Unit 8: Reactions - Mark Rosengarten
... Driving Force: The “motivation” of a reaction to occur: In nature, changes that require the least amount of energy will be the ones that happen. After all, when you let go of a bowling ball, it falls down. The motivation is gravity. It would take more energy to make the ball go up than down, so the ...
... Driving Force: The “motivation” of a reaction to occur: In nature, changes that require the least amount of energy will be the ones that happen. After all, when you let go of a bowling ball, it falls down. The motivation is gravity. It would take more energy to make the ball go up than down, so the ...
Chemistry Lab: Data Manual
... 3. Calculate the percent error between the calculated and the measured diameter, assuming the measured diameter is the true value. ...
... 3. Calculate the percent error between the calculated and the measured diameter, assuming the measured diameter is the true value. ...
Unit3_Notes - Lesmahagow High School
... Batch and Continuous Processes In a batch process the chemicals are loaded into the reaction vessel. The reaction is monitored and at the end of the reaction the product is separated and the reaction vessel cleaned out ready for the next batch. In a continuous process the reactants are continuously ...
... Batch and Continuous Processes In a batch process the chemicals are loaded into the reaction vessel. The reaction is monitored and at the end of the reaction the product is separated and the reaction vessel cleaned out ready for the next batch. In a continuous process the reactants are continuously ...
8 Chemical Equations Chapter Outline Chemical Equations
... ___________________________________ ___________________________________ ___________________________________ ___________________________________ ___________________________________ ___________________________________ © 2014 John Wiley & Sons, Inc. All rights reserved. ...
... ___________________________________ ___________________________________ ___________________________________ ___________________________________ ___________________________________ ___________________________________ © 2014 John Wiley & Sons, Inc. All rights reserved. ...
Η - Knockhardy
... making a bond is an exothermic process as it is the opposite of breaking a bond for diatomic gases, the bond enthalpy is twice the enthalpy of atomisation the smaller the bond enthalpy, the weaker the bond and the easier it is to break ...
... making a bond is an exothermic process as it is the opposite of breaking a bond for diatomic gases, the bond enthalpy is twice the enthalpy of atomisation the smaller the bond enthalpy, the weaker the bond and the easier it is to break ...
3 CO 2 (g) + 4 H 2 O(l)
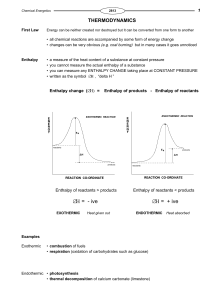
... Every chemical species has what chemists call enthalpy. The enthalpy of a substance is the ability of that species to produce Heat. Almost every chemical reaction produces or absorbs heat and we will learn how to calculate the amount of heat associated with any particular chemical change. When we he ...
... Every chemical species has what chemists call enthalpy. The enthalpy of a substance is the ability of that species to produce Heat. Almost every chemical reaction produces or absorbs heat and we will learn how to calculate the amount of heat associated with any particular chemical change. When we he ...
A Straightforward Route to Enantiopure Pyrrolizidines and
... The direct synthesis of higher alcohols from synthesis gas (syngas, CO+H2) has been extensively reviewed by Klier et al. [14] and by Forzatti et al. [15]. It is widely recognized that higher alcohols together with methanol (MeOH) can be produced from syngas by appropriate modification of MeOHsynthes ...
... The direct synthesis of higher alcohols from synthesis gas (syngas, CO+H2) has been extensively reviewed by Klier et al. [14] and by Forzatti et al. [15]. It is widely recognized that higher alcohols together with methanol (MeOH) can be produced from syngas by appropriate modification of MeOHsynthes ...
3.5 Empirical Formulas - Mayfield City Schools
... 8. Epsom salts, a strong laxative used in veterinary medicine, is a hydrate, which means that a certain number of water molecules are included in the solid structure. The formula for Epsom salts can be written as MgSO4•xH2O, where x indicates the number of moles of H2O per mole of MgSO4. When 5.061 ...
... 8. Epsom salts, a strong laxative used in veterinary medicine, is a hydrate, which means that a certain number of water molecules are included in the solid structure. The formula for Epsom salts can be written as MgSO4•xH2O, where x indicates the number of moles of H2O per mole of MgSO4. When 5.061 ...
some basic concepts of chemistry
... Many of the substances present around you are mixtures. For example, sugar solution in water, air, tea etc., are all mixtures. A mixture contains two or more substances present in it (in any ratio) which are called its components. A mixture may be homogeneous or heterogeneous. In a homogeneous mixtu ...
... Many of the substances present around you are mixtures. For example, sugar solution in water, air, tea etc., are all mixtures. A mixture contains two or more substances present in it (in any ratio) which are called its components. A mixture may be homogeneous or heterogeneous. In a homogeneous mixtu ...
PRACTICE EXAM 1-C
... 4. Do not use red pen! 5. For full credit, show all calculations; this also helps us award partial credit. 6. All numerical answers must include correct units for full credit. ...
... 4. Do not use red pen! 5. For full credit, show all calculations; this also helps us award partial credit. 6. All numerical answers must include correct units for full credit. ...
Emerging electrochemical energy conversion and
... Furthermore, the hydrogen generation by electrolysis is an energy intensive process and most commercial electrolyzers require an electric power input of 6.7–7.3 kWh/Nm3 (∼50–55% efficiency based on HHV) of hydrogen (Badwal et al., 2013), and this increases the cost of hydrogen production and advanta ...
... Furthermore, the hydrogen generation by electrolysis is an energy intensive process and most commercial electrolyzers require an electric power input of 6.7–7.3 kWh/Nm3 (∼50–55% efficiency based on HHV) of hydrogen (Badwal et al., 2013), and this increases the cost of hydrogen production and advanta ...
ХИМИЯ НА АНГЛИЙСКОМ ЯЗЫКЕ
... 3.14. A borane (containing only B and H) contained 88.45 % B. What is its empirical formula? 3.15. When 10.24 g of Cu is heated in an atmosphere of oxygen, 11.52 g of an oxide of copper is produced. What is the empirical formula of the oxide formed? 3.16. A 2.522 g sample of pure caffeine contains ...
... 3.14. A borane (containing only B and H) contained 88.45 % B. What is its empirical formula? 3.15. When 10.24 g of Cu is heated in an atmosphere of oxygen, 11.52 g of an oxide of copper is produced. What is the empirical formula of the oxide formed? 3.16. A 2.522 g sample of pure caffeine contains ...
Document
... Learning objective 3.4 The student is able to relate quantities (measured mass of substances, volumes of solutions, or volumes and pressures of gases) to identify stoichiometric relationships for a reaction, including situations involving limiting reactants and situations in which the reaction has n ...
... Learning objective 3.4 The student is able to relate quantities (measured mass of substances, volumes of solutions, or volumes and pressures of gases) to identify stoichiometric relationships for a reaction, including situations involving limiting reactants and situations in which the reaction has n ...
CHE 1400 Lab Manual - Al Akhawayn University
... a small volume. While we often say that lead is « heavy », what we really mean is that lead has a high density. Density is commonly given in terms of grams per milliliter (g/mL), although other units of mass and volume can be used. 3. Boiling point When a liquid is gradually heated, there is a point ...
... a small volume. While we often say that lead is « heavy », what we really mean is that lead has a high density. Density is commonly given in terms of grams per milliliter (g/mL), although other units of mass and volume can be used. 3. Boiling point When a liquid is gradually heated, there is a point ...
CHAPTER 1
... liquid water takes up a definite amount of space, but the water takes the shape of its container. Liquids have this characteristic because the particles in them are close together but can move past one another. The particles in a liquid move more rapidly than those in a solid. This causes them to ov ...
... liquid water takes up a definite amount of space, but the water takes the shape of its container. Liquids have this characteristic because the particles in them are close together but can move past one another. The particles in a liquid move more rapidly than those in a solid. This causes them to ov ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.