![BTD QUESTION BANK[1].](http://s1.studyres.com/store/data/009330461_1-f5de3108f7a7a17ebe3a8cbd391865db-300x300.png)
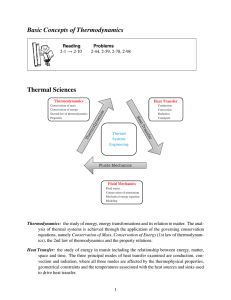
CHAPTER 4: PHASE TRANSITIONS
... Let us consider a system containing a mixture of substances 1,2, 3,…….k. If some quantities of a substance are added to the system, the energy of the system will increase. Thus for a system with variable composition, the internal energy depends not only on S and V , but also on the number of moles ( ...
... Let us consider a system containing a mixture of substances 1,2, 3,…….k. If some quantities of a substance are added to the system, the energy of the system will increase. Thus for a system with variable composition, the internal energy depends not only on S and V , but also on the number of moles ( ...
The Local-Nonequilibrium Temperature Field
... where brackets denote the difference in a value ahead of and behind the interface, V is the interface velocity, and Q is the total heat released at the interface. The interface conditions, Eqs. (6) and (7), for the hyperbolic phase change problem, Eqs. (3) and (4), clearly demonstrate that the inter ...
... where brackets denote the difference in a value ahead of and behind the interface, V is the interface velocity, and Q is the total heat released at the interface. The interface conditions, Eqs. (6) and (7), for the hyperbolic phase change problem, Eqs. (3) and (4), clearly demonstrate that the inter ...
Lecture 2 Intro to Heat Flow
... The rate of heat flow is proportional to the difference in heat between two bodies. A thin plate of thickness z with temperature difference ∆T experiences heat flow Q: ...
... The rate of heat flow is proportional to the difference in heat between two bodies. A thin plate of thickness z with temperature difference ∆T experiences heat flow Q: ...
thermodynamics
... simplicity, the system to be a certain mass of gas contained in a cylinder with a movable piston as shown in Fig. 12.4. Experience shows there are two ways of changing the state of the gas (and hence its internal energy). One way is to put the cylinder in contact with a body at a higher temperature ...
... simplicity, the system to be a certain mass of gas contained in a cylinder with a movable piston as shown in Fig. 12.4. Experience shows there are two ways of changing the state of the gas (and hence its internal energy). One way is to put the cylinder in contact with a body at a higher temperature ...
Chapter 3: The First Law of Thermodynamics for Closed Systems a
... energy resulting from the transfer of heat or work. Since specific internal energy is a property of the system, it is usually presented in the Property Tables such as in the Steam Tables. Consider for example the following solved problem. Solved Problem 3.1 - Recall the Solved Problem 2.2 in Chapter ...
... energy resulting from the transfer of heat or work. Since specific internal energy is a property of the system, it is usually presented in the Property Tables such as in the Steam Tables. Consider for example the following solved problem. Solved Problem 3.1 - Recall the Solved Problem 2.2 in Chapter ...