Lecture 3 - TAMU Chemistry
... Color: Electronic transitions due to energy levels whose gaps are in the visible range of the electromagnetic spectrum. Magnetism: partially filled orbitals, unpaired electrons. high spin: maximum no. of d electrons unpaired low spin: electrons paired up in d orbitals. ...
... Color: Electronic transitions due to energy levels whose gaps are in the visible range of the electromagnetic spectrum. Magnetism: partially filled orbitals, unpaired electrons. high spin: maximum no. of d electrons unpaired low spin: electrons paired up in d orbitals. ...
Accelerator Experiments and Theoretical Models for the Electron Screening Effect in
... The experiments have been carried out at an accelerator optimized for low energy beams. Fig. 1 illustrates the principal set-up and the data acquisition system. The accelerator consists of a radio frequency ion source, an acceleration line powered by a highly stabilized 60 kV supply and subsequent e ...
... The experiments have been carried out at an accelerator optimized for low energy beams. Fig. 1 illustrates the principal set-up and the data acquisition system. The accelerator consists of a radio frequency ion source, an acceleration line powered by a highly stabilized 60 kV supply and subsequent e ...
New interpretations of XPS spectra of nickel metal and oxides
... level XPS assignments of features in nickel metal and oxides. Beginning with nickel metal ELS, they suggest that more intense features at 6.0 and 9.5 eV are due to surface and bulk plasmon losses respectively with weaker intra- and inter-band transitions at 3.7 and 7.1 eV. In XPS spectra, the well-k ...
... level XPS assignments of features in nickel metal and oxides. Beginning with nickel metal ELS, they suggest that more intense features at 6.0 and 9.5 eV are due to surface and bulk plasmon losses respectively with weaker intra- and inter-band transitions at 3.7 and 7.1 eV. In XPS spectra, the well-k ...
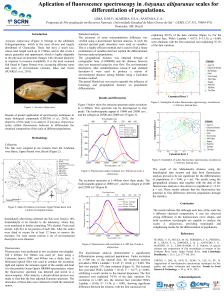
Astyanax altiparanae - Sistema de Eventos
... The discriminant analysis demonstrated a significantly differentiation among analyzed populations. Under excitation at =360 nm, in the internal face, the statistical analysis provided a Wilk’s Lambda = 0.145; F= 10.66; p < 0.001. The first root explain 71% data variation (Figure 5). The external fa ...
... The discriminant analysis demonstrated a significantly differentiation among analyzed populations. Under excitation at =360 nm, in the internal face, the statistical analysis provided a Wilk’s Lambda = 0.145; F= 10.66; p < 0.001. The first root explain 71% data variation (Figure 5). The external fa ...
Chapter 12 - NCERT books
... molecule with its neighbours. In contrast, light emitted from rarefied gases heated in a flame, or excited electrically in a glow tube such as the familiar neon sign or mercury vapour light has only certain discrete wavelengths. The spectrum appears as a series of bright lines. In such gases, the av ...
... molecule with its neighbours. In contrast, light emitted from rarefied gases heated in a flame, or excited electrically in a glow tube such as the familiar neon sign or mercury vapour light has only certain discrete wavelengths. The spectrum appears as a series of bright lines. In such gases, the av ...
Coulomb blockade in Quantum Dots
... leverarm is well defined. However in quantum dots where regions of a 2DEG are used as so-called in-plane gates the lever arm might change slowly as a function of the voltage applied to the gate making the relation between the chemical potential on the dot and gate voltage non-linear. Transport throu ...
... leverarm is well defined. However in quantum dots where regions of a 2DEG are used as so-called in-plane gates the lever arm might change slowly as a function of the voltage applied to the gate making the relation between the chemical potential on the dot and gate voltage non-linear. Transport throu ...
Ionization Energies of the Elements Manifest Natural Harmonies of
... Johnson [jon] described the surprising patterns because of which ionization data show that the Bohr atomic model is untenable: Examples: NEUTRAL helium has two (electrons), but if the helium is singly ionized, it also has one electron. NEUTRAL lithium has three electrons, but doubly-ionized lithium ...
... Johnson [jon] described the surprising patterns because of which ionization data show that the Bohr atomic model is untenable: Examples: NEUTRAL helium has two (electrons), but if the helium is singly ionized, it also has one electron. NEUTRAL lithium has three electrons, but doubly-ionized lithium ...
Ionization methods - 2-CI - Florida International University
... depends on the relative affinities of the conjugate base of the reactant ion (CH 4, NH3 and so on) and the compound M. • Decrease in PA (proton affinity) of the conjugate base (or increase in acidity of the reactant gas ion) causes increase in fragmentation because more energy is transferred to them ...
... depends on the relative affinities of the conjugate base of the reactant ion (CH 4, NH3 and so on) and the compound M. • Decrease in PA (proton affinity) of the conjugate base (or increase in acidity of the reactant gas ion) causes increase in fragmentation because more energy is transferred to them ...
atoms - eVirtualGuru
... molecule with its neighbours. In contrast, light emitted from rarefied gases heated in a flame, or excited electrically in a glow tube such as the familiar neon sign or mercury vapour light has only certain discrete wavelengths. The spectrum appears as a series of bright lines. In such gases, the av ...
... molecule with its neighbours. In contrast, light emitted from rarefied gases heated in a flame, or excited electrically in a glow tube such as the familiar neon sign or mercury vapour light has only certain discrete wavelengths. The spectrum appears as a series of bright lines. In such gases, the av ...
down - Display Materials Lab.
... Inelastic mean free path 2 atomic layer when 40eV 10 atomic layer when 1000eV MS310 Quantum Physical Chemistry ...
... Inelastic mean free path 2 atomic layer when 40eV 10 atomic layer when 1000eV MS310 Quantum Physical Chemistry ...
Drawing Electron
... In carbon dioxide, CO2, octets are achieved by sharing two pairs of electrons between atoms; this is called a double bond. ...
... In carbon dioxide, CO2, octets are achieved by sharing two pairs of electrons between atoms; this is called a double bond. ...
Chapter 5: Electrons in Atoms
... After discovering three subatomic particles in the early 1900s, scientists continued their quest to understand atomic structure and the arrangement of electrons within atoms. Rutherford proposed that all of an atom’s positive charge and virtually all of its mass are concentrated in a nucleus that is ...
... After discovering three subatomic particles in the early 1900s, scientists continued their quest to understand atomic structure and the arrangement of electrons within atoms. Rutherford proposed that all of an atom’s positive charge and virtually all of its mass are concentrated in a nucleus that is ...
LONG JOURNEY INTO TUNNELING
... In the Kronig-Penney model of a one-dimensional crystal which consists of a series of equally-spaced potential barriers, it is well known that allowed bands of perfect transmission are separated by forbidden bands of attenuation. These one-dimensional mathematical problems can often be elegantly tre ...
... In the Kronig-Penney model of a one-dimensional crystal which consists of a series of equally-spaced potential barriers, it is well known that allowed bands of perfect transmission are separated by forbidden bands of attenuation. These one-dimensional mathematical problems can often be elegantly tre ...
Chiral specific electron vortex beam spectroscopy
... We can now address the physical meaning of the double summation over p and p0 in Eq. (13) and the implications of this for the chiral-specific spectroscopy of atoms located away from the vortex beam axis. The off-axis case is illustrated in Fig. (1)(b). It can be seen that the features uncovered abo ...
... We can now address the physical meaning of the double summation over p and p0 in Eq. (13) and the implications of this for the chiral-specific spectroscopy of atoms located away from the vortex beam axis. The off-axis case is illustrated in Fig. (1)(b). It can be seen that the features uncovered abo ...
solutions - The University of Sydney
... When the intensity of light is doubled the maximum KE of electrons does not change. However, the number of electrons ejected per unit time will double. (1 mark) (c) In the classical picture the light is a wave which delivers energy continuously to electrons in the metal. The energy in the wave is pr ...
... When the intensity of light is doubled the maximum KE of electrons does not change. However, the number of electrons ejected per unit time will double. (1 mark) (c) In the classical picture the light is a wave which delivers energy continuously to electrons in the metal. The energy in the wave is pr ...
continuous vs discrete processes: the
... transitions from one level to another in accordance with the probabilities of a Markov transition-matrix. Many investigators on the other hand, who are of the opinion that quantum mechanics precludes transitions of this kind having any ontological status, interpret the transitions as measurement-ind ...
... transitions from one level to another in accordance with the probabilities of a Markov transition-matrix. Many investigators on the other hand, who are of the opinion that quantum mechanics precludes transitions of this kind having any ontological status, interpret the transitions as measurement-ind ...
PPT - jick.net
... Can μ +e − HF interactions be observed directly if Coulomb binding is ineffective? Something else must localize the electron near the muon! What could do that? What if free electrons are around to spin-exchange with the localized electron? Something must prevent electron spin-exchange! What could do ...
... Can μ +e − HF interactions be observed directly if Coulomb binding is ineffective? Something else must localize the electron near the muon! What could do that? What if free electrons are around to spin-exchange with the localized electron? Something must prevent electron spin-exchange! What could do ...
Spectroscopic Study of Argon DC Glow Discharge
... where I, λ, g, and f are the total intensity, wavelength, statistical weight, and absorption oscillator strength, respectively, of one line and E is its excitation energy. The corresponding quantities for the other line are I % , λ% , g % , f % , and E % . The values of the above parameters have bee ...
... where I, λ, g, and f are the total intensity, wavelength, statistical weight, and absorption oscillator strength, respectively, of one line and E is its excitation energy. The corresponding quantities for the other line are I % , λ% , g % , f % , and E % . The values of the above parameters have bee ...
10/18/11 - Note: Once it is downloaded, click SET
... What’s involved? Periodic table, electron, atomic number Electrons are arranged in orbitals around the nucleus Things to know: -Hund’s Rule, Aufbau Principle, Pauli’s Exclusion Principle -Electron Dot- shows how many valence electrons it has. -SPDF (orbitals) S- 1- up to 2 electrons P- 3- up to 6 el ...
... What’s involved? Periodic table, electron, atomic number Electrons are arranged in orbitals around the nucleus Things to know: -Hund’s Rule, Aufbau Principle, Pauli’s Exclusion Principle -Electron Dot- shows how many valence electrons it has. -SPDF (orbitals) S- 1- up to 2 electrons P- 3- up to 6 el ...
1 - GENCHEM
... 2) Procedure: During the quiz hour, you can take your mid-term paper scored. If you have any claims on it, you can submit a claim paper with your opinion. After writing your opinions on any paper you can get easily, attach it with a stapler to your mid-term paper scored (Please, write your name, pro ...
... 2) Procedure: During the quiz hour, you can take your mid-term paper scored. If you have any claims on it, you can submit a claim paper with your opinion. After writing your opinions on any paper you can get easily, attach it with a stapler to your mid-term paper scored (Please, write your name, pro ...
Electrons in Atoms
... The quantum concept • Prior experience had led scientists to believe that energy could be absorbed and emitted in continually varying quantities, with no minimum limit to the amount. • For example, think about heating a cup of water in a microwave oven. • It seems that you can add any amount of ther ...
... The quantum concept • Prior experience had led scientists to believe that energy could be absorbed and emitted in continually varying quantities, with no minimum limit to the amount. • For example, think about heating a cup of water in a microwave oven. • It seems that you can add any amount of ther ...
Slide 1
... The quantum concept • Prior experience had led scientists to believe that energy could be absorbed and emitted in continually varying quantities, with no minimum limit to the amount. • For example, think about heating a cup of water in a microwave oven. • It seems that you can add any amount of ther ...
... The quantum concept • Prior experience had led scientists to believe that energy could be absorbed and emitted in continually varying quantities, with no minimum limit to the amount. • For example, think about heating a cup of water in a microwave oven. • It seems that you can add any amount of ther ...
Auger electron spectroscopy
.jpg?width=300)
Auger electron spectroscopy (AES; pronounced [oʒe] in French) is a common analytical technique used specifically in the study of surfaces and, more generally, in the area of materials science. Underlying the spectroscopic technique is the Auger effect, as it has come to be called, which is based on the analysis of energetic electrons emitted from an excited atom after a series of internal relaxation events. The Auger effect was discovered independently by both Lise Meitner and Pierre Auger in the 1920s. Though the discovery was made by Meitner and initially reported in the journal Zeitschrift für Physik in 1922, Auger is credited with the discovery in most of the scientific community. Until the early 1950s Auger transitions were considered nuisance effects by spectroscopists, not containing much relevant material information, but studied so as to explain anomalies in x-ray spectroscopy data. Since 1953 however, AES has become a practical and straightforward characterization technique for probing chemical and compositional surface environments and has found applications in metallurgy, gas-phase chemistry, and throughout the microelectronics industry.