Avogadro`s Law is relation between
... 3- Given the same number of moles of two gases at STP conditions, how do the volumes of two gases compare? How do the masses of the two gas samples compare? 4- How many moles of helium are contained in each volume at STP: (a) 5.0 L; (b) 11.2 L; (c) 50.0 mL? 5- How many moles of argon are contained i ...
... 3- Given the same number of moles of two gases at STP conditions, how do the volumes of two gases compare? How do the masses of the two gas samples compare? 4- How many moles of helium are contained in each volume at STP: (a) 5.0 L; (b) 11.2 L; (c) 50.0 mL? 5- How many moles of argon are contained i ...
Chapter 10 - Chemical Quantities
... 21. Find the empirical formula of a compound, given that the compound is found to be 47.9% zinc (Zn) and 52.1% chlorine (Cl) by mass. (Zn = 65.4 g/mol; Cl = 35.5 g/mol) Ans: ZnCl2 22. Find the empirical formula of a compound, given that a 48.5-g sample of the compound is found to contain 1.75 g of c ...
... 21. Find the empirical formula of a compound, given that the compound is found to be 47.9% zinc (Zn) and 52.1% chlorine (Cl) by mass. (Zn = 65.4 g/mol; Cl = 35.5 g/mol) Ans: ZnCl2 22. Find the empirical formula of a compound, given that a 48.5-g sample of the compound is found to contain 1.75 g of c ...
Metals
... exist as an array of ions or atoms bound to each other but with no recognisable molecules. The formula NaCl instead tells us that throughout a sample of NaCl sodium and chlorine atoms are present in the ratio 1:1. Because ionic compounds do not contain molecules the sum of the relative atomic masses ...
... exist as an array of ions or atoms bound to each other but with no recognisable molecules. The formula NaCl instead tells us that throughout a sample of NaCl sodium and chlorine atoms are present in the ratio 1:1. Because ionic compounds do not contain molecules the sum of the relative atomic masses ...
3.1 Atomic Mass - Pace University Webspace
... • An Avogadro's number of standard soft drink cans would cover the surface of the earth to a depth of over 200 miles. • If you had Avogadro's number of unpopped popcorn kernels, and spread them across the United States of America, the country would be covered in popcorn to a depth of over 9 miles. • ...
... • An Avogadro's number of standard soft drink cans would cover the surface of the earth to a depth of over 200 miles. • If you had Avogadro's number of unpopped popcorn kernels, and spread them across the United States of America, the country would be covered in popcorn to a depth of over 9 miles. • ...
Wizard Test Maker
... 4891 The number of neutrons in the nucleus of an atom can be determined by (1) adding the atomic number to the mass number (2) subtracting the atomic number from the mass number (3) adding the mass number to the atomic mass (4) subtracting the mass number from the atomic number 4805 What is the tota ...
... 4891 The number of neutrons in the nucleus of an atom can be determined by (1) adding the atomic number to the mass number (2) subtracting the atomic number from the mass number (3) adding the mass number to the atomic mass (4) subtracting the mass number from the atomic number 4805 What is the tota ...
Chapter 2_Application Problems
... have one oxygen atom and two hydrogen atoms – correct; according to Dalton, atoms combine together in compounds in small whole-number ratios, so that you could describe a compound by describing the number of atoms of each element in a molecule. He used this idea to explain why compounds obey the Law ...
... have one oxygen atom and two hydrogen atoms – correct; according to Dalton, atoms combine together in compounds in small whole-number ratios, so that you could describe a compound by describing the number of atoms of each element in a molecule. He used this idea to explain why compounds obey the Law ...
GAS PRACTICE A sample of an ideal gas is cooled from 50.0 °C to
... Questions 6-7 relate to the graph shown on the right. The graph shows the temperature of a pure substance as it is heated at a constant rate in an open vessel at 1.0 atm pressure. The substance changes from the solid to the liquid to the gas phase. 6. The substance is at its normal freezing point at ...
... Questions 6-7 relate to the graph shown on the right. The graph shows the temperature of a pure substance as it is heated at a constant rate in an open vessel at 1.0 atm pressure. The substance changes from the solid to the liquid to the gas phase. 6. The substance is at its normal freezing point at ...
Example - Schoolwires.net
... number of molecules or formula units in the REACTION. They are the only things changed when balancing a reaction. ...
... number of molecules or formula units in the REACTION. They are the only things changed when balancing a reaction. ...
Chapter 6.2 Notes
... - because they do not form individual molecules, to write the chemical formulas use the smallest ratio of one ion to another, called the formula unit NaCl 1:1 Na2O 2:1 AlBr3 1:3 - smallest ratio means they will not be divisible by each other and get a whole number - will never have an ionic compound ...
... - because they do not form individual molecules, to write the chemical formulas use the smallest ratio of one ion to another, called the formula unit NaCl 1:1 Na2O 2:1 AlBr3 1:3 - smallest ratio means they will not be divisible by each other and get a whole number - will never have an ionic compound ...
Chem 171 Review - Exam 1
... uncertainty in measurements – indicated by the number of recorded significant figures significant figures: all certain digits plus the 1st uncertain digit given a number, determine how many significant figures it has understand when zeros are and are not significant determination of the number of si ...
... uncertainty in measurements – indicated by the number of recorded significant figures significant figures: all certain digits plus the 1st uncertain digit given a number, determine how many significant figures it has understand when zeros are and are not significant determination of the number of si ...
Topic 1: Quantitative Chemistry
... 1. Determine mass of elements involved (if percent composition data is given, assume a 100g sample) 2. Convert mass to moles ( moles = mass/molar mass) 3. Divide all molar amounts by the smallest molar amount; the numbers found are the amount of each atom in the empirical formula If necessary force ...
... 1. Determine mass of elements involved (if percent composition data is given, assume a 100g sample) 2. Convert mass to moles ( moles = mass/molar mass) 3. Divide all molar amounts by the smallest molar amount; the numbers found are the amount of each atom in the empirical formula If necessary force ...
Water: The Universal Solvent
... Balance the element being oxidized and reduced. Balance the elements (that is being reduced or oxidized) oxidation # by adding electrons. Oxidation adds to the right, reduction adds to the left. Balance the oxygens by adding water molecules. Balance the hydrogens by adding H+ ions. If the electrons ...
... Balance the element being oxidized and reduced. Balance the elements (that is being reduced or oxidized) oxidation # by adding electrons. Oxidation adds to the right, reduction adds to the left. Balance the oxygens by adding water molecules. Balance the hydrogens by adding H+ ions. If the electrons ...
AP Chem Test 5 preview Gases
... 2005B.6. Consider two containers of volume 1.0 L at 298 K, as shown below. One container holds 0.10 mol N2(g) and the other holds 0.10 mol H2(g) The average kinetic energy of the N2(g) molecules is 6.2 x 10-21 J. Assume that the N2(g) and the H2(g) exhibit ideal behavior. a) Is the pressure in the c ...
... 2005B.6. Consider two containers of volume 1.0 L at 298 K, as shown below. One container holds 0.10 mol N2(g) and the other holds 0.10 mol H2(g) The average kinetic energy of the N2(g) molecules is 6.2 x 10-21 J. Assume that the N2(g) and the H2(g) exhibit ideal behavior. a) Is the pressure in the c ...
Periodicity - Teach-n-Learn-Chem
... Describe how the modern periodic table is organized. State the periodic law. Explain why elements in the same family of the periodic table have similar properties. Describe the characteristics of the alkali metals, alkaline-earth metals, transition metals, actinides, lanthanides, halogens, and ...
... Describe how the modern periodic table is organized. State the periodic law. Explain why elements in the same family of the periodic table have similar properties. Describe the characteristics of the alkali metals, alkaline-earth metals, transition metals, actinides, lanthanides, halogens, and ...
CHEMISTRY 101 Name Mock Final Exam Spring 2014 Signature Dr
... equation, then reactant B must be limiting. d) If the coefficient for A is greater than the coefficient of B in the balanced equation, then reactant A must be limiting. e) None of the above choices (a-d) must be true. ...
... equation, then reactant B must be limiting. d) If the coefficient for A is greater than the coefficient of B in the balanced equation, then reactant A must be limiting. e) None of the above choices (a-d) must be true. ...
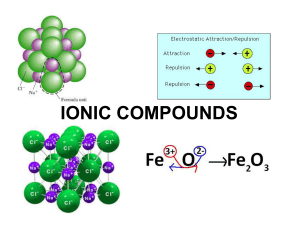
Section 8.3 Names and Formulas of Ionic Compounds Formula Unit
... • The chemical formula can be written following the same rules as monatomic ions ...
... • The chemical formula can be written following the same rules as monatomic ions ...
Determination of the Empirical Formula of an
... While a few metals, such as gold and silver, can be found in their elemental forms in nature, the majority of metals are naturally found in mineral ores in the form of compounds.1 These metals must be extracted from their ores through a process known as smelting.2 The first metals to be smelted, mor ...
... While a few metals, such as gold and silver, can be found in their elemental forms in nature, the majority of metals are naturally found in mineral ores in the form of compounds.1 These metals must be extracted from their ores through a process known as smelting.2 The first metals to be smelted, mor ...
Name: Per: Date: Unit 1. Materials: Formulating Matter B. Periodic
... smallest whole numbers! [For example, Ca2O2 simplifies to become CaO] Ions a. Al3+ + O2-– ...
... smallest whole numbers! [For example, Ca2O2 simplifies to become CaO] Ions a. Al3+ + O2-– ...
Chemistry Final Exam Practice Test
... 70. When an electron moves from a lower to a higher energy level, the electron _____. a) always doubles its energy b) absorbs a continuously variable amount of energy c) absorbs a quantum of energy d) moves closer to the nucleus ...
... 70. When an electron moves from a lower to a higher energy level, the electron _____. a) always doubles its energy b) absorbs a continuously variable amount of energy c) absorbs a quantum of energy d) moves closer to the nucleus ...
Final Exam Practice Problems: R = 0.0821 Latm/molK NA = 6.022
... A) Li+ (aq) + SO42-(aq) + Cu+(aq) + NO3-(aq) → CuS(s) + Li+(aq) + NO3-(aq) B) Li+ (aq) + S-(aq) + Cu+(aq) + NO3-(aq) → CuS(s) + LiNO3(aq) C) 2 Li+(aq) + S2-(aq) + Cu2+(aq) + 2 NO3-(aq) → Cu2+(aq) + S2-(aq) + 2 LiNO3(s) D) 2 Li+(aq) + S2-(aq) + Cu2+(aq) + 2 NO3-(aq) → CuS(s) + 2 Li+(aq) + 2 NO3-(aq) ...
... A) Li+ (aq) + SO42-(aq) + Cu+(aq) + NO3-(aq) → CuS(s) + Li+(aq) + NO3-(aq) B) Li+ (aq) + S-(aq) + Cu+(aq) + NO3-(aq) → CuS(s) + LiNO3(aq) C) 2 Li+(aq) + S2-(aq) + Cu2+(aq) + 2 NO3-(aq) → Cu2+(aq) + S2-(aq) + 2 LiNO3(s) D) 2 Li+(aq) + S2-(aq) + Cu2+(aq) + 2 NO3-(aq) → CuS(s) + 2 Li+(aq) + 2 NO3-(aq) ...
Multiple Choice Practice. A) P B) S C) Cl D) Li E) 1 F 1. Has the
... A) SiCl4 B) BrCl C) PCl3 D) Cl2O E) CaCl2 32. The simplest formula for an oxide of nitrogen that is 36.8 percent nitrogen by mass is A) N2O B) NO C) NO2 D) N2O3 E) N2O5 33. Compounds of which of the following functional groups are noted for their pleasant odors and flavors? A) alcohol B) amine C) or ...
... A) SiCl4 B) BrCl C) PCl3 D) Cl2O E) CaCl2 32. The simplest formula for an oxide of nitrogen that is 36.8 percent nitrogen by mass is A) N2O B) NO C) NO2 D) N2O3 E) N2O5 33. Compounds of which of the following functional groups are noted for their pleasant odors and flavors? A) alcohol B) amine C) or ...
Compulsory textbook Recommended textbooks Topics of the first
... 2. The precipitating agent reacts selectively or, at least, specifically with the analyte ...
... 2. The precipitating agent reacts selectively or, at least, specifically with the analyte ...
Document
... SOLUTION: (a) ClO4- is perchlorate; Fe must have a 2+ charge since there are 2 ClO4- ions. This is iron(II) perchlorate. (b) The anion sulfite is SO32-; therefore you need 2 Na+ for each sulfite. The formula is Na2SO3. (c) The ionic compound is barium hydroxide. When water is included in the formula ...
... SOLUTION: (a) ClO4- is perchlorate; Fe must have a 2+ charge since there are 2 ClO4- ions. This is iron(II) perchlorate. (b) The anion sulfite is SO32-; therefore you need 2 Na+ for each sulfite. The formula is Na2SO3. (c) The ionic compound is barium hydroxide. When water is included in the formula ...
Inductively coupled plasma mass spectrometry

Inductively coupled plasma mass spectrometry (ICP-MS) is a type of mass spectrometry which is capable of detecting metals and several non-metals at concentrations as low as one part in 1015 (part per quadrillion, ppq) on non-interfered low-background isotopes. This is achieved by ionizing the sample with inductively coupled plasma and then using a mass spectrometer to separate and quantify those ions.Compared to atomic absorption techniques, ICP-MS has greater speed, precision, and sensitivity. However, compared with other types of mass spectrometry, such as Thermal ionization mass spectrometry (TIMS) and Glow Discharge Mass Spectrometry (GD-MS), ICP-MS introduces a lot of interfering species: argon from the plasma, component gasses of air that leak through the cone orifices, and contamination from glassware and the cones.The variety of applications exceeds that of inductively coupled plasma atomic emission spectroscopy and includes isotopic speciation. Due to possible applications in nuclear technologies, ICP-MS hardware is a subject for special exporting regulations.