Recent developments in metal-catalyzed living radical
... RuCl2(PPh3)3 but seems less stable. In a way similar to ruthenium, the Cp- or Cp*-based iron(II) complexes showed higher activities and versatilities.56–59 We then changed the oxidation state of iron from +2 to +1 by applying the dinuclear Cp- or Cp*-based iron(I) complexes [Fe2Cp2(CO)4 or Fe2Cp*2(C ...
... RuCl2(PPh3)3 but seems less stable. In a way similar to ruthenium, the Cp- or Cp*-based iron(II) complexes showed higher activities and versatilities.56–59 We then changed the oxidation state of iron from +2 to +1 by applying the dinuclear Cp- or Cp*-based iron(I) complexes [Fe2Cp2(CO)4 or Fe2Cp*2(C ...
Document
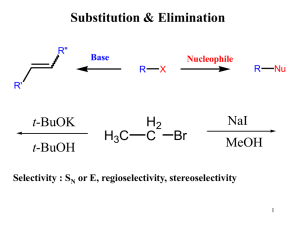
... • With base, the nucleophile is ¯OH, and the mechanism follows the usual two steps: nucleophilic attack followed by protonation. • The reaction rate increases in the presence of base because the base converts H2O into ¯OH, a stronger ...
... • With base, the nucleophile is ¯OH, and the mechanism follows the usual two steps: nucleophilic attack followed by protonation. • The reaction rate increases in the presence of base because the base converts H2O into ¯OH, a stronger ...
Iodomethylzinc_iodid.. - Groupe Charette
... with a mixture of diethylzinc and diiodomethane generated the corresponding cyclopropanols in good yield (eq 24).38 The yields obtained were much superior to those observed with boron or aluminum enolates; however, a mixture of the two diastereomeric cyclopropanes is usually observed. I ...
... with a mixture of diethylzinc and diiodomethane generated the corresponding cyclopropanols in good yield (eq 24).38 The yields obtained were much superior to those observed with boron or aluminum enolates; however, a mixture of the two diastereomeric cyclopropanes is usually observed. I ...
C−C, C−O, C−N Bond Formation on sp2 Carbon by Pd(II)
... common ligands for late-transition metals, such as phosphines, are susceptible to oxidative decomposition. In contrast, the use of O2 generally requires a ligand for efficient catalysis, which introduces the possibility of making the ...
... common ligands for late-transition metals, such as phosphines, are susceptible to oxidative decomposition. In contrast, the use of O2 generally requires a ligand for efficient catalysis, which introduces the possibility of making the ...
On The catalytic Hydrogenation of Co2 and Carboxylic acid esters
... subsequent hydrolysis of methyl formate (Scheme 1.1).7 In addition to being a commodity chemical, FA has recently been proposed as one of the potential green fuels of the future,8 ...
... subsequent hydrolysis of methyl formate (Scheme 1.1).7 In addition to being a commodity chemical, FA has recently been proposed as one of the potential green fuels of the future,8 ...
- Article One Partners
... hydrolysis. The amino function can be alkylated according to generally-accepted methods. Methylation can, for example, be effected by reaction with chloromethyl formate and subsequent reduction. The ethyl group can be introduced by reduction of the respective acetamide. In accordance with U.S. Pat. ...
... hydrolysis. The amino function can be alkylated according to generally-accepted methods. Methylation can, for example, be effected by reaction with chloromethyl formate and subsequent reduction. The ethyl group can be introduced by reduction of the respective acetamide. In accordance with U.S. Pat. ...
Nucleophilic Acyl Substitution
... the better its leaving ability. Recall from Section 10.3 that weak bases are good leaving groups because weak bases do not share their electrons as well as strong bases do. The pKa values of the conjugate acids of the leaving groups of various carbonyl compounds are listed in Table 17.1. Notice that ...
... the better its leaving ability. Recall from Section 10.3 that weak bases are good leaving groups because weak bases do not share their electrons as well as strong bases do. The pKa values of the conjugate acids of the leaving groups of various carbonyl compounds are listed in Table 17.1. Notice that ...
Part I Carbohydrate Auxiliaries - Wiley-VCH
... The asymmetric Strecker synthesis using carbohydrate auxiliaries has also been studied in some detail by Zhang using a d-glucose-based chiral template [7]. In continuation of Kunz’s studies a general protocol for the asymmetric synthesis of α,β-diamino acids involving enantiomerically pure α-amino a ...
... The asymmetric Strecker synthesis using carbohydrate auxiliaries has also been studied in some detail by Zhang using a d-glucose-based chiral template [7]. In continuation of Kunz’s studies a general protocol for the asymmetric synthesis of α,β-diamino acids involving enantiomerically pure α-amino a ...
Proofs to - Research Explorer
... system commenced with an investigation of the reaction of MeOH with the unsubstituted vinylidene [Fe(=C=CH2)(dppe)Cp]+ (2). Several protocols for the synthesis of 2 have been developed [18, 21, 22], including the reaction of [FeCl(dppe)Cp] with HC≡CSiMe3 in an alcohol solvent [22]. In the initial in ...
... system commenced with an investigation of the reaction of MeOH with the unsubstituted vinylidene [Fe(=C=CH2)(dppe)Cp]+ (2). Several protocols for the synthesis of 2 have been developed [18, 21, 22], including the reaction of [FeCl(dppe)Cp] with HC≡CSiMe3 in an alcohol solvent [22]. In the initial in ...
Document
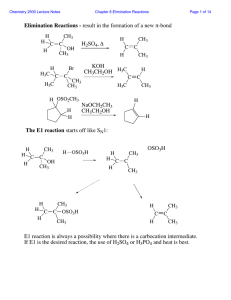
... Tosylate—Another Good Leaving Group • Because alkyl tosylates have good leaving groups, they undergo both nucleophilic substitution and elimination, exactly as alkyl halides do. ...
... Tosylate—Another Good Leaving Group • Because alkyl tosylates have good leaving groups, they undergo both nucleophilic substitution and elimination, exactly as alkyl halides do. ...
New Stereoselective Approaches to Highly Substituted
... aldol reaction, originally developed by Kazmaier, as a highly diastereoselective method for the synthesis o f the cyclisadon precursors. Both acetylenic and a,f3-unsaturated aldehydes have been successfully utilised in this reaction to afford a variety o f 0 -hydroxy-a-amino ester precursors. This p ...
... aldol reaction, originally developed by Kazmaier, as a highly diastereoselective method for the synthesis o f the cyclisadon precursors. Both acetylenic and a,f3-unsaturated aldehydes have been successfully utilised in this reaction to afford a variety o f 0 -hydroxy-a-amino ester precursors. This p ...
Ethers, Epoxides and Sulfides
... Formation of halohydrin ¾ By treating alkenes with aqueous solution of halogens. ¾ Bromine water and chlorine water add across double bonds with Markovnikov orientation. ...
... Formation of halohydrin ¾ By treating alkenes with aqueous solution of halogens. ¾ Bromine water and chlorine water add across double bonds with Markovnikov orientation. ...
haloalkanes and arenes
... has more s-character than sp3 hybridized carbon atom. Therefore, the former is more electronegative than the latter. Therefore, the density of electrons of C−Cl bond near the Clatom is less in chlorobenzene than in cydohexyl chloride. Moreover, the −R effect of the benzene ring of chlorobenzene decr ...
... has more s-character than sp3 hybridized carbon atom. Therefore, the former is more electronegative than the latter. Therefore, the density of electrons of C−Cl bond near the Clatom is less in chlorobenzene than in cydohexyl chloride. Moreover, the −R effect of the benzene ring of chlorobenzene decr ...
Iodine and Lipase Based Green Oxidation Technology
... the carboxylic acid with hydrogen peroxide (see chapter 5 for more details). 1.2 Alcohol and alkene oxidation In this paragraph two classes of reactions will be outlined with in more detail: alcohol oxidation and alkene oxidation. A variety of examples will be listed that are representative of th ...
... the carboxylic acid with hydrogen peroxide (see chapter 5 for more details). 1.2 Alcohol and alkene oxidation In this paragraph two classes of reactions will be outlined with in more detail: alcohol oxidation and alkene oxidation. A variety of examples will be listed that are representative of th ...
Efficient hydrogenation of organic carbonates, carbamates and
... groups, has captured much attention during the past four decades1–3, mainly due to its synthetic significance as an environmentally benign approach to fundamental synthetic building blocks such as alcohols and amines. Much progress has been made in the hydrogenation of ketones and aldehydes and, more ...
... groups, has captured much attention during the past four decades1–3, mainly due to its synthetic significance as an environmentally benign approach to fundamental synthetic building blocks such as alcohols and amines. Much progress has been made in the hydrogenation of ketones and aldehydes and, more ...
Chem E2b - Organic Chemistry II What is Organic Chemistry?
... Hyperconjugation is less stabilizing for a radical because the one electron in the antibonding MO has a destabilizing effect. (recall He2+ with a similar 3-electron system) ...
... Hyperconjugation is less stabilizing for a radical because the one electron in the antibonding MO has a destabilizing effect. (recall He2+ with a similar 3-electron system) ...
Excited State Reactions of Carbonyl Compounds
... are polarized in such a way that the carbonyl carbon is positively polarized. Most reactions of the carbonyl chromophore in the ground state can be understood on the basis of this simple model. The non-bonding lone pair present on the oxygen is nucleophilic, and many electrophilic reactions with the ...
... are polarized in such a way that the carbonyl carbon is positively polarized. Most reactions of the carbonyl chromophore in the ground state can be understood on the basis of this simple model. The non-bonding lone pair present on the oxygen is nucleophilic, and many electrophilic reactions with the ...
A review of new developments in the Friedel–Crafts - Beilstein
... the need for more environmentally and economically benign processes, the development of FC reactions using only catalytic amounts of a metal or acid catalyst would be highly desirable. In addition, the substitution of the alkyl chlorides by other, less toxic, alkylating reagents such as alcohols wou ...
... the need for more environmentally and economically benign processes, the development of FC reactions using only catalytic amounts of a metal or acid catalyst would be highly desirable. In addition, the substitution of the alkyl chlorides by other, less toxic, alkylating reagents such as alcohols wou ...
Diastereoselective Allylation of Carbonyl Compounds and Imines:
... silanes must be carried out in the presence of an activating reagent. A diastereoselective synthesis of 2,3,4-trisubstituted tetrahydrofurans has been reported by Cox and co-workers. In these processes, an allyl silane and a carbonyl group of an aldehyde are involved in an intramolecular allylation ...
... silanes must be carried out in the presence of an activating reagent. A diastereoselective synthesis of 2,3,4-trisubstituted tetrahydrofurans has been reported by Cox and co-workers. In these processes, an allyl silane and a carbonyl group of an aldehyde are involved in an intramolecular allylation ...
Alcohols, Phenols, and Thiols
... Alcohols are comparable in acidity to water, but phenols are much more acidic. This increased acidity is due to charge delocalization (resonance) in phenoxide ions. Electron-withdrawing groups, such as F and NO2, increase acidity, through either an inductive or a resonance effect, or both. Alkoxid ...
... Alcohols are comparable in acidity to water, but phenols are much more acidic. This increased acidity is due to charge delocalization (resonance) in phenoxide ions. Electron-withdrawing groups, such as F and NO2, increase acidity, through either an inductive or a resonance effect, or both. Alkoxid ...
- University of Bath Opus
... borrowing hydrogen methodology with masked ammonia sources. This unique approach allows selective access to primary amines without the over alkylation seen when using other ammonia sources. Furthermore, the borrowing hydrogen reaction can be coupled with an in-situ palladium catalysed hydrogenolysis ...
... borrowing hydrogen methodology with masked ammonia sources. This unique approach allows selective access to primary amines without the over alkylation seen when using other ammonia sources. Furthermore, the borrowing hydrogen reaction can be coupled with an in-situ palladium catalysed hydrogenolysis ...
Derivatization reactions for the determination of amines by gas
... the GC of free amines generally has some inherent problems related to the difficulty in handling low-molecular-mass amines because of their high water solubility and high volatility. Therefore, these amines are difficult to extract from water and are not easily chromatographed owing to their polarit ...
... the GC of free amines generally has some inherent problems related to the difficulty in handling low-molecular-mass amines because of their high water solubility and high volatility. Therefore, these amines are difficult to extract from water and are not easily chromatographed owing to their polarit ...
Chapter - FIU Faculty Websites
... Aldehydes and Ketones—Acetals as Protecting Groups • Acetals are valuable protecting groups for aldehydes and ketones: • Suppose we wish to selectively reduce the ester group in compound A to an alcohol to give compound B, leaving the ketone untouched. • Because ketones are more readily reduced than ...
... Aldehydes and Ketones—Acetals as Protecting Groups • Acetals are valuable protecting groups for aldehydes and ketones: • Suppose we wish to selectively reduce the ester group in compound A to an alcohol to give compound B, leaving the ketone untouched. • Because ketones are more readily reduced than ...
Hofmann–Löffler reaction

The Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is an organic reaction in which a cyclic amine 2 (pyrrolidine or, in some cases, piperidine) is generated by thermal or photochemical decomposition of N-halogenated amine 1 in the presence of a strong acid (concentrated sulfuric acid or concentrated CF3CO2H). The Hofmann–Löffler–Freytag reaction proceeds via an intramolecular hydrogen atom transfer to a nitrogen-centered radical and is an example of a remote intramolecular free radical C–H functionalization.