الشريحة 1
... Colligative Properties Some physical properties of solutions differ in important ways from those of pure solvent. For example, pure water freezes at 0 oC, but aqueous solutions freeze at lower temperatures. Ethylene glycol is added to the water in radiators of cars as an antifreeze to lower the fre ...
... Colligative Properties Some physical properties of solutions differ in important ways from those of pure solvent. For example, pure water freezes at 0 oC, but aqueous solutions freeze at lower temperatures. Ethylene glycol is added to the water in radiators of cars as an antifreeze to lower the fre ...
AP Chemistry Chapter 1: Chemical Foundations
... Concept Check Which of the following is a homogeneous mixture? ...
... Concept Check Which of the following is a homogeneous mixture? ...
Condensed Matter 2
... completion of this course the student is expected to have a firm understanding the relationship between pressure, temperature, and volume on the behaviour of solids liquids and gasses. They should be able to apply these relationships to understand phenomena such as vapour pressure, surface tension ...
... completion of this course the student is expected to have a firm understanding the relationship between pressure, temperature, and volume on the behaviour of solids liquids and gasses. They should be able to apply these relationships to understand phenomena such as vapour pressure, surface tension ...
Slide 1
... (a) This process is spontaneous. Whenever two objects at different temperatures are brought into contact, heat is transferred from the hotter object to the colder one. (Section 5.1) Thus, heat is transferred from the hot metal to the cooler water. The final temperature, after the metal and water ach ...
... (a) This process is spontaneous. Whenever two objects at different temperatures are brought into contact, heat is transferred from the hotter object to the colder one. (Section 5.1) Thus, heat is transferred from the hot metal to the cooler water. The final temperature, after the metal and water ach ...
practice problems of chap4_5 - Chemistry
... Hint: p. 187. Equation (5.12). Example 5.9. M=dRT/P=mRT/VP=7.10g*0.082 L• atm / (mol • K) *(40+273.15)K /[(741/760)atm*5.40L]= 34.6 18. What is the density of HCl gas in grams per liter at 700 mmHg and 25 oC? (a) 0.54 (b) 1.37 (c) 2.24 (d) 2.97 (e) 3.57 Hint:p.p.186, d=m/V=PM/RT=(700/760)atm*36.45g/ ...
... Hint: p. 187. Equation (5.12). Example 5.9. M=dRT/P=mRT/VP=7.10g*0.082 L• atm / (mol • K) *(40+273.15)K /[(741/760)atm*5.40L]= 34.6 18. What is the density of HCl gas in grams per liter at 700 mmHg and 25 oC? (a) 0.54 (b) 1.37 (c) 2.24 (d) 2.97 (e) 3.57 Hint:p.p.186, d=m/V=PM/RT=(700/760)atm*36.45g/ ...
semester 1 examination 2009 the university of the
... 3) (a) (i) A container holds two gases, A and B. Gas A has a partial pressure of 325 torr and gas B has a partial pressure of 488 torr. What percent of the molecules in the mixture is gas A? ...
... 3) (a) (i) A container holds two gases, A and B. Gas A has a partial pressure of 325 torr and gas B has a partial pressure of 488 torr. What percent of the molecules in the mixture is gas A? ...
Journal of Molecular Catalysis A: Chemical Enhancing
... presence of double bond [33] and upfield shift in the signal of saturated methylene protons containing alcoholic group. Monohalogenation of diols [34] is often a stubborn problem in different synthetic endeavoures, as there is considerable formation of disubstituted product. Immiscibility of ionic li ...
... presence of double bond [33] and upfield shift in the signal of saturated methylene protons containing alcoholic group. Monohalogenation of diols [34] is often a stubborn problem in different synthetic endeavoures, as there is considerable formation of disubstituted product. Immiscibility of ionic li ...
AP Chemistry - Problem Drill 03: Matter and Energy Question No. 1
... Although temperature changes are often common in chemical changes, not all chemical changes result in a temperature change. Therefore, the lack of a significant temperature change is not proof that the change is physical. E. Incorrect! The dry ingredients do not disappear; they spread out when they ...
... Although temperature changes are often common in chemical changes, not all chemical changes result in a temperature change. Therefore, the lack of a significant temperature change is not proof that the change is physical. E. Incorrect! The dry ingredients do not disappear; they spread out when they ...
chapter15-burno.1348..
... Significance of the Equilibrium Constant The significance of the equilibrium constant lies in the fact that for a chemical reaction taking place at a particular temperature T, the equilibrium constant (KC or Kp) has a particular numerical value. This means that no matter what the starting concentra ...
... Significance of the Equilibrium Constant The significance of the equilibrium constant lies in the fact that for a chemical reaction taking place at a particular temperature T, the equilibrium constant (KC or Kp) has a particular numerical value. This means that no matter what the starting concentra ...
Міністерство охорони здоров`я України
... dispersion of substances in the solutions can be different. Particle size is a very important feature which causes many physical and chemical properties of the solutions. According to the particle size solutions can be classified as: 1) true solutions (particle size is less than 10-9 m), which can ...
... dispersion of substances in the solutions can be different. Particle size is a very important feature which causes many physical and chemical properties of the solutions. According to the particle size solutions can be classified as: 1) true solutions (particle size is less than 10-9 m), which can ...
Decomposition Reactions
... The reaction you will investigate in this experiment is the decomposition of hydrogen peroxide. You may be familiar with this compound; hydrogen peroxide solution can be commonly purchased in any pharmacy and it is found in most home medicine cabinets. In fact, the reaction you will be studying take ...
... The reaction you will investigate in this experiment is the decomposition of hydrogen peroxide. You may be familiar with this compound; hydrogen peroxide solution can be commonly purchased in any pharmacy and it is found in most home medicine cabinets. In fact, the reaction you will be studying take ...
Moles Worksheet
... How are the terms “molar mass” and “atomic mass” different from one another? “Molar mass” is used to describe the mass of one mole of a chemical compound, while “atomic mass” is used to describe the mass of one mole of an element or the mass of one atom of an element. ...
... How are the terms “molar mass” and “atomic mass” different from one another? “Molar mass” is used to describe the mass of one mole of a chemical compound, while “atomic mass” is used to describe the mass of one mole of an element or the mass of one atom of an element. ...
K c
... 1. The concentrations of the reacting species in the condensed phase are expressed in mol/L (M). In the gaseous phase, the concentrations can be expressed in M. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium c ...
... 1. The concentrations of the reacting species in the condensed phase are expressed in mol/L (M). In the gaseous phase, the concentrations can be expressed in M. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium c ...
Supplemental Notes
... 1. In terms of mass: The mass of one mole of any substance is equal to its relative atomic or molecular mass taken in grams, e.g., 1 mole of Fe = 56 g (Atomic mass, Ar) 1 mole of H2O = 18 g (Molecular mass, Mr) 2. In terms of number: One mole of any substance contains particles equal to 6.02 x 1023. ...
... 1. In terms of mass: The mass of one mole of any substance is equal to its relative atomic or molecular mass taken in grams, e.g., 1 mole of Fe = 56 g (Atomic mass, Ar) 1 mole of H2O = 18 g (Molecular mass, Mr) 2. In terms of number: One mole of any substance contains particles equal to 6.02 x 1023. ...
Proper Piping for Vacuum Systems
... should be corrected if any hydrocarbons or other substances are present. For hydrocarbons, good installation practice is to use at least 45 ft, regardless of barometric pressure. ...
... should be corrected if any hydrocarbons or other substances are present. For hydrocarbons, good installation practice is to use at least 45 ft, regardless of barometric pressure. ...
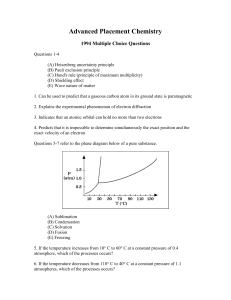
Advanced Placement Chemistry
... 12. Is added to silicon to enhance its properties as a semiconductor 13. Utilized as a shield from sources of radiation Directions: Each of the questions or incomplete statements below is followed by five suggested answers or completions. Select the one that is best in each case and then fill in the ...
... 12. Is added to silicon to enhance its properties as a semiconductor 13. Utilized as a shield from sources of radiation Directions: Each of the questions or incomplete statements below is followed by five suggested answers or completions. Select the one that is best in each case and then fill in the ...
Final Review Answers
... How does each of the following affect the solubility of (a) a solid dissolved in a liquid, and (b) a gas dissolved in a liquid. a. an increase in temperature (a) more collisions between particles causing an increase in dissolving particles (b) decreases solubility, as T increases more dissolved gas ...
... How does each of the following affect the solubility of (a) a solid dissolved in a liquid, and (b) a gas dissolved in a liquid. a. an increase in temperature (a) more collisions between particles causing an increase in dissolving particles (b) decreases solubility, as T increases more dissolved gas ...