lecture10
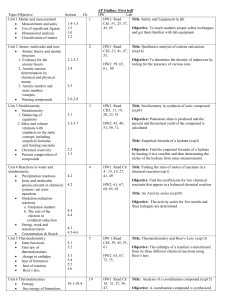
... oxidize Fe+ to Fe2+ than it is to oxidize Cu to Cu2+. The former involves the transfer of one electron for every iron, the later requires the transfer of 2 electrons for every Cu atom. Notice also that there is a minus sign in the equation. This is because the conventions for charge movement (curren ...
... oxidize Fe+ to Fe2+ than it is to oxidize Cu to Cu2+. The former involves the transfer of one electron for every iron, the later requires the transfer of 2 electrons for every Cu atom. Notice also that there is a minus sign in the equation. This is because the conventions for charge movement (curren ...
Working With Chemical Reactions
... Yields 2 ammonia, Carbon dioxide, and water. Heat is your clue that this is decomposition. Remove ammonia, and also carbonates decompose to carbon dioxide, the remaining elements form water. ...
... Yields 2 ammonia, Carbon dioxide, and water. Heat is your clue that this is decomposition. Remove ammonia, and also carbonates decompose to carbon dioxide, the remaining elements form water. ...
REDOX PowerPoint - Southmoreland School District
... it is bonded to metals in binary compounds. In these cases, its oxidation number is ___. (LiAlH4) 5. Group IA metals are ___, IIA metals are ___ and fluorine is always ___. 6. The sum of the oxidation numbers of all the atoms in a molecule or ion is equal to ________________________. ...
... it is bonded to metals in binary compounds. In these cases, its oxidation number is ___. (LiAlH4) 5. Group IA metals are ___, IIA metals are ___ and fluorine is always ___. 6. The sum of the oxidation numbers of all the atoms in a molecule or ion is equal to ________________________. ...
Selective Ethanol Synthesis from Carbon Dioxide
... (CO + H2)is a well known process (1-3). The dissociationenergy for carbon monoxide is large (107 1.8 kJ mol-I). It chemisorbs on metal surfaces via the carbon atom through the 5 0 orbital, which is the HOMO (highest occupied molecular orbital). When adsorbed CO, CO(ads), is completely hydrogenated, ...
... (CO + H2)is a well known process (1-3). The dissociationenergy for carbon monoxide is large (107 1.8 kJ mol-I). It chemisorbs on metal surfaces via the carbon atom through the 5 0 orbital, which is the HOMO (highest occupied molecular orbital). When adsorbed CO, CO(ads), is completely hydrogenated, ...
Heterogeneous catalysis (I)
... Far more convenient is the use of the concept of turnover frequency or turnover number. The turnover frequency (often designated TOF) is simply the number of times n that the overall catalytic reaction in question takes place per catalytic site per unit time for a fixed set of reaction conditions (t ...
... Far more convenient is the use of the concept of turnover frequency or turnover number. The turnover frequency (often designated TOF) is simply the number of times n that the overall catalytic reaction in question takes place per catalytic site per unit time for a fixed set of reaction conditions (t ...
Basso08_preprint - University of Strathclyde
... the different components and on the nature of the liquid phase used. Thus, there are mainly solid systems or dilute suspensions in a large liquid phase in which a product can precipitate because its solubility in the solvent used is extremely low. Although substrates are usually largely undissolved ...
... the different components and on the nature of the liquid phase used. Thus, there are mainly solid systems or dilute suspensions in a large liquid phase in which a product can precipitate because its solubility in the solvent used is extremely low. Although substrates are usually largely undissolved ...
Analytical animation-definitions electrochem3
... 1. The mass of the Cu electrode increases and the concentration of Cu2+ decreases 2. The mass of the Zinc electrode decreases and the concentration of Zn2+ increases Consider this: During a redox reaction, one species is reduced, and one species is oxidized. The one that is reduced gains electrons ( ...
... 1. The mass of the Cu electrode increases and the concentration of Cu2+ decreases 2. The mass of the Zinc electrode decreases and the concentration of Zn2+ increases Consider this: During a redox reaction, one species is reduced, and one species is oxidized. The one that is reduced gains electrons ( ...
Document
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
Kinetics of the fading of phenolphthalein in alkaline solution
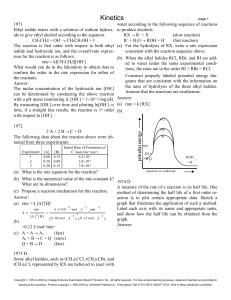
... to Beer's law, the spectral absorbance of the solution is directly proportional to [P2-1, or [P2-] = constant X (ahsorhance), so ln[P2-] = In (constant) In (ahsorhance). Therefore, a plot of in (absorbance) versus time should also be a straight line with slope of - k l , if the reaction is pseudo fi ...
... to Beer's law, the spectral absorbance of the solution is directly proportional to [P2-1, or [P2-] = constant X (ahsorhance), so ln[P2-] = In (constant) In (ahsorhance). Therefore, a plot of in (absorbance) versus time should also be a straight line with slope of - k l , if the reaction is pseudo fi ...
chm5423chapter5notes..
... molecules decompose by photodissociation. Other molecules, particularly polar molecules, can be physically removed from the troposphere by rainout, washout, and deposition. Molecules can also be removed by chemical reaction. Finally, molecules with no significant removal processes in the troposphere ...
... molecules decompose by photodissociation. Other molecules, particularly polar molecules, can be physically removed from the troposphere by rainout, washout, and deposition. Molecules can also be removed by chemical reaction. Finally, molecules with no significant removal processes in the troposphere ...