coordination of some monodentate and hybrid multident ate
... Metal-phosphoms coordination compounds provide inorganic chemistry with many elegant illustrations of the various stereochemical requirements of metal elements. For many years, coordination chemists have used trivalent phosphorus ligands to stabilise a large variety of oxidation states of transition ...
... Metal-phosphoms coordination compounds provide inorganic chemistry with many elegant illustrations of the various stereochemical requirements of metal elements. For many years, coordination chemists have used trivalent phosphorus ligands to stabilise a large variety of oxidation states of transition ...
Answers to Problem-Solving Practice Problems
... (4) Check your answer: Because the density is a little less than 1.00 g/mL, the volume in milliliters should be a little larger than the mass in grams. The calculated answer, 4.92 mL, is a little larger than the mass, 4.33 g. 1.2 Substance A must be a mixture since some of it dissolves and some, sub ...
... (4) Check your answer: Because the density is a little less than 1.00 g/mL, the volume in milliliters should be a little larger than the mass in grams. The calculated answer, 4.92 mL, is a little larger than the mass, 4.33 g. 1.2 Substance A must be a mixture since some of it dissolves and some, sub ...
Noncovalent interactions of molecules with single walled carbon
... where r is the distance between atoms, e is the well depth and s is the hard sphere radius of the atom. The e and s parameters are assigned the same value for the interior and exterior of a SWNT, an assumption that has not been rigorously shown to be true. It ...
... where r is the distance between atoms, e is the well depth and s is the hard sphere radius of the atom. The e and s parameters are assigned the same value for the interior and exterior of a SWNT, an assumption that has not been rigorously shown to be true. It ...
AQA Science GCSE Chemistry
... AQA recognizes the importance of good-quality teaching, learning and assessment resources to accompany their specification. That's why they've chosen to work exclusively with nelson Thornes. With AQA examiners providing content and quality control, you can be confident that this course is as closely ...
... AQA recognizes the importance of good-quality teaching, learning and assessment resources to accompany their specification. That's why they've chosen to work exclusively with nelson Thornes. With AQA examiners providing content and quality control, you can be confident that this course is as closely ...
08 Redox Reactions
... “The charge which an atom appears to have, when all other atoms are removed from it as ions is known as oxidation number.” OR “The oxidation number or oxidation state of an atom in a molecule or ion is defined as the number of charges it would carry if electrons were completely transferred.” Metals ...
... “The charge which an atom appears to have, when all other atoms are removed from it as ions is known as oxidation number.” OR “The oxidation number or oxidation state of an atom in a molecule or ion is defined as the number of charges it would carry if electrons were completely transferred.” Metals ...
Answers to SelectedTextbook Questions
... (e) Gibbs free energy, G = H − TS, combines enthalpy and entropy to give a quantity which must decrease for any processes that actually happens. (f) Lewisite is a chlorinate alkyl arsenic compound which was produced as a chemical weapon causing blisters and lung irritation. (g) A Lewis base ...
... (e) Gibbs free energy, G = H − TS, combines enthalpy and entropy to give a quantity which must decrease for any processes that actually happens. (f) Lewisite is a chlorinate alkyl arsenic compound which was produced as a chemical weapon causing blisters and lung irritation. (g) A Lewis base ...
CHAPTER 1 - THE MOLE SECTION 1
... In 1808 John Dalton published one of the first tables of atomic masses and actually listed the mass ratio of hydrogen to oxygen as 1 to 8 based upon his observations. He thought that the formula for water was HO with one atom of hydrogen to one atom of oxygen. In the above case the weight ratio of t ...
... In 1808 John Dalton published one of the first tables of atomic masses and actually listed the mass ratio of hydrogen to oxygen as 1 to 8 based upon his observations. He thought that the formula for water was HO with one atom of hydrogen to one atom of oxygen. In the above case the weight ratio of t ...
Polyhedral Oligomeric Silsesquioxane
... phase-transfer agent is usually required to cover the water-soluble metal salts, and then they are transferred into the organic phase. Different from hydrophilic metal salts, the palladium acetate [Pd(OAc)2] is a source of Pd salts that can be dissolved in many organic solvents (e.g., toluene, tetrah ...
... phase-transfer agent is usually required to cover the water-soluble metal salts, and then they are transferred into the organic phase. Different from hydrophilic metal salts, the palladium acetate [Pd(OAc)2] is a source of Pd salts that can be dissolved in many organic solvents (e.g., toluene, tetrah ...
Solid-State 23Na Nuclear Magnetic Resonance of Sodium
... In general, solid-state 23Na NMR spectra of sodiumionophore complexes were obtained using MAS and high-power proton decoupling. Under such circumstances, the observed central-transition 23Na NMR spectra exhibit typical features arising from second-order quadrupole interaction. In most cases only a s ...
... In general, solid-state 23Na NMR spectra of sodiumionophore complexes were obtained using MAS and high-power proton decoupling. Under such circumstances, the observed central-transition 23Na NMR spectra exhibit typical features arising from second-order quadrupole interaction. In most cases only a s ...
© www.CHEMSHEETS.co.uk 17-Jul
... it is best to show each separate step (e.g. if both elements are atomised, show this as two steps) ...
... it is best to show each separate step (e.g. if both elements are atomised, show this as two steps) ...
Chapter 1: Matter and Measurements
... (a) “Compounds containing carbon atoms are molecular” is usually true. Carbon, being a nonmetal can combine with other non-metals to form molecular compounds such as those mentioned in Section 2.4 (indeed, a whole field of chemistry, called organic chemistry, is dedicated to the study of such compou ...
... (a) “Compounds containing carbon atoms are molecular” is usually true. Carbon, being a nonmetal can combine with other non-metals to form molecular compounds such as those mentioned in Section 2.4 (indeed, a whole field of chemistry, called organic chemistry, is dedicated to the study of such compou ...
Soln Chem 2008Nov(9746)
... When the pack is squeezed, NH4NO3(s) dissolves in the water suggests that the reaction is spontaneous; i.e. ∆G is negative. Dissolution of NH4NO3(s) is accompanied by an increase in entropy (less orderly); i.e. ∆S is positive. Hence, option C. (ans) © Step-by-Step ...
... When the pack is squeezed, NH4NO3(s) dissolves in the water suggests that the reaction is spontaneous; i.e. ∆G is negative. Dissolution of NH4NO3(s) is accompanied by an increase in entropy (less orderly); i.e. ∆S is positive. Hence, option C. (ans) © Step-by-Step ...
mcdonald (pam78654) – HW 1: High School Concepts – laude
... mcdonald (pam78654) – HW 1: High School Concepts – laude – (89560) This print-out should have 40 questions. Multiple-choice questions may continue on the next column or page – find all choices before answering. 001 10.0 points Calculate the number of H2 O molecules in 1.00 cm3 of water at 0◦ C (dens ...
... mcdonald (pam78654) – HW 1: High School Concepts – laude – (89560) This print-out should have 40 questions. Multiple-choice questions may continue on the next column or page – find all choices before answering. 001 10.0 points Calculate the number of H2 O molecules in 1.00 cm3 of water at 0◦ C (dens ...
edexcel_u1_2010_2013..
... of hydrogen produced was 66 cm3, measured at room temperature and pressure. (i) Draw a labelled diagram of the apparatus you would use to carry out this experiment, showing how you would collect the hydrogen produced and ...
... of hydrogen produced was 66 cm3, measured at room temperature and pressure. (i) Draw a labelled diagram of the apparatus you would use to carry out this experiment, showing how you would collect the hydrogen produced and ...
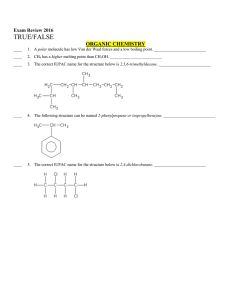
Multiple Choice Exam Review June 2016
... d. atoms which both have equally low ionization energies e. atoms which have low but unequal ionization energies According to VSEPR theory, molecules adjust their shapes to keep which of the following as far apart as possible? a. pairs of valence electrons d. mobile electrons b. inner shell electron ...
... d. atoms which both have equally low ionization energies e. atoms which have low but unequal ionization energies According to VSEPR theory, molecules adjust their shapes to keep which of the following as far apart as possible? a. pairs of valence electrons d. mobile electrons b. inner shell electron ...
Technical description of GLYDE-II 1 Supplemental Information
... a molecule, such as a monosaccharide. This monosaccharide, in turn, can be referenced (used as an archetype) to specify a glycosyl residue that is a part of a glycan molecule. This glycan molecule, in turn, can be referenced (used as an archetype) to specify a glycosyl moiety that is part of a glyco ...
... a molecule, such as a monosaccharide. This monosaccharide, in turn, can be referenced (used as an archetype) to specify a glycosyl residue that is a part of a glycan molecule. This glycan molecule, in turn, can be referenced (used as an archetype) to specify a glycosyl moiety that is part of a glyco ...
Schaum`s Outline of Theory and Problems of
... combine elements. Changing one combination of elements to another is the chief interest of the chemist. It has long been of interest to know the composition of the crust of the earth, the oceans, and the atmosphere, since these are the only sources of raw materials for all the products that humans r ...
... combine elements. Changing one combination of elements to another is the chief interest of the chemist. It has long been of interest to know the composition of the crust of the earth, the oceans, and the atmosphere, since these are the only sources of raw materials for all the products that humans r ...
physical setting chemistry
... 15 The high electrical conductivity of metals is primarily due to ...
... 15 The high electrical conductivity of metals is primarily due to ...
Coherent Spin Dynamics of a Spin-1 Bose-Einstein
... are analogous to Josephson oscillations in weakly connected superconductors and represent a type of matter-wave four-wave mixing. We also demonstrate control of the coherent evolution of the system using magnetic fields. In the first part of this thesis, the all-optical approaches to BEC that were f ...
... are analogous to Josephson oscillations in weakly connected superconductors and represent a type of matter-wave four-wave mixing. We also demonstrate control of the coherent evolution of the system using magnetic fields. In the first part of this thesis, the all-optical approaches to BEC that were f ...
Spin Physics in Two-dimensional Systems Daniel Gosálbez Martínez
... is reduced to two inequivalent points situated at the corners of the first Brillouin zone. At this point, the valence band and the conduction band touch each other with a lineal dispersion relation. The physics in the low energy spectrum is described by the π orbitals of the graphene, while the high ...
... is reduced to two inequivalent points situated at the corners of the first Brillouin zone. At this point, the valence band and the conduction band touch each other with a lineal dispersion relation. The physics in the low energy spectrum is described by the π orbitals of the graphene, while the high ...
Elementary Steps, the Role of Chemisorbed Oxygen, and the Effects
... constants initially decreased and then reached constant values as O2 pressure and O* coverage increased as a result of a transition in the surface species involved in kinetically relevant C!H bond activation steps from O*!* to O*!O* site pairs (*, vacancy site). On O*!O* site pairs, C!H bonds are cl ...
... constants initially decreased and then reached constant values as O2 pressure and O* coverage increased as a result of a transition in the surface species involved in kinetically relevant C!H bond activation steps from O*!* to O*!O* site pairs (*, vacancy site). On O*!O* site pairs, C!H bonds are cl ...
master ap chemistry - NelnetSolutions.com
... For more information, contact Peterson’s, 2000 Lenox Drive, Lawrenceville, NJ 08648; 800-338-3282; or find us on the World Wide Web at: www.petersons.com/about. © 2007 Peterson’s, a Nelnet company Previous edition © 2005 Editor: Wallie Walker Hammond; Production Editor: Mark D. Snider; Composition M ...
... For more information, contact Peterson’s, 2000 Lenox Drive, Lawrenceville, NJ 08648; 800-338-3282; or find us on the World Wide Web at: www.petersons.com/about. © 2007 Peterson’s, a Nelnet company Previous edition © 2005 Editor: Wallie Walker Hammond; Production Editor: Mark D. Snider; Composition M ...
Question Bank (Class XI - Chemistry)
... Q3- What is a chemical equation? What are its essential features? (L-2) Ans. the qualitative and quantitative representation of a chemical reaction in short form in terms of symbols and formulae is called chemical equation. For example, on heating calcium carbonate, it gives Caco3 →Ca0 + CO2 Essenti ...
... Q3- What is a chemical equation? What are its essential features? (L-2) Ans. the qualitative and quantitative representation of a chemical reaction in short form in terms of symbols and formulae is called chemical equation. For example, on heating calcium carbonate, it gives Caco3 →Ca0 + CO2 Essenti ...
Electronic-structure calculations by first-principles
... ternate way around this problem was proposed and applied within a DFT-in-DFT embedding scheme by Cortona,20 where kinetic-energy cross terms are explicitly included. This method has since been applied in a similar fashion by Wesolowski, Warshal, and Weber 22 to examine a solute in solution and by Tr ...
... ternate way around this problem was proposed and applied within a DFT-in-DFT embedding scheme by Cortona,20 where kinetic-energy cross terms are explicitly included. This method has since been applied in a similar fashion by Wesolowski, Warshal, and Weber 22 to examine a solute in solution and by Tr ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.