Plastic siphon drum pump chemical resistant Plastic siphon drum
... Polyethylene construction with discharge hose. Suction type is manual up and down piston type. Use to transfer these liquids Gasoline, Diesel, Kerosene, Toluene, Thinner, Nitric acid up to 35 percent, Hydrochloric acid, Sulfuric acid up to 35 percent, Acetic acid up to 35 percent, Water Etc. The tot ...
... Polyethylene construction with discharge hose. Suction type is manual up and down piston type. Use to transfer these liquids Gasoline, Diesel, Kerosene, Toluene, Thinner, Nitric acid up to 35 percent, Hydrochloric acid, Sulfuric acid up to 35 percent, Acetic acid up to 35 percent, Water Etc. The tot ...
Differentiated Chemistry First Term Test Review
... A commercially valuable paint and adhesive stripper, dimethyl sulfoxide (DMSO), (CH3)2SO, can be prepared by the reaction of oxygen with dimethyl sulfide, (CH3)2S, using a ratio of one mole oxygen to two moles of the sulfide: O2 + 2(CH3)2S → 2(CH3)2SO If this process has an 83% percent yield, how ma ...
... A commercially valuable paint and adhesive stripper, dimethyl sulfoxide (DMSO), (CH3)2SO, can be prepared by the reaction of oxygen with dimethyl sulfide, (CH3)2S, using a ratio of one mole oxygen to two moles of the sulfide: O2 + 2(CH3)2S → 2(CH3)2SO If this process has an 83% percent yield, how ma ...
C3.1 The Periodic Table
... Write the equation for the reversible reaction between hydrogen and nitrogen gases that produces ammonia gas State that, on cooling, the ammonia liquefies and is removed, while the remaining hydrogen and nitrogen are recycled Evaluate the conditions necessary in an industrial process to maximise yie ...
... Write the equation for the reversible reaction between hydrogen and nitrogen gases that produces ammonia gas State that, on cooling, the ammonia liquefies and is removed, while the remaining hydrogen and nitrogen are recycled Evaluate the conditions necessary in an industrial process to maximise yie ...
Re-typed from The Ultimate Chemical Equations Handbook by
... of water molecules needs to be adjusted. 5. If there is an odd number of an element on one side and an even number on the other, the odd number will need to be evened out – so use a coefficient of 2 for that substance. 6. If there are polyatomic ions that remain together as a unit during the reactio ...
... of water molecules needs to be adjusted. 5. If there is an odd number of an element on one side and an even number on the other, the odd number will need to be evened out – so use a coefficient of 2 for that substance. 6. If there are polyatomic ions that remain together as a unit during the reactio ...
Lecture 22 - Chemistry Courses
... As some base is added (but well before stoichiometric point), the concentration of A- ions comes almost entirely from the following reaction (which formally is the formation of a salt, MA): ...
... As some base is added (but well before stoichiometric point), the concentration of A- ions comes almost entirely from the following reaction (which formally is the formation of a salt, MA): ...
Chemistry Final Exam Test Yourself I
... one, is it a strong or weak acid? (strong) Which gas law includes pressure, volume, temperature, and the # of moles? ...
... one, is it a strong or weak acid? (strong) Which gas law includes pressure, volume, temperature, and the # of moles? ...
Toluenediamine
... conversion of toluene to dinitrotoluene will not work with toluene in excess as its own solvent, because toluene will always be more easily nitrated than nitrotoluene and the “by-product” water will lower the acidity. Consequently, only a two-step process or a process using an additional solvent are ...
... conversion of toluene to dinitrotoluene will not work with toluene in excess as its own solvent, because toluene will always be more easily nitrated than nitrotoluene and the “by-product” water will lower the acidity. Consequently, only a two-step process or a process using an additional solvent are ...
A Classification of AP Chemistry Reactions
... The second type of redox that involve oxygen-containing compounds such as nitrates, sulfates, permanganates, dichromates, etc. First of all, since these are redox reactions, one thing must be oxidized and another must be reduced. Jotting down oxidation numbers can be helpful. Second, almost all of t ...
... The second type of redox that involve oxygen-containing compounds such as nitrates, sulfates, permanganates, dichromates, etc. First of all, since these are redox reactions, one thing must be oxidized and another must be reduced. Jotting down oxidation numbers can be helpful. Second, almost all of t ...
UNIT 7 – CHEMICAL REACTIONS
... Reactions That Form Gases 1. A third type of double-replacement reaction that occurs in aqueous solutions results in the formation of a gas. ...
... Reactions That Form Gases 1. A third type of double-replacement reaction that occurs in aqueous solutions results in the formation of a gas. ...
South Pasadena • Chemistry Name Period Date 5 · Chemical
... 4. A solution of lead acetate is combined with a solution of hydrochloric acid forming a lead chloride precipitate and acetic acid. (a) Write the balanced equation for the reaction. ...
... 4. A solution of lead acetate is combined with a solution of hydrochloric acid forming a lead chloride precipitate and acetic acid. (a) Write the balanced equation for the reaction. ...
Spring 2001 Key
... -----------------------------------------------------------------------------------------------------------(d) is a false statement: production of 6 moles of CO2 will produce 4440 kJ of heat. The reaction is exothermic. Energy would be required if the reaction was endothermic. (a) is true since for ...
... -----------------------------------------------------------------------------------------------------------(d) is a false statement: production of 6 moles of CO2 will produce 4440 kJ of heat. The reaction is exothermic. Energy would be required if the reaction was endothermic. (a) is true since for ...
Analytical Chemistry
... equilibrium constant. To understand the effect of temperature, we must know whether the reaction is endothermic (absorption of heat) or exothermic (release ...
... equilibrium constant. To understand the effect of temperature, we must know whether the reaction is endothermic (absorption of heat) or exothermic (release ...
1) In the reaction H2O + CH3COOH H3O+ + CH3COO
... Applied Problem. You must show all your work for complete credit. 9) (15 points) The purpose of a catalyst is to lower the activation energy of a reaction (in fact, that is all that a catalyst does). The enzymes in your body which mediate chemical reactions are catalysts. One of these enzymes is ca ...
... Applied Problem. You must show all your work for complete credit. 9) (15 points) The purpose of a catalyst is to lower the activation energy of a reaction (in fact, that is all that a catalyst does). The enzymes in your body which mediate chemical reactions are catalysts. One of these enzymes is ca ...
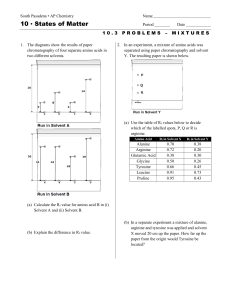
10 States of Matter
... 3. What is the vapor pressure of a solution by mixing 35.0 grams of urea (CH4N2O) with 150.0 grams of acetone (C3H6O) at 40°C? Assume urea is nonvolatile, and the vapor pressure of pure acetone is 400. torr at 40°C. ...
... 3. What is the vapor pressure of a solution by mixing 35.0 grams of urea (CH4N2O) with 150.0 grams of acetone (C3H6O) at 40°C? Assume urea is nonvolatile, and the vapor pressure of pure acetone is 400. torr at 40°C. ...
Water and Aqueous Solutions - Chemistry at Winthrop University
... Structure of the Water Molecule • Octet rule dictates that there are four electron pairs around an oxygen atom in water • These electrons are in four sp3 orbitals • Two of these pairs covalently link two hydrogen atoms to a central oxygen atom • The two remaining pairs remain ...
... Structure of the Water Molecule • Octet rule dictates that there are four electron pairs around an oxygen atom in water • These electrons are in four sp3 orbitals • Two of these pairs covalently link two hydrogen atoms to a central oxygen atom • The two remaining pairs remain ...
BIOC 462a -- General Chemistry Review
... Grisham, Biochemistry, 3rd ed., 2004. For a polyprotic acid (more than one acidic group, so more than one pKa on titration curve), it takes 1 equivalent of OH– to titrate each acidic group, so a triprotic acid like phosphoric acid requires 3 equivalents. The pKa values are pH values at each half equ ...
... Grisham, Biochemistry, 3rd ed., 2004. For a polyprotic acid (more than one acidic group, so more than one pKa on titration curve), it takes 1 equivalent of OH– to titrate each acidic group, so a triprotic acid like phosphoric acid requires 3 equivalents. The pKa values are pH values at each half equ ...
PDF notes - Chemistry and Biochemistry
... Grisham, Biochemistry, 3rd ed., 2004. For a polyprotic acid (more than one acidic group, so more than one pKa on titration curve), it takes 1 equivalent of OH– to titrate each acidic group, so a triprotic acid like phosphoric acid requires 3 equivalents. The pKa values are pH values at each half equ ...
... Grisham, Biochemistry, 3rd ed., 2004. For a polyprotic acid (more than one acidic group, so more than one pKa on titration curve), it takes 1 equivalent of OH– to titrate each acidic group, so a triprotic acid like phosphoric acid requires 3 equivalents. The pKa values are pH values at each half equ ...
General Chemistry Sample Exam 2 and Outline
... Some sulfuric acid is spilled on a lab bench. Sprinkling some sodium bicarbonate on to the sulfuric acid can neutralize it. The reaction produces sodium sulfate, carbon dioxide and water. i) Write the molecular, ionic and net ionic equation and balance the equation (include the phases). ii) What is ...
... Some sulfuric acid is spilled on a lab bench. Sprinkling some sodium bicarbonate on to the sulfuric acid can neutralize it. The reaction produces sodium sulfate, carbon dioxide and water. i) Write the molecular, ionic and net ionic equation and balance the equation (include the phases). ii) What is ...
Single Replacement Reactions
... Comprehension of key ideas and details (3) o Cites convincing evidence to support analysis, showing full comprehension of complex ideas expressed in the texts provided. Development of ideas and organization (2) o Student response addresses the prompt and provides effective development of the claim ...
... Comprehension of key ideas and details (3) o Cites convincing evidence to support analysis, showing full comprehension of complex ideas expressed in the texts provided. Development of ideas and organization (2) o Student response addresses the prompt and provides effective development of the claim ...