
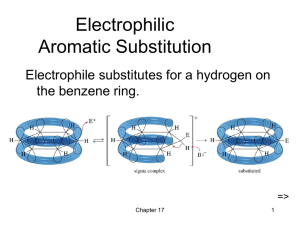
Electrophilic Aromatic Substit
... Activating, O-, PDirecting Substituents • Alkyl groups stabilize the sigma complex by induction, donating electron density through the sigma bond. • Substituents with a lone pair of electrons stabilize the sigma complex by resonance. ...
... Activating, O-, PDirecting Substituents • Alkyl groups stabilize the sigma complex by induction, donating electron density through the sigma bond. • Substituents with a lone pair of electrons stabilize the sigma complex by resonance. ...
Ethers and Epoxides
... These 3 membered rings are named using the term “epoxy” as one substituent bridging two adjacent C atoms. (NOT like cyclopropane) ...
... These 3 membered rings are named using the term “epoxy” as one substituent bridging two adjacent C atoms. (NOT like cyclopropane) ...
amine cured-epoxy matrices
... aromatic amines require elevated temperature to effectively cure most epoxy resins. It should also be mentioned that catalysts can be added to increase the reaction rate of amines with epoxy resins but if used in excess oftentimes at the cost of a reduction in performance. The amine curing agent sel ...
... aromatic amines require elevated temperature to effectively cure most epoxy resins. It should also be mentioned that catalysts can be added to increase the reaction rate of amines with epoxy resins but if used in excess oftentimes at the cost of a reduction in performance. The amine curing agent sel ...
T_AllylCF3paperBM[5]
... CF3-substituted allyl alcohols 1 promoted by Bronsted or Lewis acids were investigated (Scheme 1, нет ее). Dehydroxylation of 1 can lead to allyl cation having resonance forms A and B. However, due to strong electron-withdrawing character of CF3-group form A is destabilized significantly. As a resul ...
... CF3-substituted allyl alcohols 1 promoted by Bronsted or Lewis acids were investigated (Scheme 1, нет ее). Dehydroxylation of 1 can lead to allyl cation having resonance forms A and B. However, due to strong electron-withdrawing character of CF3-group form A is destabilized significantly. As a resul ...
A Crash Course In Organic Chemistry
... Compounds containing only bromine are generally not useful. Compounds containing only chlorine are subject to limited use, are toxic, and can cause arrhythmias. The best of the chlorinated agents are ethyl chloride and trichloroethylene ...
... Compounds containing only bromine are generally not useful. Compounds containing only chlorine are subject to limited use, are toxic, and can cause arrhythmias. The best of the chlorinated agents are ethyl chloride and trichloroethylene ...
fundamentals of structure and reactivity of organic compounds
... Solution. Organic chemistry is a chemistry of carbon compounds. The properties of organic compounds are determined for the most extent by the electronic structure of the carbon atom and nature of its chemical bonds. In the excited state (1s22s12p3) carbon atom has four unpaired electrons and can the ...
... Solution. Organic chemistry is a chemistry of carbon compounds. The properties of organic compounds are determined for the most extent by the electronic structure of the carbon atom and nature of its chemical bonds. In the excited state (1s22s12p3) carbon atom has four unpaired electrons and can the ...
Alcohols, Phenols , Phenols and Ethers Alcohols
... In ethers, the four electron pairs, i.e., the two bond pairs and two lone pairs of electrons on oxygen are arranged approximately in a tetrahedral arrangement. The bond angle is slightly greater than the tetrahedral angle due to the repulsive interaction between the two bulky (–R) groups. The C–O bo ...
... In ethers, the four electron pairs, i.e., the two bond pairs and two lone pairs of electrons on oxygen are arranged approximately in a tetrahedral arrangement. The bond angle is slightly greater than the tetrahedral angle due to the repulsive interaction between the two bulky (–R) groups. The C–O bo ...
Chapter 1--Title - Chemistry Workshop
... Chiral quaternary ammonium salts cannot undergo nitrogen inversion because they lack an unshared electron pair on the nitrogen atom ...
... Chiral quaternary ammonium salts cannot undergo nitrogen inversion because they lack an unshared electron pair on the nitrogen atom ...













![T_AllylCF3paperBM[5]](http://s1.studyres.com/store/data/003584459_1-3decab572f7fca68901a941affab18ea-300x300.png)







