Thermodynamics and Carnot Cycle Lab Exercise
... Experiment #2 – Carnot Cycle You will find this experiment under the "heat" tab of the program's menu. After clicking the Carnot Cycle link within the Heat tab menu you will get to the experimental setup. This experiment shows a Carnot cycle in action. You can choose the values of pressure and volum ...
... Experiment #2 – Carnot Cycle You will find this experiment under the "heat" tab of the program's menu. After clicking the Carnot Cycle link within the Heat tab menu you will get to the experimental setup. This experiment shows a Carnot cycle in action. You can choose the values of pressure and volum ...
Heat of Liberation
... – Heat of shortening dependent on load – Mommaerts separated heat of activation into two components, A – true heat of activation and f(P,t) – heat liberation which is a function of tension of muscle (P) and length of time (t) tension is ...
... – Heat of shortening dependent on load – Mommaerts separated heat of activation into two components, A – true heat of activation and f(P,t) – heat liberation which is a function of tension of muscle (P) and length of time (t) tension is ...
1. This question is about thermodynamic processes. (a) Distinguish
... An ideal gas is held in a container by a moveable piston and thermal energy is supplied to the gas such that it expands at a constant pressure of 1.2 × 105 Pa. ...
... An ideal gas is held in a container by a moveable piston and thermal energy is supplied to the gas such that it expands at a constant pressure of 1.2 × 105 Pa. ...
Chapter 12 Notes
... • If form A can be completely converted to form B, but the reverse is never complete, A is a higher grade of energy than B • When a high-grade energy is converted to internal energy, it can never be fully recovered as high-grade energy • Degradation of energy is the conversion of highgrade energy to ...
... • If form A can be completely converted to form B, but the reverse is never complete, A is a higher grade of energy than B • When a high-grade energy is converted to internal energy, it can never be fully recovered as high-grade energy • Degradation of energy is the conversion of highgrade energy to ...
First law
... a third, then they are in thermal equilibrium with each other. When two systems are put in contact with each other, there will be a net exchange of energy between them unless or until they are in thermal equilibrium, that is, they are at the same temperature. While this is a fundamental concept of t ...
... a third, then they are in thermal equilibrium with each other. When two systems are put in contact with each other, there will be a net exchange of energy between them unless or until they are in thermal equilibrium, that is, they are at the same temperature. While this is a fundamental concept of t ...
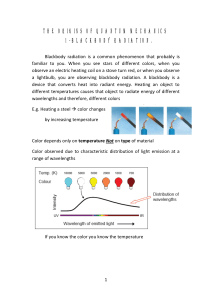
the origins of quantum mechanics 1
... is the energy density in units of energy per volume (e.g., J m-3), is the frequency of emitted radiation, T is the temperature of the blackbody, k is Boltzmann's constant, and c is the speed of light. This is known as the Rayleigh-Jeans law. A clear implication of this law is that as the frequency ...
... is the energy density in units of energy per volume (e.g., J m-3), is the frequency of emitted radiation, T is the temperature of the blackbody, k is Boltzmann's constant, and c is the speed of light. This is known as the Rayleigh-Jeans law. A clear implication of this law is that as the frequency ...
CYL100 2013–14 I semester Homework 2 Solutions 1. Consider a
... 0.917 g cm−3 respectively, and the heat of fusion is 6.004 kJ mol−1 . Since common experience is that ice skating is possible even when the ambient temperature is well below the normal freezing point, does the pressure induced lowering of the melting point explain clearly this observation? Straightf ...
... 0.917 g cm−3 respectively, and the heat of fusion is 6.004 kJ mol−1 . Since common experience is that ice skating is possible even when the ambient temperature is well below the normal freezing point, does the pressure induced lowering of the melting point explain clearly this observation? Straightf ...
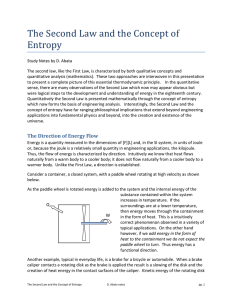
Professor David M. Stepp
... The Entropy of a system may increase or decrease during a process. The Entropy of the universe, taken as a system plus surroundings, can only increase. (The Second Law of Thermodynamics) “Entropy is Time’s Arrow” Note: The laws of thermodynamics are empirical, based on considerable experimental evid ...
... The Entropy of a system may increase or decrease during a process. The Entropy of the universe, taken as a system plus surroundings, can only increase. (The Second Law of Thermodynamics) “Entropy is Time’s Arrow” Note: The laws of thermodynamics are empirical, based on considerable experimental evid ...
Physics MCAS Study Guide Motion and Forces Distance
... Radiation-heat that is transferred by electromagnetic radiation (infrared rays). Only heat transfer that can travel through a vacuum. Travels at speed of light. Phase changes occur when substances are at their melting/freezing or boiling/condensing temperatures and energy is absorbed or released. No ...
... Radiation-heat that is transferred by electromagnetic radiation (infrared rays). Only heat transfer that can travel through a vacuum. Travels at speed of light. Phase changes occur when substances are at their melting/freezing or boiling/condensing temperatures and energy is absorbed or released. No ...
Document
... The internal energy (E) of a system is the sum of KE and PE of all particles in the system. The internal energy of a system can be changed by a flow of work, heat, or both. i.e. ΔE = q + w ΔE: change of E, q: heat, w: work Thermodynamic quantities always consist of two parts: a number, giving the ma ...
... The internal energy (E) of a system is the sum of KE and PE of all particles in the system. The internal energy of a system can be changed by a flow of work, heat, or both. i.e. ΔE = q + w ΔE: change of E, q: heat, w: work Thermodynamic quantities always consist of two parts: a number, giving the ma ...
• Conservation of energy principle • Total energy • Energy transfer
... between a solid surface and the adjacent liquid or gas which in motion and it involves the combined effects of “conduction and fluid motion”. The faster the fluid motion, the greater the convection heat transfer. In the absence of any bulk fluid motion, heat transfer between a solid surface and the ...
... between a solid surface and the adjacent liquid or gas which in motion and it involves the combined effects of “conduction and fluid motion”. The faster the fluid motion, the greater the convection heat transfer. In the absence of any bulk fluid motion, heat transfer between a solid surface and the ...
Chapter 5 Thermochemistry
... Methylhydrazine (CH6N2) is used as a liquid rocket fuel. The combustion of methylhydrazine with oxygen produces N2(g), CO2(g), and H2O(l): 2 CH6N2(l) + 5 O2(g) → 2 N2(g) + 2 CO2(g) + 6 H2O(l) When 4.00 g of methylhydrazine is combusted in a bomb calorimeter, the temperature of the calorimeter increa ...
... Methylhydrazine (CH6N2) is used as a liquid rocket fuel. The combustion of methylhydrazine with oxygen produces N2(g), CO2(g), and H2O(l): 2 CH6N2(l) + 5 O2(g) → 2 N2(g) + 2 CO2(g) + 6 H2O(l) When 4.00 g of methylhydrazine is combusted in a bomb calorimeter, the temperature of the calorimeter increa ...