Molecules and Ions
... Molecules and their Chemical Formulas There are two ways of describing the components (i.e. the number and type of atoms) found inside any molecule: Molecular Formula: the actual number and type of atoms in a compound, e.g. hydrogen peroxide = H2O2 Empirical Formula: the lowest whole number ratio of ...
... Molecules and their Chemical Formulas There are two ways of describing the components (i.e. the number and type of atoms) found inside any molecule: Molecular Formula: the actual number and type of atoms in a compound, e.g. hydrogen peroxide = H2O2 Empirical Formula: the lowest whole number ratio of ...
OXIDATION OF CYCLOHEXANOL TO CYCLOHEXANONE The
... The oxidation of cyclohexanol to cyclohexanone involves the removal of hydrogen from the OH group. After separation and purification, an Infrared Spectrum will be run to determine the composition of the recovered material. Infrared Spectroscopy is a very powerful technique used in the determination ...
... The oxidation of cyclohexanol to cyclohexanone involves the removal of hydrogen from the OH group. After separation and purification, an Infrared Spectrum will be run to determine the composition of the recovered material. Infrared Spectroscopy is a very powerful technique used in the determination ...
Chemistry notes Important terms *Mass of element in a sample
... Ionic compound – electron transfer from a metal to a non-metal Covalent compound- electron sharing between two non-metals Hydrates- have a specific number of water molecules associated with each formula unit ( shown with a *#H2O) Aqueous solutions are solutions in water Combustion analysis- add O2 ...
... Ionic compound – electron transfer from a metal to a non-metal Covalent compound- electron sharing between two non-metals Hydrates- have a specific number of water molecules associated with each formula unit ( shown with a *#H2O) Aqueous solutions are solutions in water Combustion analysis- add O2 ...
CLASSES AND NOMENCLATURE OF INORGANIC COMPOUNDS
... D 16 times D increase of the pressure E 6 times E decrease of the temperature 9. Speed of what reactions increases if the 10. The law of mass action describes the temperature is increased? dependence of rate of chemical reaction on: A endothermic A the concentration of reactans B exothermic B areas ...
... D 16 times D increase of the pressure E 6 times E decrease of the temperature 9. Speed of what reactions increases if the 10. The law of mass action describes the temperature is increased? dependence of rate of chemical reaction on: A endothermic A the concentration of reactans B exothermic B areas ...
Mole Ratio and Stoichiometry
... 0.25 moles of element X was found to have a mass of 28g. Calculate its atomic mass. Identify element X. ...
... 0.25 moles of element X was found to have a mass of 28g. Calculate its atomic mass. Identify element X. ...
PRACTICAL ORGANIC CHEMISTRY
... formed which gives on further heating gum-like materials known as dextrin. The final hydrolysis products are maltose and glucose. ...
... formed which gives on further heating gum-like materials known as dextrin. The final hydrolysis products are maltose and glucose. ...
Membrane Permeability A. Permeability If you take a pure solution of
... B. Membrane proteins. So how do ions and other substance pass through the membrane? Many transmembrane proteins exist, that form an aqueous channel in the lipid bilayer. How does a protein cross the membrane, if membranes are nonpolar and peptides are polar? It turns out that almost all protein stre ...
... B. Membrane proteins. So how do ions and other substance pass through the membrane? Many transmembrane proteins exist, that form an aqueous channel in the lipid bilayer. How does a protein cross the membrane, if membranes are nonpolar and peptides are polar? It turns out that almost all protein stre ...
Chapter 4: Solution Chemistry and the Hydrosphere
... 1. The oxidation number of an element in its natural form is 0. Examples: the oxidation number is zero for each element in H2, O2, Cl2, P4, Na, etc. 2. The oxidation number of a monatomic ion is the charge on the ion. Examples: Na3N, the ions are Na+ and N3–, so oxidation #’s: Na = +1 and N = -3. In ...
... 1. The oxidation number of an element in its natural form is 0. Examples: the oxidation number is zero for each element in H2, O2, Cl2, P4, Na, etc. 2. The oxidation number of a monatomic ion is the charge on the ion. Examples: Na3N, the ions are Na+ and N3–, so oxidation #’s: Na = +1 and N = -3. In ...
Single crystal structure determination using synchrotron X
... (m2/s), which indicated a single product formation of Pt12L24 sphere 2 shown in Fig. 2 and was comparable to that of the Pd 12 L 24 counterpart. The control experiment for the reaction without TFE, i.e., in DMSO only, showed no convergence to a single product: the unassignable mixture of unstable in ...
... (m2/s), which indicated a single product formation of Pt12L24 sphere 2 shown in Fig. 2 and was comparable to that of the Pd 12 L 24 counterpart. The control experiment for the reaction without TFE, i.e., in DMSO only, showed no convergence to a single product: the unassignable mixture of unstable in ...
- Kendriya Vidyalaya NKJ Katni
... 16. Why SO2 is a better reducing agent in alkaline medium as compared to that in acidic medium ? Explain. ...
... 16. Why SO2 is a better reducing agent in alkaline medium as compared to that in acidic medium ? Explain. ...
king fahd university of petroleum and minerals chemistry
... 2. For the reaction A product, successive half-lives (i.e., first, second and third half-lives) are observed to be 10, 20, 40 min for an experiment in which [A]0 = 0.10 M. Calculate the concentration of A at t = 30 min. A) B) C) D) ...
... 2. For the reaction A product, successive half-lives (i.e., first, second and third half-lives) are observed to be 10, 20, 40 min for an experiment in which [A]0 = 0.10 M. Calculate the concentration of A at t = 30 min. A) B) C) D) ...
System International Base Units
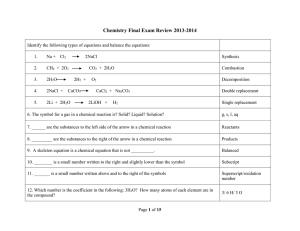
... Double Replacement Reaction – ions recombine in solution to form a solid, liquid, or gas; reactions will not occur if solid, liquid, or gas is not formed Precipitation Reaction AB(aq) + ED(aq) AD(s) + EB(aq) o Example: AgNO3(aq) + NaCl(aq) AgCl(s) + NaNO3(aq) o Use the solubility table to pred ...
... Double Replacement Reaction – ions recombine in solution to form a solid, liquid, or gas; reactions will not occur if solid, liquid, or gas is not formed Precipitation Reaction AB(aq) + ED(aq) AD(s) + EB(aq) o Example: AgNO3(aq) + NaCl(aq) AgCl(s) + NaNO3(aq) o Use the solubility table to pred ...
Document
... 3. Richard complained about a thin black film that formed on his aluminum windows. Kim told him that this film was the result of oxidation (rusting) of the aluminum by oxygen in the air. Which balanced equation represents this reaction? A) 2 Al + O2 → Al2O3 C) 2 Al + 3 O2 → Al2O3 B) 4 Al + 3 O2 → 2 ...
... 3. Richard complained about a thin black film that formed on his aluminum windows. Kim told him that this film was the result of oxidation (rusting) of the aluminum by oxygen in the air. Which balanced equation represents this reaction? A) 2 Al + O2 → Al2O3 C) 2 Al + 3 O2 → Al2O3 B) 4 Al + 3 O2 → 2 ...
ert207 analytical chemistry
... What is Analytical Chemistry? • Concerned with the chemical characterization of matter and the answer of : 1) What is it (Qualitative) –identification of elements, ions or compound 2) How much is it (Quantitative) ...
... What is Analytical Chemistry? • Concerned with the chemical characterization of matter and the answer of : 1) What is it (Qualitative) –identification of elements, ions or compound 2) How much is it (Quantitative) ...
Chemistry Review
... 1. expansion - gases completely fill any container in which they are enclosed, and they take its shape. 2. fluidity - gas particles glide easily past one another because the attractive forces are insignificant; because liquids and gases flow, they are referred to as fluids. 3. low density – gases ar ...
... 1. expansion - gases completely fill any container in which they are enclosed, and they take its shape. 2. fluidity - gas particles glide easily past one another because the attractive forces are insignificant; because liquids and gases flow, they are referred to as fluids. 3. low density – gases ar ...
FM 10-67-2 Chapter 7
... responsible to identify chemical substances (perform qualitative analysis) and estimate quantities present (perform quantitative analysis) in their examination of fuel samples. This chapter addresses some of the basic terms, formulas, tests, and equipmentthey will use. DEFINITION Matter is anything ...
... responsible to identify chemical substances (perform qualitative analysis) and estimate quantities present (perform quantitative analysis) in their examination of fuel samples. This chapter addresses some of the basic terms, formulas, tests, and equipmentthey will use. DEFINITION Matter is anything ...