacity, which can be calculated by the equation shown in the figure . Water's high heat capacity is a property caused by hydrogen bonding among water molecules. When heat is absorbed, hydrogen bonds are broken and water molecules can move freely. When the temperature of ...
Thermochemistry, thermodynamics Thermochemistry
... The thermodynamic state of a system is defined by a set of conditions that completely specifies all the properties of the system. This set is commonly includes the temperature, pressure, composition, and physical state (gas, liquid or solid) of each part of the system. The properties of a system – s ...
... The thermodynamic state of a system is defined by a set of conditions that completely specifies all the properties of the system. This set is commonly includes the temperature, pressure, composition, and physical state (gas, liquid or solid) of each part of the system. The properties of a system – s ...