Proximity Effects on Reaction Rates
... to deprotonate water • The enhanced nucleophilicity of deprotonated water ...
... to deprotonate water • The enhanced nucleophilicity of deprotonated water ...
Chapter 12: Chemical Equilibrium • Chemical Equilibrium
... • In aqueous solutions, H3O+ (aq) & OH- (aq) are the strongest acids & bases that exist. Stronger bases react with water to form OH- (aq) O2- (aq) + H2O () OH- (aq) + OH- (aq) Stronger acids react with water to form H3O+ (aq). HCl (aq) + H2O () H3O+ (aq) + Cl- (aq) This is known as the Levelin ...
... • In aqueous solutions, H3O+ (aq) & OH- (aq) are the strongest acids & bases that exist. Stronger bases react with water to form OH- (aq) O2- (aq) + H2O () OH- (aq) + OH- (aq) Stronger acids react with water to form H3O+ (aq). HCl (aq) + H2O () H3O+ (aq) + Cl- (aq) This is known as the Levelin ...
Cambridge International Examinations Cambridge
... (c) When iron is made into the alloy steel, the properties of iron are changed. High carbon steels are stronger than iron but are brittle. State a property of low carbon steels. ...
... (c) When iron is made into the alloy steel, the properties of iron are changed. High carbon steels are stronger than iron but are brittle. State a property of low carbon steels. ...
Chemical Equations
... “yields”; indicates result of a reaction Indicates a reversible reaction A reactant or product in the solid state Alternative to (s); used only for a precipitate (solid) falling out of solution A reactant or product in the liquid state A reactant or product in aqueous solution (dissolved in water) A ...
... “yields”; indicates result of a reaction Indicates a reversible reaction A reactant or product in the solid state Alternative to (s); used only for a precipitate (solid) falling out of solution A reactant or product in the liquid state A reactant or product in aqueous solution (dissolved in water) A ...
Chemistry (SPA)
... 440 BC, the Greek Leucippus and his pupil Democritus coined the term atomos to describe the smallest particle of matter. It translates to mean something that is indivisible. In the eighteenth century, chemist, John Dalton, revived the term when he suggested that each element was made up of unique at ...
... 440 BC, the Greek Leucippus and his pupil Democritus coined the term atomos to describe the smallest particle of matter. It translates to mean something that is indivisible. In the eighteenth century, chemist, John Dalton, revived the term when he suggested that each element was made up of unique at ...
35 IChO Problems 1-13
... be coupled into a singlet) is P, predict the ground state electron configuration for these complexes. Hint: The Aufbau principle and the Pauli exclusion principle must be obeyed. Problem 16: Isomerism in Inorganic Chemistry Isomerism is traditionally introduced in Organic chemistry courses. However ...
... be coupled into a singlet) is P, predict the ground state electron configuration for these complexes. Hint: The Aufbau principle and the Pauli exclusion principle must be obeyed. Problem 16: Isomerism in Inorganic Chemistry Isomerism is traditionally introduced in Organic chemistry courses. However ...
PDF version - ltcconline.net
... destruction of outdoor marble statues and the exteriors of limestone buildings. The chemistry is simple: marble and limestone are forms of calcium carbonate, which can react with sulfuric acid to form calcium sulfate: ...
... destruction of outdoor marble statues and the exteriors of limestone buildings. The chemistry is simple: marble and limestone are forms of calcium carbonate, which can react with sulfuric acid to form calcium sulfate: ...
chemistry sp.indd
... syllabus that will be examined in 2010, 2011 and 2012. The purpose of these materials is to provide Centres with a reasonable idea of the general shape and character of the planned question papers in advance of the first operational examination. If there are any changes to the syllabus CIE will writ ...
... syllabus that will be examined in 2010, 2011 and 2012. The purpose of these materials is to provide Centres with a reasonable idea of the general shape and character of the planned question papers in advance of the first operational examination. If there are any changes to the syllabus CIE will writ ...
practice problems of chap4_5 - Chemistry
... 6. Which of the following redox reactions will occur according to activity series? (a) Cu(s) + 2HCl(aq) CuCl2(aq) + H2(g) (b) Zn(s) + CuSO4(aq) ZnSO4(aq) + Cu(s) (c) Fe(s) + ZnSO4(aq) FeSO4(aq) + Zn(s) (d) 2Al(s) + 3Ca(NO3)2(aq) 2Al((NO3)3(aq) + 3Ca(s) Hint: Activity table, Fig. 4.6, p. 139. ...
... 6. Which of the following redox reactions will occur according to activity series? (a) Cu(s) + 2HCl(aq) CuCl2(aq) + H2(g) (b) Zn(s) + CuSO4(aq) ZnSO4(aq) + Cu(s) (c) Fe(s) + ZnSO4(aq) FeSO4(aq) + Zn(s) (d) 2Al(s) + 3Ca(NO3)2(aq) 2Al((NO3)3(aq) + 3Ca(s) Hint: Activity table, Fig. 4.6, p. 139. ...
Exam 2
... • give simplified answers with an appropriate number of significant figures to all numerical questions; unsimplified answers will not be given full marks. • show all working in your answers to numerical questions. No credit will be given for an incorrect answer unless it is accompanied by details of ...
... • give simplified answers with an appropriate number of significant figures to all numerical questions; unsimplified answers will not be given full marks. • show all working in your answers to numerical questions. No credit will be given for an incorrect answer unless it is accompanied by details of ...
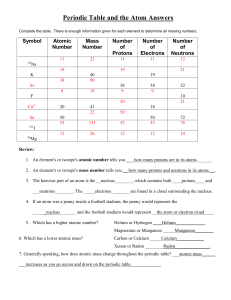
Periodic Table and the Atom Answers
... stoichiometry problems, I would highly suggest consulting this section of the site before answering these questions. When doing stoichiometry problems, people are frequently worried by statements such as “if you have an excess of (compound X)”. This statement shouldn’t worry you… what it really mean ...
... stoichiometry problems, I would highly suggest consulting this section of the site before answering these questions. When doing stoichiometry problems, people are frequently worried by statements such as “if you have an excess of (compound X)”. This statement shouldn’t worry you… what it really mean ...
Chapter 8 - Chemical Equations
... Step 1 – Look at the element by itself. Is this element a metal or a nonmetal? Al (aluminum) is a metal because it is located to the left side of the staircase line on the Periodic Table. Step 2 – You will compare the type of element by itself to the similar type of element in the compound. In this ...
... Step 1 – Look at the element by itself. Is this element a metal or a nonmetal? Al (aluminum) is a metal because it is located to the left side of the staircase line on the Periodic Table. Step 2 – You will compare the type of element by itself to the similar type of element in the compound. In this ...
Appendix - Cengage
... molecules (● Figure B-4a). (A subscript following a chemical symbol indicates the number of that type of atom present in the molecule.) Several other nonmetallic elements also exist as molecules, because covalent bonds form between identical atoms; oxygen (O2) is an example (● Figure B-4b). Often, a ...
... molecules (● Figure B-4a). (A subscript following a chemical symbol indicates the number of that type of atom present in the molecule.) Several other nonmetallic elements also exist as molecules, because covalent bonds form between identical atoms; oxygen (O2) is an example (● Figure B-4b). Often, a ...
Answer Key, Problem Set 6 – complete, with explanations
... ions, I have shown the ions as “touching” here—you could have shown them with a bit of space in between them as well, as long as the amount of space in between was roughly “equal” for all adjacent ions). To further ...
... ions, I have shown the ions as “touching” here—you could have shown them with a bit of space in between them as well, as long as the amount of space in between was roughly “equal” for all adjacent ions). To further ...