Everything is made of atoms.
... Modern atomic theory began with the publication in 1808 by British chemist and physicist John Dalton of his experimental conclusions that: 1. all atoms of an element have the same size and weight, and 2. atoms of elements unite chemically in simple numerical ratios to form compounds. http://www.neut ...
... Modern atomic theory began with the publication in 1808 by British chemist and physicist John Dalton of his experimental conclusions that: 1. all atoms of an element have the same size and weight, and 2. atoms of elements unite chemically in simple numerical ratios to form compounds. http://www.neut ...
The Structure of the Atom Chapter 4
... Atoms of the same element are chemically and physically the same. Atoms of different elements are chemically and physically different. When atoms combine in a chemical reaction, they do so in small, whole number ratios. In a chemical reaction, atoms aren’t Created or destroyed, they are simply rearr ...
... Atoms of the same element are chemically and physically the same. Atoms of different elements are chemically and physically different. When atoms combine in a chemical reaction, they do so in small, whole number ratios. In a chemical reaction, atoms aren’t Created or destroyed, they are simply rearr ...
Henry Moseley, the Atomic Number, and Synthesis
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
SCIENCE: EIGHTH GRADE CRT FIRST QUARTER
... Which subatomic particles in an atom have no charge? Which subatomic particles are found in the nucleus of an atom? Where are electrons located in an atom? Who was the Greek philosopher responsible for naming the atom? Which two subatomic particles have the greatest mass? An element that is a very r ...
... Which subatomic particles in an atom have no charge? Which subatomic particles are found in the nucleus of an atom? Where are electrons located in an atom? Who was the Greek philosopher responsible for naming the atom? Which two subatomic particles have the greatest mass? An element that is a very r ...
SCIENCE: EIGHTH GRADE CRT FIRST QUARTER
... Which subatomic particles in an atom have no charge? Which subatomic particles are found in the nucleus of an atom? Where are electrons located in an atom? Who was the Greek philosopher responsible for naming the atom? Which two subatomic particles have the greatest mass? An element that is a very r ...
... Which subatomic particles in an atom have no charge? Which subatomic particles are found in the nucleus of an atom? Where are electrons located in an atom? Who was the Greek philosopher responsible for naming the atom? Which two subatomic particles have the greatest mass? An element that is a very r ...
Unit 2, Day 25
... is used because the mass of each subatomic particle is too small to measure in grams The mass of each atom is equal to the number of protons plus the number of neutrons. (Electrons are ignored, because their mass is so small that they don’t affect the mass enough) ...
... is used because the mass of each subatomic particle is too small to measure in grams The mass of each atom is equal to the number of protons plus the number of neutrons. (Electrons are ignored, because their mass is so small that they don’t affect the mass enough) ...
Henry Moseley, the Atomic Number, and Synthesis
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
SCIENCE: EIGHTH GRADE CRT FIRST QUARTER
... Which subatomic particles in an atom have no charge? Which subatomic particles are found in the nucleus of an atom? Where are electrons located in an atom? Who was the Greek philosopher responsible for naming the atom? Which two subatomic particles have the greatest mass? An element that is a very r ...
... Which subatomic particles in an atom have no charge? Which subatomic particles are found in the nucleus of an atom? Where are electrons located in an atom? Who was the Greek philosopher responsible for naming the atom? Which two subatomic particles have the greatest mass? An element that is a very r ...
Chapter 5 Atomic Structure & the Periodic Table
... stone • Neptunium = after the planet Neptune • Hydrogen (H), Sulfur (S), Carbon (C) • Gold (Au), Lead (Pb), Iron (Fe), Copper (Cu) = symbols come from latin names. ...
... stone • Neptunium = after the planet Neptune • Hydrogen (H), Sulfur (S), Carbon (C) • Gold (Au), Lead (Pb), Iron (Fe), Copper (Cu) = symbols come from latin names. ...
Atoms Ions Valence Electrons Isotopes
... – an atom with 2 valence electrons will lose 2 electrons to reach 0 – an atom with 6 valence electrons will gain 2 electrons to reach 8 ...
... – an atom with 2 valence electrons will lose 2 electrons to reach 0 – an atom with 6 valence electrons will gain 2 electrons to reach 8 ...
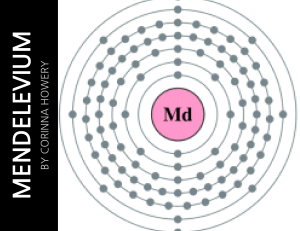
Mendelevium
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
Atoms - Chemistry Land
... cold beaker above it, you can see water condensing on it. So the dry paper actually has water in it. Finally as it burns out you see the ashes, which is like dirt or earth. ...
... cold beaker above it, you can see water condensing on it. So the dry paper actually has water in it. Finally as it burns out you see the ashes, which is like dirt or earth. ...
Atomic Mass Lab (Flaskum)
... 10. The atomic mass of chlorine on the periodic table is not a whole number. What does this indicate? What is the most common isotope of chlorine? ...
... 10. The atomic mass of chlorine on the periodic table is not a whole number. What does this indicate? What is the most common isotope of chlorine? ...
File
... Definition: Atomic Number: # of p+ in the nucleus Of an element is the _______________________________________________ of its atoms. No two elements have the _______ Same atomic number. This number defines an ________________. ...
... Definition: Atomic Number: # of p+ in the nucleus Of an element is the _______________________________________________ of its atoms. No two elements have the _______ Same atomic number. This number defines an ________________. ...
Oct 14th ,2015
... atom in terms of its subatomic particles; isotopes and ions; differentiate between the classification and separation of matter ...
... atom in terms of its subatomic particles; isotopes and ions; differentiate between the classification and separation of matter ...
The ocean is a mixture.
... Transition elements have properties similar to one another and to other metals, but their properties do not fit in with those of any other family. Many transition metals combine chemically with oxygen to form compounds called oxides. They have one or two electrons in the outer level Reactivity: less ...
... Transition elements have properties similar to one another and to other metals, but their properties do not fit in with those of any other family. Many transition metals combine chemically with oxygen to form compounds called oxides. They have one or two electrons in the outer level Reactivity: less ...
Ch 3 Outline- Intro to Atom and Periodic Table
... b. Synthesized (made) when nuclear particles are forced to crash into each other. c. Called “synthetic elements” i. Example Plutonium (Pu) is made in a nuclear reactor. d. Elements with atomic numbers over 95 such as Curium (Cm), Einsteinium (Es) and others can only be synthesized using a particle a ...
... b. Synthesized (made) when nuclear particles are forced to crash into each other. c. Called “synthetic elements” i. Example Plutonium (Pu) is made in a nuclear reactor. d. Elements with atomic numbers over 95 such as Curium (Cm), Einsteinium (Es) and others can only be synthesized using a particle a ...
North Haven Public Schools Curriculum
... The quantity one mole is set by defining one mole of carbon; 12 atoms to have a mass of exactly 12 grams. One mole equals 6.02 x 1023 particles (atoms or moles) The mass of a substance can be converted to moles, number of particles, or volume of gas at standard temperature and pressure. ...
... The quantity one mole is set by defining one mole of carbon; 12 atoms to have a mass of exactly 12 grams. One mole equals 6.02 x 1023 particles (atoms or moles) The mass of a substance can be converted to moles, number of particles, or volume of gas at standard temperature and pressure. ...
Charge of Ion = number of protons – number of electrons A neutral
... What causes an atom to become an ion? • When an atom loses or gains one or more electrons it acquires a net electrical charge called an ion. • The net charge of an ion is found by subtracting the number of electrons from the number of protons Charge of Ion = number of protons – number of electrons ...
... What causes an atom to become an ion? • When an atom loses or gains one or more electrons it acquires a net electrical charge called an ion. • The net charge of an ion is found by subtracting the number of electrons from the number of protons Charge of Ion = number of protons – number of electrons ...
Chapter 5: Atomic Structure & The Periodic Table
... was unknown at the time. 2000 years after Democritus, the real nature of atoms and observable changes at the atomic level were established. John Dalton (1766-1844)—English school teacher, performed experiments to test and correct his atomic theory. ...
... was unknown at the time. 2000 years after Democritus, the real nature of atoms and observable changes at the atomic level were established. John Dalton (1766-1844)—English school teacher, performed experiments to test and correct his atomic theory. ...
4.1 Early Theories of Matter The Philosophers Democritus – Greek
... Accidentally discovered radioactivity while working with uranium salts Radioactivity – the phenomenon of rays or particles being produced spontaneously by unstable atomic nuclei (also referred to as radiation) Radioactivity can accompany changes in the identities of elements Transmutation – ...
... Accidentally discovered radioactivity while working with uranium salts Radioactivity – the phenomenon of rays or particles being produced spontaneously by unstable atomic nuclei (also referred to as radiation) Radioactivity can accompany changes in the identities of elements Transmutation – ...
ATOMS AND ELEMENTS Evolution of Atomic Theory
... scale, composed of particles described as atomos, meaning “indivisible” ! While atoms can in fact be broken down into smaller particles, the atoms of each element are distinct from each other, making them the fundamental unit of ...
... scale, composed of particles described as atomos, meaning “indivisible” ! While atoms can in fact be broken down into smaller particles, the atoms of each element are distinct from each other, making them the fundamental unit of ...
Chapter 4: Concept 4.2
... subatomic particles. A proton is a subatomic particle with a single unit of positive electrical charge (+). An electron is a subatomic particle with a single unit of negative electrical charge (-). A third type of subatomic particle, the neutron, is electrically neutral, meaning it has no electrical ...
... subatomic particles. A proton is a subatomic particle with a single unit of positive electrical charge (+). An electron is a subatomic particle with a single unit of negative electrical charge (-). A third type of subatomic particle, the neutron, is electrically neutral, meaning it has no electrical ...
Atoms & Radioactivity
... When an atom loses electrons it becomes a positive ion. When an atom gains electrons it becomes a negative ion. ...
... When an atom loses electrons it becomes a positive ion. When an atom gains electrons it becomes a negative ion. ...
No Slide Title
... Periodic Table 30 • Which group of elements share characteristics of both metals and nonmetals? • What are metalloids or semi-metals? ...
... Periodic Table 30 • Which group of elements share characteristics of both metals and nonmetals? • What are metalloids or semi-metals? ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.