Classical Thermodynamics Written by Jussi Eloranta
... At equilibrium a system is described by its thermodynamic variables. Thermodynamic equation of state introduces dependencies between the variables. An example of thermodynamic equation of state is the ideal gas law (P V = nRT ), which allows to express any of the five variables as a function of the ...
... At equilibrium a system is described by its thermodynamic variables. Thermodynamic equation of state introduces dependencies between the variables. An example of thermodynamic equation of state is the ideal gas law (P V = nRT ), which allows to express any of the five variables as a function of the ...
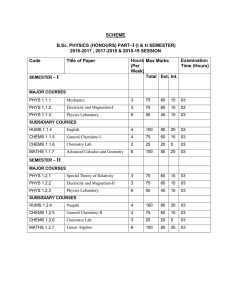
B.Sc.PHYSICS(HONOURS) PART I(SEMESTER
... particles of different mass, Cross-section of elastic scattering, Rutherford scattering. Dynamics of Rigid Bodies : Equation of motion, angular momentum and kinetic energy of a Rotating Body, Moment of Inertia and Radius of Gyration, Rotation of about fixed axes – time dependence of motion, cylinder ...
... particles of different mass, Cross-section of elastic scattering, Rutherford scattering. Dynamics of Rigid Bodies : Equation of motion, angular momentum and kinetic energy of a Rotating Body, Moment of Inertia and Radius of Gyration, Rotation of about fixed axes – time dependence of motion, cylinder ...
Work done and energy transfer
... The cyclist used the brakes to slow down and stop the bicycle. A constant braking force of 140 N stopped the bicycle in a distance of 24 m. Calculate the work done by the braking force to stop the bicycle. Give the unit. ...
... The cyclist used the brakes to slow down and stop the bicycle. A constant braking force of 140 N stopped the bicycle in a distance of 24 m. Calculate the work done by the braking force to stop the bicycle. Give the unit. ...
TEST-Chapters 2-4-Clayton Answer Section
... ____ 14. According to Newton's second law of motion, ____. a. F = m a c. F = p a b. F = m v d. F = p v ____ 15. For any object, the greater the force that's applied to it, the greater its ____ will be. a. acceleration c. inertia b. gravity d. velocity ____ 16. When a force is exerted on a bo ...
... ____ 14. According to Newton's second law of motion, ____. a. F = m a c. F = p a b. F = m v d. F = p v ____ 15. For any object, the greater the force that's applied to it, the greater its ____ will be. a. acceleration c. inertia b. gravity d. velocity ____ 16. When a force is exerted on a bo ...
Thermodynamics Chapter 4
... During a steady-flow process, the fluid flows through the control volume steadily, experiencing no change with time at a fixed position. The mass and energy content of the control volume remain constant during a steady-flow process. ...
... During a steady-flow process, the fluid flows through the control volume steadily, experiencing no change with time at a fixed position. The mass and energy content of the control volume remain constant during a steady-flow process. ...
EDEXCEL HIGHERS ENGINEERING THERMODYNAMICS H2 NQF
... cp = cv + R 5.5 LIQUIDS Since the volume of a liquid does not change much when heated or cooled, very little work is done against the surrounding pressure so it follows that cv and cp are for all intents and purposes the same and usually the heat transfer to a liquid is given as : Q = mc ∆T Where c ...
... cp = cv + R 5.5 LIQUIDS Since the volume of a liquid does not change much when heated or cooled, very little work is done against the surrounding pressure so it follows that cv and cp are for all intents and purposes the same and usually the heat transfer to a liquid is given as : Q = mc ∆T Where c ...
Polarization dependence of X-ray absorption spectra of
... incident photon energy is equal to the energy difference between the initial state and final unoccupied state. In the HEELS process, the number of scattered electron of the beam was counted. Since this electron excited the core electron to the same final states as of X-ray absorption, if the incomin ...
... incident photon energy is equal to the energy difference between the initial state and final unoccupied state. In the HEELS process, the number of scattered electron of the beam was counted. Since this electron excited the core electron to the same final states as of X-ray absorption, if the incomin ...
Thermochemistry
... “heat flows”—until the average molecular kinetic energies of the two bodies become the same, until the temperatures become equal. Heat, like work, describes energy in transit between a system and its surroundings. Not only can heat transfer cause a change in temperature but in some instances, it can ...
... “heat flows”—until the average molecular kinetic energies of the two bodies become the same, until the temperatures become equal. Heat, like work, describes energy in transit between a system and its surroundings. Not only can heat transfer cause a change in temperature but in some instances, it can ...
Chapter 1 Principles of Probability
... (a) The simplest way to solve this problem is to recall that when probabilities are independent, and you want the probability of events A and B, you can multiply them. When events are mutually exclusive and you want the probability of events A or B, you can add the probabilities. Therefore we try to ...
... (a) The simplest way to solve this problem is to recall that when probabilities are independent, and you want the probability of events A and B, you can multiply them. When events are mutually exclusive and you want the probability of events A or B, you can add the probabilities. Therefore we try to ...