couverture these PRES Toulouse M ESCARCEGA 2011
... the formation of new bonds. Syngas conversion, hydrogenation and oxidation processes are by far the most important industrial applications. Catalyst synthesis technology is applied to the manufacture of high surface area metal species, including metallic nanoparticles (which will be further discusse ...
... the formation of new bonds. Syngas conversion, hydrogenation and oxidation processes are by far the most important industrial applications. Catalyst synthesis technology is applied to the manufacture of high surface area metal species, including metallic nanoparticles (which will be further discusse ...
Brønsted Acidity in Metal−Organic Frameworks
... they are obtained from a mixture of the guest molecules and the molecular building units needed for MOF synthesis. The guest molecules can be acidified after their inclusion to further increase the amount or strength of their Brønsted acidity. This approach enlarges the scope of Brønsted acidic guest ...
... they are obtained from a mixture of the guest molecules and the molecular building units needed for MOF synthesis. The guest molecules can be acidified after their inclusion to further increase the amount or strength of their Brønsted acidity. This approach enlarges the scope of Brønsted acidic guest ...
sample
... E) None of the above forms remains constant. Ans: D Difficulty: H 7. The most significant contribution to modern science made by alchemists was A) their fundamental work in the transmutation of the elements. B) their widespread acceptance of observation and experimentation. C) their systematic metho ...
... E) None of the above forms remains constant. Ans: D Difficulty: H 7. The most significant contribution to modern science made by alchemists was A) their fundamental work in the transmutation of the elements. B) their widespread acceptance of observation and experimentation. C) their systematic metho ...
Chemistry In action
... This book was typeset in 10/12 Times New Roman at cMPreparé and printed and bound by R. R. Donnelley/Jefferson City. The cover was printed by R. R. Donnelley/Jefferson City. The paper in this book was manufactured by a mill whose forest management programs include sustained yield—harvesting of its t ...
... This book was typeset in 10/12 Times New Roman at cMPreparé and printed and bound by R. R. Donnelley/Jefferson City. The cover was printed by R. R. Donnelley/Jefferson City. The paper in this book was manufactured by a mill whose forest management programs include sustained yield—harvesting of its t ...
enjoy chemistry
... Ans:Halogens are coloured. This is due to absorption of radiations in visible region which results in the excitation of outer electrons to higher energy level. By absorbing different quanta of radiation, they display different colours. For example, F2, has yellow, Cl2 , greenish yellow, Br2, red and ...
... Ans:Halogens are coloured. This is due to absorption of radiations in visible region which results in the excitation of outer electrons to higher energy level. By absorbing different quanta of radiation, they display different colours. For example, F2, has yellow, Cl2 , greenish yellow, Br2, red and ...
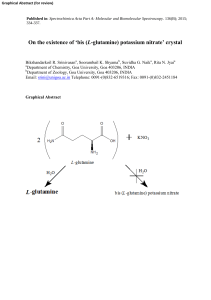
`bis (L-glutamine) potassium nitrate` crystal
... name for example bis (L-glutamine) potassium nitrate [11], abbreviated by a strange code. L-glutamine is one of the twenty naturally occurring amino acids and is an amide of Lglutamic acid. The compound monosodium glutamate, which contains sodium and the monoanion of L-glutamic acid, is present in t ...
... name for example bis (L-glutamine) potassium nitrate [11], abbreviated by a strange code. L-glutamine is one of the twenty naturally occurring amino acids and is an amide of Lglutamic acid. The compound monosodium glutamate, which contains sodium and the monoanion of L-glutamic acid, is present in t ...
COMPETITION PTOBLEMS 1
... Preface This publication contains the competition problems from the first twenty International Chemistry Olympiads (ICHO) organized in the years 1968 – 1988. It has been published by the ICHO International Information Centre in Bratislava (Slovakia) on the occasion of the 40th anniversary of this i ...
... Preface This publication contains the competition problems from the first twenty International Chemistry Olympiads (ICHO) organized in the years 1968 – 1988. It has been published by the ICHO International Information Centre in Bratislava (Slovakia) on the occasion of the 40th anniversary of this i ...
4420 Natural History of the Vertebrates. (3-3) Environmental
... 4420 Natural History of the Vertebrates. (3-3) Environmental relationships and natural history of vertebrates. Emphasis is upon taxonomy, speciation and biotic provinces. The laboratory will include field trips for the study and collection of animals in their natural habitats. Students will assemble ...
... 4420 Natural History of the Vertebrates. (3-3) Environmental relationships and natural history of vertebrates. Emphasis is upon taxonomy, speciation and biotic provinces. The laboratory will include field trips for the study and collection of animals in their natural habitats. Students will assemble ...
Ans:- (i) Gluconic acid - Kendriya Vidyalaya No.2, Kribhco, Surat
... Ans-.a) one b) two c) four Q-3.What type of semiconductors are when Si doped with – a) Phosphorous and b) Gallium. Ans. a) n-type and b) p-type Q-4. What is the coordination no. of an octahedral void and tetrahedral void? A-. Six(6) and Four(4) Q-5. Which of the two will show the Schottky defect whe ...
... Ans-.a) one b) two c) four Q-3.What type of semiconductors are when Si doped with – a) Phosphorous and b) Gallium. Ans. a) n-type and b) p-type Q-4. What is the coordination no. of an octahedral void and tetrahedral void? A-. Six(6) and Four(4) Q-5. Which of the two will show the Schottky defect whe ...
Chemical Redox Agents for Organometallic
... solutions, the usual media for organometallic ET reactions. It is hoped that the information in this review will also be useful to those interested in ET reactions of organic and inorganic systems, which have more often involved the use of labile metal ions such as M(III), M ) Fe, Tl1 (but see ref 2 ...
... solutions, the usual media for organometallic ET reactions. It is hoped that the information in this review will also be useful to those interested in ET reactions of organic and inorganic systems, which have more often involved the use of labile metal ions such as M(III), M ) Fe, Tl1 (but see ref 2 ...
Chemistry Science Notebook: Student Edition
... Third, you will notice that vocabulary is introduced and practiced throughout the Science Notebook. When students know the meaning of the words used to discuss information, they are able to understand that information better. Also, students are more likely to be successful in school when they have v ...
... Third, you will notice that vocabulary is introduced and practiced throughout the Science Notebook. When students know the meaning of the words used to discuss information, they are able to understand that information better. Also, students are more likely to be successful in school when they have v ...
08 Redox Reactions
... In the direct redox reaction, the transferance of electrons is limited to very small distances and therefore, no useful electrical work could be obtained. In these reactions, chemical energy appears as heat. If the transferance of electrons from zinc to copper ions is allowed to occur through some m ...
... In the direct redox reaction, the transferance of electrons is limited to very small distances and therefore, no useful electrical work could be obtained. In these reactions, chemical energy appears as heat. If the transferance of electrons from zinc to copper ions is allowed to occur through some m ...
- Academy Test Bank
... OBJ: Perform calculations using the mole. TOP: stoichiometry | mass and moles of substance 24. A sample of gallium(III) sulfite, Ga2(SO3)3, contains 1.95 mol of sulfite ions. The number of moles of gallium(III) ions in the sample is A) 1.30 mol. B) 2.92 mol. C) 1.95 mol. D) 5.84 mol. E) 3.90 mol. A ...
... OBJ: Perform calculations using the mole. TOP: stoichiometry | mass and moles of substance 24. A sample of gallium(III) sulfite, Ga2(SO3)3, contains 1.95 mol of sulfite ions. The number of moles of gallium(III) ions in the sample is A) 1.30 mol. B) 2.92 mol. C) 1.95 mol. D) 5.84 mol. E) 3.90 mol. A ...
Chapter 3: Ionic and Covalent Compounds Chapter 3: Ionic and
... 29. An atom has the electron-dot symbol shown below. If this atom is a main group element, what charge will it form in its ionic state? ...
... 29. An atom has the electron-dot symbol shown below. If this atom is a main group element, what charge will it form in its ionic state? ...
Chapter 9 Review, pages 628–633
... 25. (a) Separate the equation H2O(l) + Au3+(aq) → O2(g) + Au(s) into two half-reactions. H2O(l) → O2(g) (oxidation) Au3+(aq) → Au(s) (reduction) For the oxidation half-reaction, first balance oxygen. 2 H2O(l) → O2(g) Balance hydrogen by adding hydrogen ions. 2 H2O(l) → O2(g) + 4 H+(aq) Balance the c ...
... 25. (a) Separate the equation H2O(l) + Au3+(aq) → O2(g) + Au(s) into two half-reactions. H2O(l) → O2(g) (oxidation) Au3+(aq) → Au(s) (reduction) For the oxidation half-reaction, first balance oxygen. 2 H2O(l) → O2(g) Balance hydrogen by adding hydrogen ions. 2 H2O(l) → O2(g) + 4 H+(aq) Balance the c ...
Inorganic chemistry

Inorganic chemistry deals with the synthesis and behavior of inorganic and organometallic compounds. This field covers all chemical compounds except the myriad organic compounds (carbon based compounds, usually containing C-H bonds), which are the subjects of organic chemistry. The distinction between the two disciplines is far from absolute, and there is much overlap, most importantly in the sub-discipline of organometallic chemistry. It has applications in every aspect of the chemical industry–including catalysis, materials science, pigments, surfactants, coatings, medicine, fuel, and agriculture.