Thermodynamics and Equilibrium
... spontaneously because doing so lowers its energy, but it does not move back up the hill spontaneously because an input of energy is required to do so. Thus, it would be tempting to conclude that processes should be spontaneous if they are exothermic. However, there are many examples of spontaneous e ...
... spontaneously because doing so lowers its energy, but it does not move back up the hill spontaneously because an input of energy is required to do so. Thus, it would be tempting to conclude that processes should be spontaneous if they are exothermic. However, there are many examples of spontaneous e ...
Subatomic Physics: the Notes - McMaster Physics and Astronomy
... By this time the electric charge of the electron had been measured (through the Millikan oil-drop experiment of 1909) and so it was known that the electron had a charge equal in size to (but opposite in sign from) the charge, q = e, of the Hydrogen ion (what we now call the Hydrogen nucleus, or pro ...
... By this time the electric charge of the electron had been measured (through the Millikan oil-drop experiment of 1909) and so it was known that the electron had a charge equal in size to (but opposite in sign from) the charge, q = e, of the Hydrogen ion (what we now call the Hydrogen nucleus, or pro ...
Einstein`s Big Idea Teacher`s Guide
... oxygen), water (hydrogen, oxygen), living organisms (carbon, nitrogen, oxygen, hydrogen), the sun (hydrogen, helium), and jewelry (nickel, silver, gold). Do these elements have mass? (Yes. The periodic table provides the relative mass of each element in atomic mass units.) Clarify with students the ...
... oxygen), water (hydrogen, oxygen), living organisms (carbon, nitrogen, oxygen, hydrogen), the sun (hydrogen, helium), and jewelry (nickel, silver, gold). Do these elements have mass? (Yes. The periodic table provides the relative mass of each element in atomic mass units.) Clarify with students the ...
Exercise II
... The restriction of having identical attacking and leaving groups was purposefully chosen for one component of the present exercise in order to allow an easier analysis without any loss of the concepts inherent in more general SN 2 reactions. In this exercise we will examine both an identity SN 2 rea ...
... The restriction of having identical attacking and leaving groups was purposefully chosen for one component of the present exercise in order to allow an easier analysis without any loss of the concepts inherent in more general SN 2 reactions. In this exercise we will examine both an identity SN 2 rea ...
confined plasmas theory toroidally
... interferometer data22 in Fig. 1共c兲 reveals modes in the EAE frequency range very early in the discharge and these modes persist for only 0.3 s before disappearing. The EAE is expected to occur at about twice the TAE frequency. The EAE is also observed on electron cyclotron emission measurements from ...
... interferometer data22 in Fig. 1共c兲 reveals modes in the EAE frequency range very early in the discharge and these modes persist for only 0.3 s before disappearing. The EAE is expected to occur at about twice the TAE frequency. The EAE is also observed on electron cyclotron emission measurements from ...
Topic 6 Kinetics File
... increase of number of moles of gaseous molecules; 2) change of state from solid to liquid or liquid to gas; 3) increase of temperature Exothermic: A reaction in which energy is evolved. ΔH is –. Products more stable than reactants. Gibb’s free energy: Must be negative for reaction to be spontaneous. ...
... increase of number of moles of gaseous molecules; 2) change of state from solid to liquid or liquid to gas; 3) increase of temperature Exothermic: A reaction in which energy is evolved. ΔH is –. Products more stable than reactants. Gibb’s free energy: Must be negative for reaction to be spontaneous. ...
CHAPTER 9
... Sn undergoes an oxidation number change of 2 units. Fe undergoes an oxidation number change of 6 units. ...
... Sn undergoes an oxidation number change of 2 units. Fe undergoes an oxidation number change of 6 units. ...
Neutron Stars
... which it was realized that neutron stars were likely to be rapidly rotating and to have intense magnetic fields. Pacini3 predicted that a rotating magnetized neutron starwould emit radio waves. But it was not until the 1967 Bell and Hewish4 discovery of radio pulsars that the existence of neutron st ...
... which it was realized that neutron stars were likely to be rapidly rotating and to have intense magnetic fields. Pacini3 predicted that a rotating magnetized neutron starwould emit radio waves. But it was not until the 1967 Bell and Hewish4 discovery of radio pulsars that the existence of neutron st ...
Nuclear Physics Fundamentals and Application Prof. H.C Verma
... What we had seen? The results of these experiments is that if you plot this density charge density as a function of r, then you find that something like some constant value almost constant value. Then, it tapers off and for different nuclei; you have different slightly different values of this. But ...
... What we had seen? The results of these experiments is that if you plot this density charge density as a function of r, then you find that something like some constant value almost constant value. Then, it tapers off and for different nuclei; you have different slightly different values of this. But ...
Theoretical Nuclear Physics
... 11.5.1 Thomas expression for the width . . . . . . . . . 11.6 Homework problems . . . . . . . . . . . . . . . . . . . . ...
... 11.5.1 Thomas expression for the width . . . . . . . . . 11.6 Homework problems . . . . . . . . . . . . . . . . . . . . ...
Past papers - Wilson`s Physics
... An ultrasound investigation was used to identify a small volume of substance in a patient. It is suspected that this substance is either blood or muscle. During the ultrasound investigation, an ultrasound pulse of frequency of 3.5 × 106 Hz passed through soft tissue and then into the small volume of ...
... An ultrasound investigation was used to identify a small volume of substance in a patient. It is suspected that this substance is either blood or muscle. During the ultrasound investigation, an ultrasound pulse of frequency of 3.5 × 106 Hz passed through soft tissue and then into the small volume of ...
Slide 1
... • Particles decay spontaneously only when their combined products have less mass after decay than before. • The mass of a neutron is slightly greater than the total mass of a proton plus electron (and the antineutrino). • When a neutron decays, there is less mass. • Decay will not spontaneously occu ...
... • Particles decay spontaneously only when their combined products have less mass after decay than before. • The mass of a neutron is slightly greater than the total mass of a proton plus electron (and the antineutrino). • When a neutron decays, there is less mass. • Decay will not spontaneously occu ...
Option C Energy - Cambridge Resources for the IB Diploma
... The three main fossil fuels are coal, oil and natural gas. They are called fossil fuels because they are formed from things that were once alive and have been buried underground for millions of years. Coal is formed from the remains of plants and trees which fell into swamps millions of years ago. T ...
... The three main fossil fuels are coal, oil and natural gas. They are called fossil fuels because they are formed from things that were once alive and have been buried underground for millions of years. Coal is formed from the remains of plants and trees which fell into swamps millions of years ago. T ...
Reaction Energy
... sample, the hotter it feels. • For calculations in thermochemistry, the Celsius and Kelvin temperature scales are used. Celsius and Kelvin temperatures are related by the following equation. K = 273.15 + °C ...
... sample, the hotter it feels. • For calculations in thermochemistry, the Celsius and Kelvin temperature scales are used. Celsius and Kelvin temperatures are related by the following equation. K = 273.15 + °C ...
Energy and Chemistry
... One area where the calorie is used is in nutrition. Energy contents of foods are often expressed in calories. However, the calorie unit used for foods is actually the kilocalorie (kcal). Most foods indicate this by spelling the word with a capital C—Calorie. Figure 7.1 shows one example. So be caref ...
... One area where the calorie is used is in nutrition. Energy contents of foods are often expressed in calories. However, the calorie unit used for foods is actually the kilocalorie (kcal). Most foods indicate this by spelling the word with a capital C—Calorie. Figure 7.1 shows one example. So be caref ...
Unit 4, Lesson #3 - Patterson Science
... The value of Keq is determined experimentally. Chemists allow reactions to occur at stated temperatures, until the system no longer changes. At this point, they measure the amounts of both the reactants and products. Just as chemists monitor changes in pH, colour, gas pressure or conductivity of sol ...
... The value of Keq is determined experimentally. Chemists allow reactions to occur at stated temperatures, until the system no longer changes. At this point, they measure the amounts of both the reactants and products. Just as chemists monitor changes in pH, colour, gas pressure or conductivity of sol ...
Chapter 17 - Cengage Learning
... Le Châtelier’s principle helps us predict what happens to systems at equilibrium when conditions are changed. Le Châtelier’s principle states that when a system at equilibrium is changed, the system will shift its equilibrium position in order to reduce the change. The most common changes are change ...
... Le Châtelier’s principle helps us predict what happens to systems at equilibrium when conditions are changed. Le Châtelier’s principle states that when a system at equilibrium is changed, the system will shift its equilibrium position in order to reduce the change. The most common changes are change ...
- Institute of plasma physics
... e.g. Tore Supra : L//~100 m, D~10-5 m ~ 107 grid points in 1-D. Equilibrium is reached after several ion transit times =L///vTi ~ 109 particles and ~ 108 time steps Fully self consistent PIC simulations can only be attempted with greatly reduced SOL dimension (~cm) and unphysically high collis ...
... e.g. Tore Supra : L//~100 m, D~10-5 m ~ 107 grid points in 1-D. Equilibrium is reached after several ion transit times =L///vTi ~ 109 particles and ~ 108 time steps Fully self consistent PIC simulations can only be attempted with greatly reduced SOL dimension (~cm) and unphysically high collis ...
On neutral-beam injection counter to the plasma current
... is larger in absolute terms than the injected angular momentum. The injection of countercurrent momentum is supplemented by the loss of cocurrent momentum, leading to a larger effect than if there were no first-orbit losses. Second, if the loss occurs some distance away from the separatrix 共or ...
... is larger in absolute terms than the injected angular momentum. The injection of countercurrent momentum is supplemented by the loss of cocurrent momentum, leading to a larger effect than if there were no first-orbit losses. Second, if the loss occurs some distance away from the separatrix 共or ...
1. Which idea of John Dalton is no longer considered part of the
... End of Goal 2 Sample Items In compliance with federal law, including the provisions of Title IX of the Education Amendments of 1972, the Department of Public Instruction does not discriminate on the basis of race, sex, religion, color, national or ethnic origin, age, disability, or military service ...
... End of Goal 2 Sample Items In compliance with federal law, including the provisions of Title IX of the Education Amendments of 1972, the Department of Public Instruction does not discriminate on the basis of race, sex, religion, color, national or ethnic origin, age, disability, or military service ...
4. chemical kinetics
... If concentration is higher, the number of molecules per unit volume is, more and the number of active collisions increases. According to collision theory, rate of reaction increases with increase in the number of ...
... If concentration is higher, the number of molecules per unit volume is, more and the number of active collisions increases. According to collision theory, rate of reaction increases with increase in the number of ...
Equilibrium
... If you roll clay into a rope and begin to shake one end more and more vigorously, eventually it will break. If enough energy is applied to a molecule, the bonds holding it together can break. ...
... If you roll clay into a rope and begin to shake one end more and more vigorously, eventually it will break. If enough energy is applied to a molecule, the bonds holding it together can break. ...
Issue 10 - Free-Energy Devices
... registered neutron radiation during plasma electrolysis of water and reported that not only the nuclear process, but also the process of the electron capture by the free protons can be the source of this radiation. ...
... registered neutron radiation during plasma electrolysis of water and reported that not only the nuclear process, but also the process of the electron capture by the free protons can be the source of this radiation. ...
14th International Conference on Condensed Matter Nuclear Science Cold Fusion (ICCF-14)
... and then a plasma of increasing temperature, when it is near fully collapsed. And, the jet of material is like a beam impacting the surface of the foil. So, cavitation loading spans at least two of the major classes of loading, plasma and beam, both of which involve higher energies per particle than ...
... and then a plasma of increasing temperature, when it is near fully collapsed. And, the jet of material is like a beam impacting the surface of the foil. So, cavitation loading spans at least two of the major classes of loading, plasma and beam, both of which involve higher energies per particle than ...
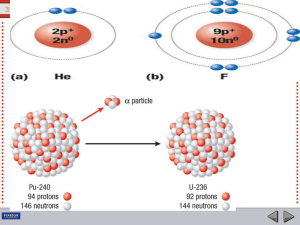
Nuclear fusion

In nuclear physics, nuclear fusion is a nuclear reaction in which two or more atomic nuclei come very close and then collide at a very high speed and join to form a new nucleus. During this process, matter is not conserved because some of the matter of the fusing nuclei is converted to photons (energy). Fusion is the process that powers active or ""main sequence"" stars.The fusion of two nuclei with lower masses than Iron-56 (which, along with Nickel-62, has the largest binding energy per nucleon) generally releases energy, while the fusion of nuclei heavier than iron absorbs energy. The opposite is true for the reverse process, nuclear fission. This means that fusion generally occurs for lighter elements only, and likewise, that fission normally occurs only for heavier elements. There are extreme astrophysical events that can lead to short periods of fusion with heavier nuclei. This is the process that gives rise to nucleosynthesis, the creation of the heavy elements during events such as supernova.Following the discovery of quantum tunneling by Friedrich Hund, in 1929 Robert Atkinson and Fritz Houtermans used the measured masses of light elements to predict that large amounts of energy could be released by fusing small nuclei. Building upon the nuclear transmutation experiments by Ernest Rutherford, carried out several years earlier, the laboratory fusion of hydrogen isotopes was first accomplished by Mark Oliphant in 1932. During the remainder of that decade the steps of the main cycle of nuclear fusion in stars were worked out by Hans Bethe. Research into fusion for military purposes began in the early 1940s as part of the Manhattan Project. Fusion was accomplished in 1951 with the Greenhouse Item nuclear test. Nuclear fusion on a large scale in an explosion was first carried out on November 1, 1952, in the Ivy Mike hydrogen bomb test.Research into developing controlled thermonuclear fusion for civil purposes also began in earnest in the 1950s, and it continues to this day. The present article is about the theory of fusion. For details of the quest for controlled fusion and its history, see the article Fusion power.