Material
... uniform temperature is attained throughout the combined system. The system is then in thermal equilibrium. Experience shows, all systems which are in thermal equilibrium with a given system are also in thermal equilibrium with each other. This principle defines the temperature of a thermodynamic sys ...
... uniform temperature is attained throughout the combined system. The system is then in thermal equilibrium. Experience shows, all systems which are in thermal equilibrium with a given system are also in thermal equilibrium with each other. This principle defines the temperature of a thermodynamic sys ...
Chapter 6 HEAT CAPACITY, ENTHALPY, ENTROPY, AND
... As a result of experimental measurement, Dulong and Petit introduced an empirical rule in 1819 which states that the molar heat capacities of all solid elements have the value 3R(=24.9 J/K), and, in 1865, Kopp introduced a rule which states that, at ordinary temperatures, the molar heat capacity of ...
... As a result of experimental measurement, Dulong and Petit introduced an empirical rule in 1819 which states that the molar heat capacities of all solid elements have the value 3R(=24.9 J/K), and, in 1865, Kopp introduced a rule which states that, at ordinary temperatures, the molar heat capacity of ...
KEY
... 4. decreases Explanation: Increasing the total pressure on the system by decreasing its volume will shift the equilibrium toward the side of the reaction with fewer numbers of moles of gaseous components. If the total number of moles of gas is the same on the product and reactant sides of the balanc ...
... 4. decreases Explanation: Increasing the total pressure on the system by decreasing its volume will shift the equilibrium toward the side of the reaction with fewer numbers of moles of gaseous components. If the total number of moles of gas is the same on the product and reactant sides of the balanc ...
Chemical Equilibrium - Chemistry with Mrs. Caruso Let the Bonding
... Kp- _partial pressures__are used in the calculation of this equilibrium. Relationship between Kp and Kc: Based on the ideal gas law: Therefore, Kp = Kc(RT)∆n Ex. At 327°C, the equilibrium concentrations are [CH3OH]= .15M, [CO]= .24M, and [H2]=1.1M for the reaction CH3OH (g) ⇌ CO (g) + 2H2 (g) Calcul ...
... Kp- _partial pressures__are used in the calculation of this equilibrium. Relationship between Kp and Kc: Based on the ideal gas law: Therefore, Kp = Kc(RT)∆n Ex. At 327°C, the equilibrium concentrations are [CH3OH]= .15M, [CO]= .24M, and [H2]=1.1M for the reaction CH3OH (g) ⇌ CO (g) + 2H2 (g) Calcul ...
selected experiments in organic chemistry
... Capillary melting point tubes are about 6-7 cm in length and 1 mm in diameter. They are sealed by rotating one end of the capillary tube in the edge of a small hot flame. The dry solid is ground to a fine powder on a piece of paper with a spatula. The open end of the capillary is then pushed into th ...
... Capillary melting point tubes are about 6-7 cm in length and 1 mm in diameter. They are sealed by rotating one end of the capillary tube in the edge of a small hot flame. The dry solid is ground to a fine powder on a piece of paper with a spatula. The open end of the capillary is then pushed into th ...
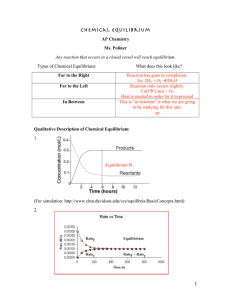
Equilibrium notes (complete)
... Why bother having 2 constants in the same expression? Combine the constants to get ...
... Why bother having 2 constants in the same expression? Combine the constants to get ...
Laboratory Practices from Physical Chemistry
... Using analytical scales, we determine the weight, m1, of empty Dumas flask with stopper. We add ca 1–2 ml of sample to Dumas flask and we place it in the thermostatic bath with temperature Tk. Only the end of the flask neck (1–2 cm) has to project from the bath. During sample evaporation, all air fr ...
... Using analytical scales, we determine the weight, m1, of empty Dumas flask with stopper. We add ca 1–2 ml of sample to Dumas flask and we place it in the thermostatic bath with temperature Tk. Only the end of the flask neck (1–2 cm) has to project from the bath. During sample evaporation, all air fr ...
Topic 3 MOLE Avodagro`s number = 6.02 x 1023 things = 1 mole 1
... Topic 3 MOLE Avodagro’s number = 6.02 x 1023 things = 1 mole 1 mole of any substance weighs its formula weight in grams (molar mass) EMPIRICAL/MOLECULAR FORMULAS From % to empirical = % to mass, mass to mole, divide by small, times ’til whole. From molecular to empirical = (molecular mass) / ...
... Topic 3 MOLE Avodagro’s number = 6.02 x 1023 things = 1 mole 1 mole of any substance weighs its formula weight in grams (molar mass) EMPIRICAL/MOLECULAR FORMULAS From % to empirical = % to mass, mass to mole, divide by small, times ’til whole. From molecular to empirical = (molecular mass) / ...
HC_Chapter_15_-_Solutions_files/Chapter 15 Objectives and Notes
... combined materials that are uniformly distributed throughout the mixture. The component parts do not have to be in a fixed ratio and they do not lose their own identity. a. Because the components are physically mixed together, they can be separated by physical methods such as distillation or crystal ...
... combined materials that are uniformly distributed throughout the mixture. The component parts do not have to be in a fixed ratio and they do not lose their own identity. a. Because the components are physically mixed together, they can be separated by physical methods such as distillation or crystal ...
equilibrium
... Removing NH3 the reverse reaction slows down - (fewer collisions) (the forward reaction is now the faster one) Reaction SHIFTS RIGHT (to replace the lost NH3 ) Result: N2 and H2 decrease, NH3 increases ...
... Removing NH3 the reverse reaction slows down - (fewer collisions) (the forward reaction is now the faster one) Reaction SHIFTS RIGHT (to replace the lost NH3 ) Result: N2 and H2 decrease, NH3 increases ...
Engineering Thermodynamics
... system of liquid water and water vapor (steam) contains two phases. A pure substance is one that is uniform and invariable in chemical composition. A pure substance can exist in more than one phase, but its chemical composition must be the same in each phase. For example, if liquid water and water v ...
... system of liquid water and water vapor (steam) contains two phases. A pure substance is one that is uniform and invariable in chemical composition. A pure substance can exist in more than one phase, but its chemical composition must be the same in each phase. For example, if liquid water and water v ...
U6B _13-14
... Titrating a strong acid with a strong base pH is always = 7 The titration curve graph shows the pH of the equivalence point. Take the vertical region and cut the length in half and then look to what pH value aligns to that point. ...
... Titrating a strong acid with a strong base pH is always = 7 The titration curve graph shows the pH of the equivalence point. Take the vertical region and cut the length in half and then look to what pH value aligns to that point. ...
Liquid-gas transition of neon in quasi-one
... obtaining wrong thermodynamic averages from the calculation. To avoid that, we simulated the corresponding 1D neon systems at temperatures at which we were certain that the correlation length was smaller than the simulation cells used. As a check, we performed an additional set of simulations at den ...
... obtaining wrong thermodynamic averages from the calculation. To avoid that, we simulated the corresponding 1D neon systems at temperatures at which we were certain that the correlation length was smaller than the simulation cells used. As a check, we performed an additional set of simulations at den ...
Term 111, Final Exam (All correct choices are A): 1. What is the
... 18. At very high pressures (~ 1000 atm), the measured pressure exerted by real gases is greater than that predicted by the ideal gas equation. This is mainly because A) the volume occupied by the gas molecules themselves becomes significant. B) real gases will condense to form solids at 1000 atm pre ...
... 18. At very high pressures (~ 1000 atm), the measured pressure exerted by real gases is greater than that predicted by the ideal gas equation. This is mainly because A) the volume occupied by the gas molecules themselves becomes significant. B) real gases will condense to form solids at 1000 atm pre ...
Document
... B) Equilibrium in molecular systems is dynamic, with two opposing processes balancing one another. C) The value of the equilibrium constant for a given reaction mixture is the same regardless of the direction from which equilibrium is attained. D) A system moves spontaneously toward a state of equil ...
... B) Equilibrium in molecular systems is dynamic, with two opposing processes balancing one another. C) The value of the equilibrium constant for a given reaction mixture is the same regardless of the direction from which equilibrium is attained. D) A system moves spontaneously toward a state of equil ...
In Class Problems and Notes AP Chemistry General Equilibrium
... When the reaction is at equilibrium and the [B2] is increased the reaction proceeds from left-to-right to relieve the stress. When B2 is added the reaction shifts in a direction to relieve the stress, by trying to decrease the amount of B2. So the reactions proceeds from left-to-right. Similarly whe ...
... When the reaction is at equilibrium and the [B2] is increased the reaction proceeds from left-to-right to relieve the stress. When B2 is added the reaction shifts in a direction to relieve the stress, by trying to decrease the amount of B2. So the reactions proceeds from left-to-right. Similarly whe ...
SOLVING L-L EXTRACTION PROBLEMS WITH EXCEL
... iquid-liquid (L-L) extraction, sometimes called solvent extraction, is the separation of the components of a liquid solution by contact with another insoluble liquid. One method used to solve this problem is the equilibrium stage concept.[1] Most L-L extraction processes concern only one component t ...
... iquid-liquid (L-L) extraction, sometimes called solvent extraction, is the separation of the components of a liquid solution by contact with another insoluble liquid. One method used to solve this problem is the equilibrium stage concept.[1] Most L-L extraction processes concern only one component t ...













![Keq = [A] [B] [C] [D]](http://s1.studyres.com/store/data/014463360_1-50a2de0db1e8b9a361c4b31c6e85c28d-300x300.png)