H - Bruder Chemistry
... What amount of work is done when 15 L of gas is expanded to 25 L at 2.4 atm pressure? If 2.36 J of heat are absorbed by the gas above. what is the change in energy? How much heat would it take to change the gas without changing the internal energy of the gas? ...
... What amount of work is done when 15 L of gas is expanded to 25 L at 2.4 atm pressure? If 2.36 J of heat are absorbed by the gas above. what is the change in energy? How much heat would it take to change the gas without changing the internal energy of the gas? ...
EART 160: Planetary Sciences
... • Heat flows from hot to cold (thermodynamics) and is proportional to the temperature gradient • Here k is the thermal conductivity (W m-1 K-1) and units of F are W m-2 (heat flux is power per unit area) • Typical values for k are 2-4 Wm-1K-1 (rock, ice) and 3060 Wm-1K-1 (metal) • Solar heat flux at ...
... • Heat flows from hot to cold (thermodynamics) and is proportional to the temperature gradient • Here k is the thermal conductivity (W m-1 K-1) and units of F are W m-2 (heat flux is power per unit area) • Typical values for k are 2-4 Wm-1K-1 (rock, ice) and 3060 Wm-1K-1 (metal) • Solar heat flux at ...
Chapter 5 Thermochemistry Notes File
... Heat capacity = the amount of heat required to raise its temperature by 1 K (or 1ºC) -- larger the heat capacity the more heat required to raise a rise in temperature. Molar heat capacity = the heat capacity of 1 mole of a substance Specific heat = heat capacity of 1 gram of a substance q = (specifi ...
... Heat capacity = the amount of heat required to raise its temperature by 1 K (or 1ºC) -- larger the heat capacity the more heat required to raise a rise in temperature. Molar heat capacity = the heat capacity of 1 mole of a substance Specific heat = heat capacity of 1 gram of a substance q = (specifi ...
Thermodynamics
... In a coffee cup calorimeter, 50.0 mL of 0.100 M silver nitrate and 50.0 mL of 0.100 M HCl are mixed. The following reaction occurs: Ag+ (aq) + Cl- (aq) AgCl (s). If the two solutions are initially at 22.60 oC, and if the final temperature is 23.40 oC, calculate the change in enthalpy for the react ...
... In a coffee cup calorimeter, 50.0 mL of 0.100 M silver nitrate and 50.0 mL of 0.100 M HCl are mixed. The following reaction occurs: Ag+ (aq) + Cl- (aq) AgCl (s). If the two solutions are initially at 22.60 oC, and if the final temperature is 23.40 oC, calculate the change in enthalpy for the react ...
MS PowerPoint - Catalysis Eprints database
... The word “Thermodynamics” can be defined in various ways. One of the simplest ways being flow of heat. Thermodynamics deals with the energy changes associated with all types of physicochemical processes (reactions). The basis of thermodynamics is human experience rather than formal proof. Thermodyna ...
... The word “Thermodynamics” can be defined in various ways. One of the simplest ways being flow of heat. Thermodynamics deals with the energy changes associated with all types of physicochemical processes (reactions). The basis of thermodynamics is human experience rather than formal proof. Thermodyna ...
Kinetic Molecular Theory of Gases
... Heat is transferred into the system q > 0 Heat is transferred out of the system q < 0 ...
... Heat is transferred into the system q > 0 Heat is transferred out of the system q < 0 ...
Tutorial 3 – Thermodynamics of Dielectric Relaxations in Complex
... microscopic polarizability and macroscopic permittivity. From the phenomenological point of view, it is necessary to know the kinetic of the Polarization. From molecular one it’s required the knowledge of the effective Electric field at which the dipole is subjected. 4 different ways are proposed to ...
... microscopic polarizability and macroscopic permittivity. From the phenomenological point of view, it is necessary to know the kinetic of the Polarization. From molecular one it’s required the knowledge of the effective Electric field at which the dipole is subjected. 4 different ways are proposed to ...
Process
... THIRD LAW OF THERMODYNAMICS: The third law of thermodynamics, formulated by Walter Nernst and also known as the Nernst heat theorem, states that if one could reach absolute zero, all bodies would have the same entropy. Temperature approaches absolute zero (0 K), the entropy of a system approaches a ...
... THIRD LAW OF THERMODYNAMICS: The third law of thermodynamics, formulated by Walter Nernst and also known as the Nernst heat theorem, states that if one could reach absolute zero, all bodies would have the same entropy. Temperature approaches absolute zero (0 K), the entropy of a system approaches a ...
CHAPTER 9: Statistical Physics
... be linearly proportional to the applied electric field that is consistent with Ohm’s law. Prediction of the electrical conductivity: ...
... be linearly proportional to the applied electric field that is consistent with Ohm’s law. Prediction of the electrical conductivity: ...
Fundamentals of Energy Conversion
... In contrast, properties that may vary from point to point and that do not change with the mass of the system are called intensive properties. Temperature and pressure are well-known examples. For instance, thermometers at different locations in a system may indicate differing temperatures. But if a ...
... In contrast, properties that may vary from point to point and that do not change with the mass of the system are called intensive properties. Temperature and pressure are well-known examples. For instance, thermometers at different locations in a system may indicate differing temperatures. But if a ...
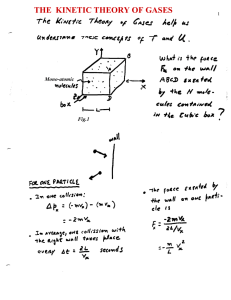
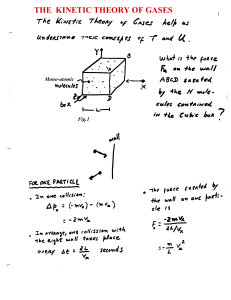
The Kinetic Theory of Gases
... As an application of the expression above for the equation of state, let’s consider an adiabatic process, one in which the gas container is thermally well isolated, i.e. no heat-transfer occurs. No heat-transfer is added or removed to the gas, Q = 0 As the piston is pressed, work is done on the gas ...
... As an application of the expression above for the equation of state, let’s consider an adiabatic process, one in which the gas container is thermally well isolated, i.e. no heat-transfer occurs. No heat-transfer is added or removed to the gas, Q = 0 As the piston is pressed, work is done on the gas ...
Chapter 6 CHEM 121
... • No ideal gases actually exist. • If they did exist, they would behave exactly as predicted by the gas laws at all temperatures and pressures. • Real gases deviate from the behavior predicted by the gas laws, but under normally encountered temperatures and pressures, the deviations are small. • Con ...
... • No ideal gases actually exist. • If they did exist, they would behave exactly as predicted by the gas laws at all temperatures and pressures. • Real gases deviate from the behavior predicted by the gas laws, but under normally encountered temperatures and pressures, the deviations are small. • Con ...
Physics 321 Final Exam May 1, `09
... 4. (10 pts.) The temperature of the Earth’s surface is maintained by radiation from the Sun. Use the Stefan-Boltzmann law for the energy emitted from the Sun and reradiated from the Earth to show that the ratio of the temperature of the Earth to that of the Sun is: ...
... 4. (10 pts.) The temperature of the Earth’s surface is maintained by radiation from the Sun. Use the Stefan-Boltzmann law for the energy emitted from the Sun and reradiated from the Earth to show that the ratio of the temperature of the Earth to that of the Sun is: ...
1 Introduction - Jefferson Lab
... approximately 3kW of thermal energy is required to produce 1kW of electrical energy. • In contrast, for ideal systems the conversion from mechanical to electrical energy (or visa-versa) can be 100%. • This thermodynamic limitation is expressed by the 2nd Law of Thermodynamics and embodies the concep ...
... approximately 3kW of thermal energy is required to produce 1kW of electrical energy. • In contrast, for ideal systems the conversion from mechanical to electrical energy (or visa-versa) can be 100%. • This thermodynamic limitation is expressed by the 2nd Law of Thermodynamics and embodies the concep ...