Final Review 3-8 Answers_2
... a) S8(s) + 8 O2(g) 8 SO2(g) b) S(s) + O2(g) SO2(g) c) SO2(g) + H2O (l) H2SO3(aq) d) SO2(g) + ½ O2(g) SO3(g) 4. The experimental design of most directly determining the mass of solute in a specific volume of solution is called a) titration b) crystallization c) filtration d) distillation 5. I ...
... a) S8(s) + 8 O2(g) 8 SO2(g) b) S(s) + O2(g) SO2(g) c) SO2(g) + H2O (l) H2SO3(aq) d) SO2(g) + ½ O2(g) SO3(g) 4. The experimental design of most directly determining the mass of solute in a specific volume of solution is called a) titration b) crystallization c) filtration d) distillation 5. I ...
Physiological and phylogenetic studies of thermophilic
... The present study is based on two earlier projects. One, supervised by Ólafur Friðjónsson at Prokaria ehf, was based on the study of three mesophilic hydrogen oxidizing (HOX) bacteria, Wautersia eutropha, Hydrogenomonas pseudoflava and Acidovorax facilis. This research emphasised on genetic engineer ...
... The present study is based on two earlier projects. One, supervised by Ólafur Friðjónsson at Prokaria ehf, was based on the study of three mesophilic hydrogen oxidizing (HOX) bacteria, Wautersia eutropha, Hydrogenomonas pseudoflava and Acidovorax facilis. This research emphasised on genetic engineer ...
Chemistry of CHLORINE
... II. Explain the trend in bond Energy of the halogens above: -Decrease down the group from chlorine to Iodine -Atomic radius increase down the group decreasing the energy required to break the covalent bonds between the larger atom with reduced effective nuclear charge an outer energy level that take ...
... II. Explain the trend in bond Energy of the halogens above: -Decrease down the group from chlorine to Iodine -Atomic radius increase down the group decreasing the energy required to break the covalent bonds between the larger atom with reduced effective nuclear charge an outer energy level that take ...
Here
... (h) aluminium sulphide (ionic with some covalent character) (i) phosphorus trichloride (j) potassium phosphate (ionic) (a) NaOCl (ionic) (iii) electrons are set free within a lattice of positive ions A chemical reaction of oxygen requires breaking the oxygen‐oxygen double bond, and generally ...
... (h) aluminium sulphide (ionic with some covalent character) (i) phosphorus trichloride (j) potassium phosphate (ionic) (a) NaOCl (ionic) (iii) electrons are set free within a lattice of positive ions A chemical reaction of oxygen requires breaking the oxygen‐oxygen double bond, and generally ...
COCRYSTAL FORMATION OF PARACETAMOL WITH INDOMETHACIN AND MEFENAMIC ACID:
... The design of co-crystals seems to be straight forward because donor and acceptor functionalities can be brought together more easily than with single component systems since the partners are more accessible to arrange themselves into an optimal geometry, leading to favorable intermolecular interact ...
... The design of co-crystals seems to be straight forward because donor and acceptor functionalities can be brought together more easily than with single component systems since the partners are more accessible to arrange themselves into an optimal geometry, leading to favorable intermolecular interact ...
Quantitative Comparison of the Hydrogen Bond
... flexibility, and the lack of stable tertiary contacts (1-4). Besides a general interest in the understanding of interactions responsible for protein stabilization, the research of partially folded states is motivated by the finding that certain of these destabilized structures represent equilibrium ...
... flexibility, and the lack of stable tertiary contacts (1-4). Besides a general interest in the understanding of interactions responsible for protein stabilization, the research of partially folded states is motivated by the finding that certain of these destabilized structures represent equilibrium ...
Thermal Decomposition of the Non-Interstitial Hydrides for the
... of materials for hydrogen storage, namely the catalytically enhanced complex metal hydrides. We support our predictions by thermodynamics considerations, calculations derived from molecular orbital (MO) theory and backed up by simple chemical insights and intuition. ...
... of materials for hydrogen storage, namely the catalytically enhanced complex metal hydrides. We support our predictions by thermodynamics considerations, calculations derived from molecular orbital (MO) theory and backed up by simple chemical insights and intuition. ...
Growth and characterization of pure and disodium hydrogen
... peaks and also by the broadening and shift in the vibrational absorption frequencies of the FTIR spectra. The complexity of infrared spectra in 1450 to 600 cm-1 region makes it difficult to assign all the absorption bands, because of the unique patterns found there, it is often called the fingerprin ...
... peaks and also by the broadening and shift in the vibrational absorption frequencies of the FTIR spectra. The complexity of infrared spectra in 1450 to 600 cm-1 region makes it difficult to assign all the absorption bands, because of the unique patterns found there, it is often called the fingerprin ...
The SimSoup Guide - Chris Gordon
... Atoms are of different types. These types are called elements. An atom has a mass and a number of electrons that orbit a nucleus. Some of these are valence electrons that can participate in bonds with other atoms. ...
... Atoms are of different types. These types are called elements. An atom has a mass and a number of electrons that orbit a nucleus. Some of these are valence electrons that can participate in bonds with other atoms. ...
CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... When zinc reacts with hydrochloric acid, hydrogen gas is released. The volume of gas produced can be recorded at regular time intervals. The illustration shows the apparatus used and the results. Q4: What volume of hydrogen is released in the first 5 seconds? ...
... When zinc reacts with hydrochloric acid, hydrogen gas is released. The volume of gas produced can be recorded at regular time intervals. The illustration shows the apparatus used and the results. Q4: What volume of hydrogen is released in the first 5 seconds? ...
AVOGADRO EXAMS 1991 - 2002 PRACTICE BOOKLET
... 16. An element occurring in nature as a metal(such as copper or gold) is likely to (a) react readily with oxygen to from a protective oxide coating (b) be at the high end of the activity series of metals (c) cause strong acids to release hydrogen gas (d) undergo oxidation only with difficulty (e) lo ...
... 16. An element occurring in nature as a metal(such as copper or gold) is likely to (a) react readily with oxygen to from a protective oxide coating (b) be at the high end of the activity series of metals (c) cause strong acids to release hydrogen gas (d) undergo oxidation only with difficulty (e) lo ...
Type - Enrico Fermi High
... (Note these are structural formulas which partially show the Lewis structures of the molecules) HO-CH2-OH b/c it has 2 sites for H-bonding, vs only 1 for H3C-OH. ...
... (Note these are structural formulas which partially show the Lewis structures of the molecules) HO-CH2-OH b/c it has 2 sites for H-bonding, vs only 1 for H3C-OH. ...
SQA CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... All substances are made up of particles called atoms, ions or molecules, and these particles are constantly moving. The degree of movement depends upon the state of the substance. This is known as the "kinetic model" of matter. In any sample of solution, liquid or gas there is a range of kinetic ene ...
... All substances are made up of particles called atoms, ions or molecules, and these particles are constantly moving. The degree of movement depends upon the state of the substance. This is known as the "kinetic model" of matter. In any sample of solution, liquid or gas there is a range of kinetic ene ...
Aluminum Borohydride Complex with
... in the development of these compounds is trying to formulate the borohydrides with groups of Hδ+ enriched molecules such as NH 3 to form novel nitrogen and boron-combined materials.18 Both hydridic and protic hydrogen atoms are contained in these compounds; as the result, they are proposed to releas ...
... in the development of these compounds is trying to formulate the borohydrides with groups of Hδ+ enriched molecules such as NH 3 to form novel nitrogen and boron-combined materials.18 Both hydridic and protic hydrogen atoms are contained in these compounds; as the result, they are proposed to releas ...
Teaching with SCIGRESS - Photochemical Dynamics Group
... provides much of the information upon which these predictions are made. From lectures you have learned to use several models to describe molecules. For example Lewis dot structures delineate connectivity and bonding relationships between atoms of a molecule or ion and can predict bond energies and b ...
... provides much of the information upon which these predictions are made. From lectures you have learned to use several models to describe molecules. For example Lewis dot structures delineate connectivity and bonding relationships between atoms of a molecule or ion and can predict bond energies and b ...
Stoichiometery
... Real Chemistry is all about doing chemical reactions. Chemistry is about making or breaking bonds in order to rearrange atoms and make new compounds. ...
... Real Chemistry is all about doing chemical reactions. Chemistry is about making or breaking bonds in order to rearrange atoms and make new compounds. ...
Surface Structure/Property Relationship for (001) Surface of Magnetite
... the oldest magnet, it has been studied for decades, but it is still attracting attention owing to its relevance to technological development. It is used as the core material of hardware, such as electromagnetic coils, microwave resonant circuits, computer memory cores and high density magnetic recor ...
... the oldest magnet, it has been studied for decades, but it is still attracting attention owing to its relevance to technological development. It is used as the core material of hardware, such as electromagnetic coils, microwave resonant circuits, computer memory cores and high density magnetic recor ...
chm 205 - National Open University of Nigeria
... alternate atom in it is Si in place of carbon. In diamond, the strong covalent bonds formed within the giant macromolecule result in a structure which is without any mobile electrons and thus it behaves as an insulator. The rigid, three dimensional linkages make diamond one of the hardest substances ...
... alternate atom in it is Si in place of carbon. In diamond, the strong covalent bonds formed within the giant macromolecule result in a structure which is without any mobile electrons and thus it behaves as an insulator. The rigid, three dimensional linkages make diamond one of the hardest substances ...
Chapter 8 "Ionic versus Covalent Bonding"
... compounds are usually rigid, brittle, crystalline substances with flat surfaces that intersect at characteristic angles. They are not easily deformed, and they melt at relatively high temperatures. NaCl, for example, melts at 801°C. These properties result from the regular arrangement of the ions in ...
... compounds are usually rigid, brittle, crystalline substances with flat surfaces that intersect at characteristic angles. They are not easily deformed, and they melt at relatively high temperatures. NaCl, for example, melts at 801°C. These properties result from the regular arrangement of the ions in ...
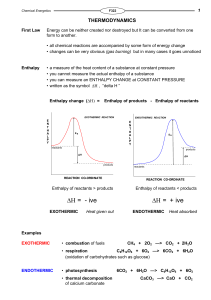
Enthalpy change
... The enthalpy change when ONE MOLE of a substance undergoes complete combustion under standard conditions. All reactants and products are in their ...
... The enthalpy change when ONE MOLE of a substance undergoes complete combustion under standard conditions. All reactants and products are in their ...
File
... Answers to the chapter summary worksheets (see the textbook CD-ROM) are given at the end of each chapter of this Teacher Guide. The answers to the practice unit tests are model answers that include all that examiners look for when awarding marks, together with some extra explanation. Each marking po ...
... Answers to the chapter summary worksheets (see the textbook CD-ROM) are given at the end of each chapter of this Teacher Guide. The answers to the practice unit tests are model answers that include all that examiners look for when awarding marks, together with some extra explanation. Each marking po ...
Class-XII, Summer assignment
... 2. PH3 has lower boiling point than NH3. Why? Ans: Unlike NH3, PH3 molecules are not associated through hydrogen bonding in liquid state. That is why the boiling point of PH3 is lower than NH3. 3. Why are pentahalides more covalent than trihalides ? Ans: Higher the positive oxidation state of centra ...
... 2. PH3 has lower boiling point than NH3. Why? Ans: Unlike NH3, PH3 molecules are not associated through hydrogen bonding in liquid state. That is why the boiling point of PH3 is lower than NH3. 3. Why are pentahalides more covalent than trihalides ? Ans: Higher the positive oxidation state of centra ...
chemical bonding i: basic concepts
... The Lewis theory of bonding describes a covalent bond as the sharing of a pair of electrons, but this does not necessarily mean that each atom contributes an electron to the bond. A covalent bond in which a single atom contributes both of the electrons to a shared pair is called a coordinate covalen ...
... The Lewis theory of bonding describes a covalent bond as the sharing of a pair of electrons, but this does not necessarily mean that each atom contributes an electron to the bond. A covalent bond in which a single atom contributes both of the electrons to a shared pair is called a coordinate covalen ...
Hydrogen bond
A hydrogen bond is the electrostatic attraction between polar molecules that occurs when a hydrogen (H) atom bound to a highly electronegative atom such as nitrogen (N), oxygen (O) or fluorine (F) experiences attraction to some other nearby highly electronegative atom.These hydrogen-bond attractions can occur between molecules (intermolecular) or within different parts of a single molecule (intramolecular). The hydrogen bond (5 to 30 kJ/mole) is stronger than a van der Waals interaction, but weaker than covalent or ionic bonds. This type of bond can occur in inorganic molecules such as water and in organic molecules like DNA and proteins.Intermolecular hydrogen bonding is responsible for the high boiling point of water (100 °C) compared to the other group 16 hydrides that have no hydrogen bonds. Intramolecular hydrogen bonding is partly responsible for the secondary and tertiary structures of proteins and nucleic acids. It also plays an important role in the structure of polymers, both synthetic and natural.In 2011, an IUPAC Task Group recommended a modern evidence-based definition of hydrogen bonding, which was published in the IUPAC journal Pure and Applied Chemistry. This definition specifies that The hydrogen bond is an attractive interaction between a hydrogen atom from a molecule or a molecular fragment X–H in which X is more electronegative than H, and an atom or a group of atoms in the same or a different molecule, in which there is evidence of bond formation. An accompanying detailed technical report provides the rationale behind the new definition.