Chapter 1 questions
... Q21. Hydrogen peroxide decomposes over time to produce oxygen gas and water. What volume of O2 (g) at S.T.P. will be produced by the complete decomposition of 200ml of 6.0 M H2O2 solution. Q22. An oxygen cylinder of internal volume 6.65L contains oxygen gas under a pressure of 16.7 MPa at a temperat ...
... Q21. Hydrogen peroxide decomposes over time to produce oxygen gas and water. What volume of O2 (g) at S.T.P. will be produced by the complete decomposition of 200ml of 6.0 M H2O2 solution. Q22. An oxygen cylinder of internal volume 6.65L contains oxygen gas under a pressure of 16.7 MPa at a temperat ...
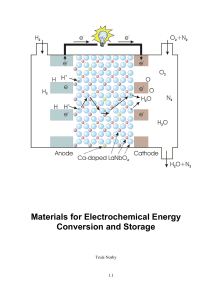
Materials for Electrochemical Energy Conversion and Storage
... Anodes ........................................................................................................... 3.61 Cathodes ......................................................................................................... 3.62 ...
... Anodes ........................................................................................................... 3.61 Cathodes ......................................................................................................... 3.62 ...
File
... For acidic solutions, can have H+(aq), H2O(l), not OH(aq), never e(aq) or O2(aq); - for basic solutions can have OH(aq), H2O(l), not H+(aq) never e(aq) or O2(aq); so must correct for H+ from step (4). – For reactions in basic media add appropriate number of OH to each side of equation so that ...
... For acidic solutions, can have H+(aq), H2O(l), not OH(aq), never e(aq) or O2(aq); - for basic solutions can have OH(aq), H2O(l), not H+(aq) never e(aq) or O2(aq); so must correct for H+ from step (4). – For reactions in basic media add appropriate number of OH to each side of equation so that ...
Monte Carlo Simulation of Water Radiolysis for
... incident particle and its products, covering all ranges of energy transfers in individual collisions. For fast ions, the majority of energy is transferred in ionizing collisions, resulting in energetic free electrons and the potential energy of residual ions. Excitation cross sections and elastic sc ...
... incident particle and its products, covering all ranges of energy transfers in individual collisions. For fast ions, the majority of energy is transferred in ionizing collisions, resulting in energetic free electrons and the potential energy of residual ions. Excitation cross sections and elastic sc ...
Chapter12
... b. number of molecules - the balanced equation shows that 1 molecule of nitrogen reacts with 3 molecules of hydrogen in order to form 2 molecules of ammonia. The ratio of molecules of N2:H2:NH3 is always 1:3:2. This means that if you could get 10 molecules of nitrogen to react with 30 molecules of h ...
... b. number of molecules - the balanced equation shows that 1 molecule of nitrogen reacts with 3 molecules of hydrogen in order to form 2 molecules of ammonia. The ratio of molecules of N2:H2:NH3 is always 1:3:2. This means that if you could get 10 molecules of nitrogen to react with 30 molecules of h ...
LESSON 23: Exploding Bags
... properties. We use our senses or take measurements to identify physical properties. Some examples of physical properties are color, shape, boiling point, melting point, and density. Chemical properties can be identified by observing how a chemical reacts with other substances. Some examples of chemi ...
... properties. We use our senses or take measurements to identify physical properties. Some examples of physical properties are color, shape, boiling point, melting point, and density. Chemical properties can be identified by observing how a chemical reacts with other substances. Some examples of chemi ...
Effect of Potassium on Sol-Gel Cerium and Lanthanum Oxide
... Therefore, the catalysts used in the catalytic combustion of soot must be capable of presenting catalyst activity at low temperatures [10]. An important number of catalytic formulations have been developed in this field, including oxides [11]-[14], perovskites [9] [15]-[17], spinels [18] and metals ...
... Therefore, the catalysts used in the catalytic combustion of soot must be capable of presenting catalyst activity at low temperatures [10]. An important number of catalytic formulations have been developed in this field, including oxides [11]-[14], perovskites [9] [15]-[17], spinels [18] and metals ...
Answers to examination questions
... CH2CH3COOH since it contains hydrogen bonded directly to oxygen. The −OH group is able to engage in hydrogen bonding. In the other two compounds the hydrogen is bonded to carbon. Q8 C Both ethane and neon are non-polar. Hence, the predominant intermolecular and inter-atomic forces are van der Wa ...
... CH2CH3COOH since it contains hydrogen bonded directly to oxygen. The −OH group is able to engage in hydrogen bonding. In the other two compounds the hydrogen is bonded to carbon. Q8 C Both ethane and neon are non-polar. Hence, the predominant intermolecular and inter-atomic forces are van der Wa ...
Chemistry (English) Grade 11 and 12
... hold the molecules of the species together; that is, the easier it is to break the bonds, the lower the boiling point will be. The more carbon atoms an alkane molecule has the more places are available for induced dipole – induced dipole “bonds” to form, so that more energy is needed to break the “b ...
... hold the molecules of the species together; that is, the easier it is to break the bonds, the lower the boiling point will be. The more carbon atoms an alkane molecule has the more places are available for induced dipole – induced dipole “bonds” to form, so that more energy is needed to break the “b ...
Comparison of 2008 to 2000 SCH3U_ud
... potentially harmful chemical substance (e.g., fertilizer, pesticide, a household cleaning product, materials used in electronics and batteries) and how that substance affects the environment, and propose ways to lessen the harmfulness of the substance (e.g., by reducing the amount used, by modifying ...
... potentially harmful chemical substance (e.g., fertilizer, pesticide, a household cleaning product, materials used in electronics and batteries) and how that substance affects the environment, and propose ways to lessen the harmfulness of the substance (e.g., by reducing the amount used, by modifying ...
Learning Outcomes Leaving Certificate Chemistry
... define oxidation and reduction in terms of electron transfer use simple examples , e.g. Na with Cl2, Mg with O2, Zn with Cu2+ to describe oxidation and reduction in terms of electron transfer apply knowledge of oxidation and reduction to explain the rusting of iron define oxidising agent and reducin ...
... define oxidation and reduction in terms of electron transfer use simple examples , e.g. Na with Cl2, Mg with O2, Zn with Cu2+ to describe oxidation and reduction in terms of electron transfer apply knowledge of oxidation and reduction to explain the rusting of iron define oxidising agent and reducin ...
chemical reaction equation - parmod cobra insititution.
... Effects of oxidation reaction in everyday life:- Oxidation has damaging effect on metal as well as on food. The damaging effect of oxidation on metal is studied as corrosion and that on food is studied as rancidity. The two common effect of oxidation reaction are as (i) Corrosion of metal (ii) Ran ...
... Effects of oxidation reaction in everyday life:- Oxidation has damaging effect on metal as well as on food. The damaging effect of oxidation on metal is studied as corrosion and that on food is studied as rancidity. The two common effect of oxidation reaction are as (i) Corrosion of metal (ii) Ran ...
Chemical change is a process that involves recombining atoms and
... A lead-acid storage battery is an electrochemical device that produces voltage and delivers electrical current. The battery is the primary "source" of electrical energy used in vehicles today. It's important to remember that a battery does not store electricity, but rather it stores a series of chem ...
... A lead-acid storage battery is an electrochemical device that produces voltage and delivers electrical current. The battery is the primary "source" of electrical energy used in vehicles today. It's important to remember that a battery does not store electricity, but rather it stores a series of chem ...
____ 1. The energy required to convert a ground
... 45. The graph above shows the results of a study of the reaction of X with a large excess of Y to yield Z. The concentrations of X and Y were measured over a period of time. According to the results, which of the following can be concluded about the rate law for the reaction under the conditions stu ...
... 45. The graph above shows the results of a study of the reaction of X with a large excess of Y to yield Z. The concentrations of X and Y were measured over a period of time. According to the results, which of the following can be concluded about the rate law for the reaction under the conditions stu ...
Energy Practice
... 65. How much energy could we get from eating 10.0 g of sucrose? (Assume the sucrose is oxidized in the body cells to produce carbon dioxide gas and liquid water). ...
... 65. How much energy could we get from eating 10.0 g of sucrose? (Assume the sucrose is oxidized in the body cells to produce carbon dioxide gas and liquid water). ...
GROUP 13 ELEMENTS -THE BORON FAMILY -
... melts by increasing room temperature by a little. Gallium is important because it forms gallium arsenide (GaAs), which can convert light directly into electricity. Also due to thermite reaction, aluminum can extract oxygen from water and hydrogen is released. However, as mentioned above, aluminum fo ...
... melts by increasing room temperature by a little. Gallium is important because it forms gallium arsenide (GaAs), which can convert light directly into electricity. Also due to thermite reaction, aluminum can extract oxygen from water and hydrogen is released. However, as mentioned above, aluminum fo ...
chemistry
... Record the number of your choice for each Part A and Part B–1 multiple-choice question on your separate answer sheet. Write your answers to the Part B–2 and Part C questions in your answer booklet. All work should be written in pen, except for graphs and drawings, which should be done in pencil. You ...
... Record the number of your choice for each Part A and Part B–1 multiple-choice question on your separate answer sheet. Write your answers to the Part B–2 and Part C questions in your answer booklet. All work should be written in pen, except for graphs and drawings, which should be done in pencil. You ...
Oxidation-Reduction Reactions
... Many elements simply combine with oxygen to form the oxide of that element. Heating magnesium in air allows it to combine with oxygen to form magnesium oxide. 2 Mg(s) + O2 (g) → 2MgO(s) Many compounds react with oxygen as well, often in very exothermic processes that are generally referred to as com ...
... Many elements simply combine with oxygen to form the oxide of that element. Heating magnesium in air allows it to combine with oxygen to form magnesium oxide. 2 Mg(s) + O2 (g) → 2MgO(s) Many compounds react with oxygen as well, often in very exothermic processes that are generally referred to as com ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.