AP Chemistry 2015-‐‑2016 Name: Chapter 5: Thermodynamics
... 5. If 300. J of heat energy were used to heat up a 5.00 gram sample of copper metal and a 5.00 gram sample of water both starting at 10.0°C, calculate the final temperature of each sample? ...
... 5. If 300. J of heat energy were used to heat up a 5.00 gram sample of copper metal and a 5.00 gram sample of water both starting at 10.0°C, calculate the final temperature of each sample? ...
Unit 7 Homework and Lab Packet
... 3. Some household drain cleaners consist of a mixture of granules of sodium hydroxide and powdered aluminum. When dissolved in water, the sodium hydroxide reacts with the aluminum to produce hydrogen gas: 2 Al + 2 NaOH + 6 H2O 2 NaAl(OH)4 + 3 H2 The sodium hydroxide helps dissolve grease, and the ...
... 3. Some household drain cleaners consist of a mixture of granules of sodium hydroxide and powdered aluminum. When dissolved in water, the sodium hydroxide reacts with the aluminum to produce hydrogen gas: 2 Al + 2 NaOH + 6 H2O 2 NaAl(OH)4 + 3 H2 The sodium hydroxide helps dissolve grease, and the ...
Magic of Chemical Reactions 2. - mt
... retains its luster for years. Since gold does not corrode, gold ornaments look new after years. break. 3. This is achieved by bringing reactants in contact with each other and then by supplying energy in the form of either heat, light or electricity. 4. This results in breaking of bonds in the react ...
... retains its luster for years. Since gold does not corrode, gold ornaments look new after years. break. 3. This is achieved by bringing reactants in contact with each other and then by supplying energy in the form of either heat, light or electricity. 4. This results in breaking of bonds in the react ...
File
... Copyright © 1984 by Educational Testing Service, Princeton, NJ 08541. All rights reserved. Reproductions of these examination questions by classroom teachers is permitted for face-to-face teaching purposes only. ...
... Copyright © 1984 by Educational Testing Service, Princeton, NJ 08541. All rights reserved. Reproductions of these examination questions by classroom teachers is permitted for face-to-face teaching purposes only. ...
percent composition and formulas
... COMPOUND CONTAINING (A) 2.1 Percent H; 65.3 Percent O; 32.6 percent S ...
... COMPOUND CONTAINING (A) 2.1 Percent H; 65.3 Percent O; 32.6 percent S ...
Ch 8 Lecture Notes
... Sample Exercise 8.7 – Prediction the Mass of a Precipitate Barium sulfate is used to enhance Xray imaging of the upper and lower GI tract. In upper GI imaging, patients drink a suspension of solid barium sulfate in H 2O. The compound is not toxic because of its limited solubility. To make pure bari ...
... Sample Exercise 8.7 – Prediction the Mass of a Precipitate Barium sulfate is used to enhance Xray imaging of the upper and lower GI tract. In upper GI imaging, patients drink a suspension of solid barium sulfate in H 2O. The compound is not toxic because of its limited solubility. To make pure bari ...
Oxidation numbers
... The word species is used in chemistry to indicate either a compound, a molecule, an ion, an atom or an element. We will use the reaction between magnesium and chlorine as an example. The chemical equation for this reaction is: As a reactant, magnesium has an oxidation number of zero, but as part of ...
... The word species is used in chemistry to indicate either a compound, a molecule, an ion, an atom or an element. We will use the reaction between magnesium and chlorine as an example. The chemical equation for this reaction is: As a reactant, magnesium has an oxidation number of zero, but as part of ...
Worksheet to accompany demos on exchange reactions
... (any atoms in an element have oxidation numbers of zero because elements are made of neutral, essentially identical atoms). In water, O is assigned -2 and H +1 (this adds up to zero overall). In carbon dioxide, O is assigned a value of -2, so C ends up having an oxidation number of +4. Thus, we can ...
... (any atoms in an element have oxidation numbers of zero because elements are made of neutral, essentially identical atoms). In water, O is assigned -2 and H +1 (this adds up to zero overall). In carbon dioxide, O is assigned a value of -2, so C ends up having an oxidation number of +4. Thus, we can ...
Chemical reaction model:
... Secondary Nitrate Peak Height (1630 cm 1 ) H .I . Methylene Peak Height (1370 cm 1 ) ...
... Secondary Nitrate Peak Height (1630 cm 1 ) H .I . Methylene Peak Height (1370 cm 1 ) ...
examination review
... Strong bases are bases that produce a high concentration of OH-(aq) ions in aqueous solution. As with strong acids, there are also relatively few common strong bases. Soluble hydroxides and soluble carbonates will produce strong bases. Since most hydroxides have low solubilities, very few hydroxides ...
... Strong bases are bases that produce a high concentration of OH-(aq) ions in aqueous solution. As with strong acids, there are also relatively few common strong bases. Soluble hydroxides and soluble carbonates will produce strong bases. Since most hydroxides have low solubilities, very few hydroxides ...
Bis2A 06.Appendix A review of Red/Ox reactions
... Redox reactions that take place in aqueous media often involve water, hydronium ions, and hydroxide ions as reactants or products. Although these species are not oxidized or reduced, they do participate in chemical change in other ways (e.g., by providing the elements required to form oxyanions). Eq ...
... Redox reactions that take place in aqueous media often involve water, hydronium ions, and hydroxide ions as reactants or products. Although these species are not oxidized or reduced, they do participate in chemical change in other ways (e.g., by providing the elements required to form oxyanions). Eq ...
reducing total package oxygen
... To lower TPO, beer bottling operators have developed various methods for displacing the air or atmospheric oxygen present in the container. Early systems included bottle knockers and ultrasonic vibrations to induce foaming. Newer systems have experimented with the use of CO2 or liquid nitrogen (Oliv ...
... To lower TPO, beer bottling operators have developed various methods for displacing the air or atmospheric oxygen present in the container. Early systems included bottle knockers and ultrasonic vibrations to induce foaming. Newer systems have experimented with the use of CO2 or liquid nitrogen (Oliv ...
Reactions Balancing Chemical Equations uses Law of conservation
... Sum of oxidation numbers is equal to overall charge of molecule or ion: • For a neutral compound the sum of oxidation numbers equals zero. • For a polyatomic ion, the sum of the oxidation numbers is equal to the charge on the ion. Shared electrons are assigned to the more electronegative atom of t ...
... Sum of oxidation numbers is equal to overall charge of molecule or ion: • For a neutral compound the sum of oxidation numbers equals zero. • For a polyatomic ion, the sum of the oxidation numbers is equal to the charge on the ion. Shared electrons are assigned to the more electronegative atom of t ...
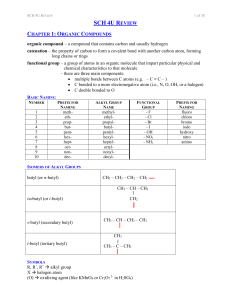
SCH 4U REVIEW Notes
... a compound with a structure based on benzene (a ring of six carbons) consider the benzene ring to be the parent molecule alkyl groups are named to give the lowest combination of numbers, with no particular starting carbon (as it is a ring) when it is easier to consider the benzene ring as an alk ...
... a compound with a structure based on benzene (a ring of six carbons) consider the benzene ring to be the parent molecule alkyl groups are named to give the lowest combination of numbers, with no particular starting carbon (as it is a ring) when it is easier to consider the benzene ring as an alk ...
The p-Block Elements The p-Block Elements
... Ammonia is a colourless gas with a pungent odour. Its freezing and boiling points are 198.4 and 239.7 K respectively. In the solid and liquid states, it is associated through hydrogen bonds as in the case of water and that accounts for its higher melting and boiling points than expected on the basis ...
... Ammonia is a colourless gas with a pungent odour. Its freezing and boiling points are 198.4 and 239.7 K respectively. In the solid and liquid states, it is associated through hydrogen bonds as in the case of water and that accounts for its higher melting and boiling points than expected on the basis ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.