Boundless Study Slides
... • An atom is composed of two regions: the nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom which holds its electrons in orbit around the nucleus. • Protons and neutrons have approximately the same mass, about 1.67 × 10-24 grams, which sc ...
... • An atom is composed of two regions: the nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom which holds its electrons in orbit around the nucleus. • Protons and neutrons have approximately the same mass, about 1.67 × 10-24 grams, which sc ...
Chapter 2 ATOMS AND ELEMENTS
... • Phosphorus is essential to life. It is an important constituent in bones, teeth, and DNA. The element glows in the dark if it is in the air. Its name is based on Greek words meaning “lightbearing”. ...
... • Phosphorus is essential to life. It is an important constituent in bones, teeth, and DNA. The element glows in the dark if it is in the air. Its name is based on Greek words meaning “lightbearing”. ...
Chapter 2 ATOMS AND ELEMENTS
... • Phosphorus is essential to life. It is an important constituent in bones, teeth, and DNA. The element glows in the dark if it is in the air. Its name is based on Greek words meaning “lightbearing”. ...
... • Phosphorus is essential to life. It is an important constituent in bones, teeth, and DNA. The element glows in the dark if it is in the air. Its name is based on Greek words meaning “lightbearing”. ...
Name: Period:______ Table Number:______
... 51. Each element found on the periodic table of elements has a unique single letter (Hydrogen – H), two letter (Helium – He ) or three letter (Unnilquadiam – Unq) abbreviation which is called the CHEMICAL SYMBOL of that element. P. 83, Bill Nye the Science Guy Video 52. JONS BERZELIUS created the un ...
... 51. Each element found on the periodic table of elements has a unique single letter (Hydrogen – H), two letter (Helium – He ) or three letter (Unnilquadiam – Unq) abbreviation which is called the CHEMICAL SYMBOL of that element. P. 83, Bill Nye the Science Guy Video 52. JONS BERZELIUS created the un ...
Atomic Mass
... The atomic number tells you the number of protons in one an atom of an element. It also tells you the number of electrons in a neutral atom of that element. The atomic number gives the identity of an element as well as its location on the periodic table. No two different elements will have the same ...
... The atomic number tells you the number of protons in one an atom of an element. It also tells you the number of electrons in a neutral atom of that element. The atomic number gives the identity of an element as well as its location on the periodic table. No two different elements will have the same ...
Oxidation numbers
... In fact, oxidation never takes place on its own - nor does reduction. When one substance is oxidised in a reaction, another one is reduced. A Redox reaction is one in which both reduction and oxidation take place. To work out which element is oxidised and which is reduced in a reaction, we go throug ...
... In fact, oxidation never takes place on its own - nor does reduction. When one substance is oxidised in a reaction, another one is reduced. A Redox reaction is one in which both reduction and oxidation take place. To work out which element is oxidised and which is reduced in a reaction, we go throug ...
Chapter 4- Elements and the Periodic Table
... the sealed box in the Standards Warm-Up activity. Ideas about the shape and structure of atoms have changed many times. The first people to think about the nature of matter were the ancient Greeks. Around the year 430 B.C., a Greek philosopher named Democritus proposed the idea that matter is formed ...
... the sealed box in the Standards Warm-Up activity. Ideas about the shape and structure of atoms have changed many times. The first people to think about the nature of matter were the ancient Greeks. Around the year 430 B.C., a Greek philosopher named Democritus proposed the idea that matter is formed ...
Atomic Structure Powerpoints - Warren County Public Schools
... • Mass # = # of protons and # of neutrons in an atom. • Atomic Mass = The average mass for an element. It is determined by taking in account all the isotopes that make-up an element. You must know the isotopes relative abundance and mass # to calculate the atomic mass of the element. -Atomic mass = ...
... • Mass # = # of protons and # of neutrons in an atom. • Atomic Mass = The average mass for an element. It is determined by taking in account all the isotopes that make-up an element. You must know the isotopes relative abundance and mass # to calculate the atomic mass of the element. -Atomic mass = ...
Types of Radiation - Kasson
... • For example, gamma rays accompany the alpha-decay reaction of uranium-238. • The 2 in front of the γ symbol indicates that two gamma rays of different frequencies are emitted. • Because gamma rays have no effect on mass number or atomic number, it is customary to omit them from nuclear equations. ...
... • For example, gamma rays accompany the alpha-decay reaction of uranium-238. • The 2 in front of the γ symbol indicates that two gamma rays of different frequencies are emitted. • Because gamma rays have no effect on mass number or atomic number, it is customary to omit them from nuclear equations. ...
UNIT 3 - ATOMS 1 HISTORY OF ATOMIC THEORY NOTES I
... 22. Calculate the average atomic mass of oxygen given the percent abundance of each of its naturallyoccurring isotopes: oxygen-16 is 99.76%, oxygen-17 is 0.037%, and oxygen-18 is 0.204%. 23. What is the mass of 6.00 moles of aluminum? 24. How many moles contain 8.53 x 1023 atoms of platinum? 25. How ...
... 22. Calculate the average atomic mass of oxygen given the percent abundance of each of its naturallyoccurring isotopes: oxygen-16 is 99.76%, oxygen-17 is 0.037%, and oxygen-18 is 0.204%. 23. What is the mass of 6.00 moles of aluminum? 24. How many moles contain 8.53 x 1023 atoms of platinum? 25. How ...
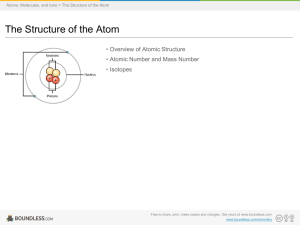
4 Structure of The Atom
... the protons, the neutrons and the electrons. The protons have a positive charge of 1.6 × 10–19 C and mass equal to 1 amu or 1 u (unified mass). The neutrons have no charge and mass equal to 1 amu. The electrons have a negative charge of 1.6 × 10–19 C and mass 1/1837 amu. The protons and neutrons con ...
... the protons, the neutrons and the electrons. The protons have a positive charge of 1.6 × 10–19 C and mass equal to 1 amu or 1 u (unified mass). The neutrons have no charge and mass equal to 1 amu. The electrons have a negative charge of 1.6 × 10–19 C and mass 1/1837 amu. The protons and neutrons con ...
Chapter 2 - HCC Learning Web
... You can determine that the atomic number (Z) of carbon is 6 (from the periodic table). This means that carbon atoms have 6 protons. The mass number (A) for the isotope with 7 neutrons is the sum of the number of protons and the number of neutrons. ...
... You can determine that the atomic number (Z) of carbon is 6 (from the periodic table). This means that carbon atoms have 6 protons. The mass number (A) for the isotope with 7 neutrons is the sum of the number of protons and the number of neutrons. ...
Name
... A(n) __________________________ is a pure substance that can not be broken down into any other substance by some physical or chemical method and from which all more complex forms of matter or substances are made when they are combined together in different ways and in different amounts. P. 9, 70, ...
... A(n) __________________________ is a pure substance that can not be broken down into any other substance by some physical or chemical method and from which all more complex forms of matter or substances are made when they are combined together in different ways and in different amounts. P. 9, 70, ...
Period:______ Table Number
... 79. A(n) _____________________________ is a subatomic particle which has a negative electrical charge. P. 74, 77, VCR: Atoms and Molecules 80. The negative electrical charge of one electron is equal to but opposite the positive electrical charge of one ______________________________. P. 77, VCR: Ato ...
... 79. A(n) _____________________________ is a subatomic particle which has a negative electrical charge. P. 74, 77, VCR: Atoms and Molecules 80. The negative electrical charge of one electron is equal to but opposite the positive electrical charge of one ______________________________. P. 77, VCR: Ato ...
Distinguishing the Atom Reading
... Table 4.2 shows that a fluorine atom has an atomic number of 9 and a mass number of 19. Since the atomic number equals the number of protons, which equals the number of electrons, a fluorine atom has nine protons and nine electrons. The mass number of fluorine is equal to the number of protons plus ...
... Table 4.2 shows that a fluorine atom has an atomic number of 9 and a mass number of 19. Since the atomic number equals the number of protons, which equals the number of electrons, a fluorine atom has nine protons and nine electrons. The mass number of fluorine is equal to the number of protons plus ...
Chap4 Review - armstrongchemistry
... The diagram above shows how alpha, beta, and gamma rays are affected by two electrically charged plates. Based on the paths the rays follow, what are the respective charges of alpha, beta, and gamma rays? ...
... The diagram above shows how alpha, beta, and gamma rays are affected by two electrically charged plates. Based on the paths the rays follow, what are the respective charges of alpha, beta, and gamma rays? ...
EARLY ATOMIC THEORY AND STRUCTURE
... 9. Atomic masses are not whole numbers because: (a) the neutron and proton do not have identical masses and neither is exactly 1 amu. (b) most elements exist in nature as a mixture of isotopes with different atomic masses due to different numbers of neutrons. The atomic mass given in the periodic ta ...
... 9. Atomic masses are not whole numbers because: (a) the neutron and proton do not have identical masses and neither is exactly 1 amu. (b) most elements exist in nature as a mixture of isotopes with different atomic masses due to different numbers of neutrons. The atomic mass given in the periodic ta ...
File
... arrangement is significant. The elements in each vertical column or group have similar properties. The elements in the first period or row all have one shell. The elements in period 2 all have 2 shells and so on. There are a number of major groups with similar properties. They are as follows: Hydrog ...
... arrangement is significant. The elements in each vertical column or group have similar properties. The elements in the first period or row all have one shell. The elements in period 2 all have 2 shells and so on. There are a number of major groups with similar properties. They are as follows: Hydrog ...
sec 3- Counting atoms - Nutley Public Schools
... • Imagine that your semester grade depends 60% on exam scores and 40% on laboratory explorations. • Your exam scores would count more heavily toward your final grade. • In this section, you will learn that the atomic mass of an element is a weighted average of the masses of the naturally occurring i ...
... • Imagine that your semester grade depends 60% on exam scores and 40% on laboratory explorations. • Your exam scores would count more heavily toward your final grade. • In this section, you will learn that the atomic mass of an element is a weighted average of the masses of the naturally occurring i ...
Counting Atoms
... • Imagine that your semester grade depends 60% on exam scores and 40% on laboratory explorations. • Your exam scores would count more heavily toward your final grade. • In this section, you will learn that the atomic mass of an element is a weighted average of the masses of the naturally occurring i ...
... • Imagine that your semester grade depends 60% on exam scores and 40% on laboratory explorations. • Your exam scores would count more heavily toward your final grade. • In this section, you will learn that the atomic mass of an element is a weighted average of the masses of the naturally occurring i ...
sec 3- Counting atoms - Nutley Public Schools
... • Imagine that your semester grade depends 60% on exam scores and 40% on laboratory explorations. • Your exam scores would count more heavily toward your final grade. • In this section, you will learn that the atomic mass of an element is a weighted average of the masses of the naturally occurring i ...
... • Imagine that your semester grade depends 60% on exam scores and 40% on laboratory explorations. • Your exam scores would count more heavily toward your final grade. • In this section, you will learn that the atomic mass of an element is a weighted average of the masses of the naturally occurring i ...
Elements Compounds
... Ionic bond – electron from Na is transferred to Cl, this causes a charge imbalance in each atom. The Na becomes (Na+) and the Cl becomes (Cl-), charged particles or ions. ...
... Ionic bond – electron from Na is transferred to Cl, this causes a charge imbalance in each atom. The Na becomes (Na+) and the Cl becomes (Cl-), charged particles or ions. ...
Problem Solving Drill - Rapid Learning Center
... Instructions: (1) Read the problem and answer choices carefully (2) Work the problems on paper as needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 10. Copper is a metal with high thermal and electric conductivity. This metal and its alloys have been used for thou ...
... Instructions: (1) Read the problem and answer choices carefully (2) Work the problems on paper as needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 10. Copper is a metal with high thermal and electric conductivity. This metal and its alloys have been used for thou ...
Problem Solving Drill - Rapid Learning Center
... Instructions: (1) Read the problem and answer choices carefully (2) Work the problems on paper as needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 10. Copper is a metal with high thermal and electric conductivity. This metal and its alloys have been used for thou ...
... Instructions: (1) Read the problem and answer choices carefully (2) Work the problems on paper as needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 10. Copper is a metal with high thermal and electric conductivity. This metal and its alloys have been used for thou ...
Is Kr the symbol for Kryptonite?
... Formulas of compounds • Write the formulas for the following compounds: – A phosphorus atom bonded to three chlorine atoms – A molecule containing two boron atoms and six hydrogen atoms – A compound containing one calcium atom for every two chlorine atoms – Four hydrogen atoms bonded to a singl ...
... Formulas of compounds • Write the formulas for the following compounds: – A phosphorus atom bonded to three chlorine atoms – A molecule containing two boron atoms and six hydrogen atoms – A compound containing one calcium atom for every two chlorine atoms – Four hydrogen atoms bonded to a singl ...
Promethium

Promethium, originally prometheum, is a chemical element with symbol Pm and atomic number 61. All of its isotopes are radioactive; it is one of only two such elements that are followed in the periodic table by elements with stable forms, a distinction shared with technetium. Chemically, promethium is a lanthanide, which forms salts when combined with other elements. Promethium shows only one stable oxidation state of +3; however, a few +2 compounds may exist.In 1902, Bohuslav Brauner suggested there was an element with properties intermediate between those of the known elements neodymium (60) and samarium (62); this was confirmed in 1914 by Henry Moseley who, having measured the atomic numbers of all the elements then known, found there was an element with atomic number 61. In 1926, an Italian and an American group claimed to have isolated a sample of element 61; both ""discoveries"" were soon proven to be false. In 1938, during a nuclear experiment conducted at Ohio State University, a few radioactive nuclides were produced that certainly were not radioisotopes of neodymium or samarium, but there was a lack of chemical proof that element 61 was produced, and the discovery was not generally recognized. Promethium was first produced and characterized at Oak Ridge National Laboratory in 1945 by the separation and analysis of the fission products of uranium fuel irradiated in a graphite reactor. The discoverers proposed the name ""prometheum"" (the spelling was subsequently changed), derived from Prometheus, the Titan in Greek mythology who stole fire from Mount Olympus and brought it down to humans, to symbolize ""both the daring and the possible misuse of mankind's intellect"". However, a sample of the metal was made only in 1963.There are two possible sources for natural promethium: rare decays of natural europium-151 (producing promethium-147), and uranium (various isotopes). Practical applications exist only for chemical compounds of promethium-147, which are used in luminous paint, atomic batteries, and thickness measurement devices, even though promethium-145 is the most stable promethium isotope. Because natural promethium is exceedingly scarce, it is typically synthesized by bombarding uranium-235 (enriched uranium) with thermal neutrons to produce promethium-147.