Unit 9 Chemical Equations and Reactions Balancing Equations Notes
... Synthesis- _____________________ elements or compounds combine to form one compound. Decomposition- a _________________ compound decomposes into two or more elements or smaller compounds. Single Replacement- a metal will _________________ a less active metal in an ionic compound OR a nonmetal will r ...
... Synthesis- _____________________ elements or compounds combine to form one compound. Decomposition- a _________________ compound decomposes into two or more elements or smaller compounds. Single Replacement- a metal will _________________ a less active metal in an ionic compound OR a nonmetal will r ...
Paper - Asee peer logo
... stirred reactor (PSR) feeding the fuel/oxidant gas mixture. Such calculations can be performed for many simple fuel/oxidant feeds using detailed chemical reaction mechanisms available in the literature and the PSR program from Chemkin (6). Using pure component k, ρ, and Cp values, a simple mole fra ...
... stirred reactor (PSR) feeding the fuel/oxidant gas mixture. Such calculations can be performed for many simple fuel/oxidant feeds using detailed chemical reaction mechanisms available in the literature and the PSR program from Chemkin (6). Using pure component k, ρ, and Cp values, a simple mole fra ...
AP Chem II Instructor: Mr. Malasky Name Period ______ Due Date
... ____ 5. The value of ΔG˚ at 25˚C for the decomposition of gaseous sulfur dioxide to solid elemental sulfur and gaseous oxygen, SO2(g) → 2 S (s,rhombic) + O2(g) is __________ kJ/mol. A) +395.2 B) +269.9 C) -269.9 D) +300.4 E) -300.4 ____ 6. The value of ΔG˚ at 25˚C for the formation of POCl3 from it ...
... ____ 5. The value of ΔG˚ at 25˚C for the decomposition of gaseous sulfur dioxide to solid elemental sulfur and gaseous oxygen, SO2(g) → 2 S (s,rhombic) + O2(g) is __________ kJ/mol. A) +395.2 B) +269.9 C) -269.9 D) +300.4 E) -300.4 ____ 6. The value of ΔG˚ at 25˚C for the formation of POCl3 from it ...
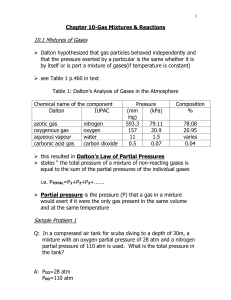
Chapter 10 Notes
... mixture with an oxygen partial pressure of 28 atm and a nitrogen partial pressure of 110 atm is used. What is the total pressure in the tank? ...
... mixture with an oxygen partial pressure of 28 atm and a nitrogen partial pressure of 110 atm is used. What is the total pressure in the tank? ...
284
... in pure chlorine gas, the iron “burns” according to the following (unbalanced) reaction: How many milligrams of iron(III) chloride result when 15.5 mg of iron is reacted with an excess of chlorine gas? Fe(s) + Cl2(g) FeCl(s) 32. You have probably seen images of a chef preparing a “flaming” dessert ...
... in pure chlorine gas, the iron “burns” according to the following (unbalanced) reaction: How many milligrams of iron(III) chloride result when 15.5 mg of iron is reacted with an excess of chlorine gas? Fe(s) + Cl2(g) FeCl(s) 32. You have probably seen images of a chef preparing a “flaming” dessert ...
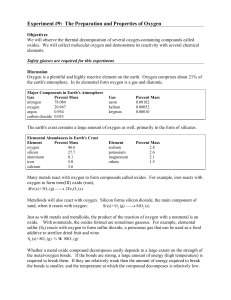
Experiment # 9 Properties of Oxygen
... oxide. With nonmetals, the oxides formed are sometimes gaseous. For example, elemental sulfur (S8) reacts with oxygen to form sulfur dioxide, a poisonous gas that can be used as a food additive to sterilize dried fruit and wine. S8 (s) +8O 2 (g) ¾ ¾ ® 8SO 2 (g) ...
... oxide. With nonmetals, the oxides formed are sometimes gaseous. For example, elemental sulfur (S8) reacts with oxygen to form sulfur dioxide, a poisonous gas that can be used as a food additive to sterilize dried fruit and wine. S8 (s) +8O 2 (g) ¾ ¾ ® 8SO 2 (g) ...
Impact of Welding Processes on Environment and Health
... Most of the small and medium enterprises (SMEs) have poor working conditions contributing to worker’s safety and health problem. Most welders who work in construction, factories, mining, manufacturing, metallurgy, railroad, petrochemical, ironworks, shipbuilding or steel industries, suffer from some ...
... Most of the small and medium enterprises (SMEs) have poor working conditions contributing to worker’s safety and health problem. Most welders who work in construction, factories, mining, manufacturing, metallurgy, railroad, petrochemical, ironworks, shipbuilding or steel industries, suffer from some ...
Group 2 Elements
... The presence of certain metal ions can be identified by noting the characteristic flame colour that results from burning. The colours for group 2 metal ions are: ...
... The presence of certain metal ions can be identified by noting the characteristic flame colour that results from burning. The colours for group 2 metal ions are: ...
Prof. Yong Lei & Stefan Bösemann (& Liying Liang)
... are separated by ion exchange membrane to split water into oxygen and hydrogen, respectively, at the same time. If two types of semiconductor are contacted directly to form a p-n junction, what would happen using the junction as photoelectrode for water splitting? What are the advantages of the junc ...
... are separated by ion exchange membrane to split water into oxygen and hydrogen, respectively, at the same time. If two types of semiconductor are contacted directly to form a p-n junction, what would happen using the junction as photoelectrode for water splitting? What are the advantages of the junc ...
thermal cutting and flame straightening
... steels is quenching, or hardening, from which the designation is derived. Thus, hardened steel is not actually tempered, which accounts for its different properties from quenched and tempered steel. Special care should be taken when flame straightening hardened steels. The maximum recommended temper ...
... steels is quenching, or hardening, from which the designation is derived. Thus, hardened steel is not actually tempered, which accounts for its different properties from quenched and tempered steel. Special care should be taken when flame straightening hardened steels. The maximum recommended temper ...
variable heat pattern burners
... furnace should have narrow or wide flame for best temperature uniformity. Try both with Variable Heat Pattern Burner, or select the best in-between combination. There is no need to build a second furnace to complete the experiment for your particular conditions--just make on-site adjustments. After ...
... furnace should have narrow or wide flame for best temperature uniformity. Try both with Variable Heat Pattern Burner, or select the best in-between combination. There is no need to build a second furnace to complete the experiment for your particular conditions--just make on-site adjustments. After ...
Unit 2
... b) When liquid phosphorus trichloride is added to water it reacts to form phosphous acid (sometimes called phosphorous acid) and hydrochloric acid. ...
... b) When liquid phosphorus trichloride is added to water it reacts to form phosphous acid (sometimes called phosphorous acid) and hydrochloric acid. ...
Unit 2
... b) When liquid phosphorus trichloride is added to water it reacts to form phosphous acid (sometimes called phosphorous acid) and hydrochloric acid. ...
... b) When liquid phosphorus trichloride is added to water it reacts to form phosphous acid (sometimes called phosphorous acid) and hydrochloric acid. ...
Unit 2
... b) When liquid phosphorus trichloride is added to water it reacts to form phosphous acid (sometimes called phosphorous acid) and hydrochloric acid. ...
... b) When liquid phosphorus trichloride is added to water it reacts to form phosphous acid (sometimes called phosphorous acid) and hydrochloric acid. ...
Unit 2
... b) When liquid phosphorus trichloride is added to water it reacts to form phosphous acid (sometimes called phosphorous acid) and hydrochloric acid. ...
... b) When liquid phosphorus trichloride is added to water it reacts to form phosphous acid (sometimes called phosphorous acid) and hydrochloric acid. ...
examples of chemical and physical reactions.
... things burn, they react with ________________ gas in the air. The products formed are called ______. _____________ gas relights a _____________ splint. 2. You have burned magnesium in air. Remember that air is a mixture of gases; so what do you think will happen if we burn magnesium in pure oxygen g ...
... things burn, they react with ________________ gas in the air. The products formed are called ______. _____________ gas relights a _____________ splint. 2. You have burned magnesium in air. Remember that air is a mixture of gases; so what do you think will happen if we burn magnesium in pure oxygen g ...
gerresheimer_nfpa_design_q... 250KB Jan 21 2016 09:21:29 AM
... flames. Because they are exceptionally hot, these flames can damage burners, ruin work in process and furnace internals, and even destroy refractory insulation that was ...
... flames. Because they are exceptionally hot, these flames can damage burners, ruin work in process and furnace internals, and even destroy refractory insulation that was ...
quote - AgriSETA
... Set up oxygen-fuel gas welding equipment including light up, adjustment of gas pressures and shut down procedures. Differentiate gas welding consumables. Prepare material for gas welding. Gas weld work pieces incidental to the trade. Assemble gas cutting and heating equipment, including light up and ...
... Set up oxygen-fuel gas welding equipment including light up, adjustment of gas pressures and shut down procedures. Differentiate gas welding consumables. Prepare material for gas welding. Gas weld work pieces incidental to the trade. Assemble gas cutting and heating equipment, including light up and ...
Dissolved acetylene. - Linde North America
... acetylene can still explosively decompose. The ignition temperatures of acetylene, mixtures of acetylene and air, and mixtures of acetylene with oxygen will vary according to composition, pressure, water vapour content and initial temperature. As a typical example, mixtures containing 30% acetylene ...
... acetylene can still explosively decompose. The ignition temperatures of acetylene, mixtures of acetylene and air, and mixtures of acetylene with oxygen will vary according to composition, pressure, water vapour content and initial temperature. As a typical example, mixtures containing 30% acetylene ...
Word and Skeleton Equations Practice (ws Fall 2010)
... Write the WORD and SKELETON equations for each chemical reaction. Indicate the state of each reactant and product in the skeleton equation. Remember that the following seven elements are diatomic: hydrogen, H2(g); nitrogen, N2(g); oxygen O2(g); fluorine, F2(g); chlorine Cl2(g); bromine, Br2(l); and ...
... Write the WORD and SKELETON equations for each chemical reaction. Indicate the state of each reactant and product in the skeleton equation. Remember that the following seven elements are diatomic: hydrogen, H2(g); nitrogen, N2(g); oxygen O2(g); fluorine, F2(g); chlorine Cl2(g); bromine, Br2(l); and ...
products
... •Uzan Automotive meets all requirements of the ISO/TS:16949 quality management system scrupulously and is certified by BV. A laboratory, for raising the efficiency of quality control to higher levels, is available. • In our laboratory the mechanical and chemical tests of plastic-metal components are ...
... •Uzan Automotive meets all requirements of the ISO/TS:16949 quality management system scrupulously and is certified by BV. A laboratory, for raising the efficiency of quality control to higher levels, is available. • In our laboratory the mechanical and chemical tests of plastic-metal components are ...
BURNERS AND FLAMES:
... A ball that has been kicked up the stairs will fall back down the steps giving back the energy that was added to it. Likewise, an atom that is excited is unstable and will immediately give up the energy added and return to the ground state. When a ball is kicked up the stairs there are options of ho ...
... A ball that has been kicked up the stairs will fall back down the steps giving back the energy that was added to it. Likewise, an atom that is excited is unstable and will immediately give up the energy added and return to the ground state. When a ball is kicked up the stairs there are options of ho ...
~The different types of gases~ Oxygen (O2) The most common
... sustain life. They are inert gases which do not react. These gases can only be combined with other chemical substances with great difficulty. This extreme inertness quality in these gases makes them very valuable for certain applications. ...
... sustain life. They are inert gases which do not react. These gases can only be combined with other chemical substances with great difficulty. This extreme inertness quality in these gases makes them very valuable for certain applications. ...
Characteristics of Gases Pressure Gas Laws The Ideal
... connected to a 5.00-L tank that is completely empty (evacuated), and a valve connects the two tanks., If the valve is opened, determine the total pressure in this two-tank system after the nitrogen stops flowing. No temperature change occurs in the process. ...
... connected to a 5.00-L tank that is completely empty (evacuated), and a valve connects the two tanks., If the valve is opened, determine the total pressure in this two-tank system after the nitrogen stops flowing. No temperature change occurs in the process. ...
Oxy-fuel welding and cutting

Oxy-fuel welding (commonly called oxyacetylene welding, oxy welding, or gas welding in the U.S.) and oxy-fuel cutting are processes that use fuel gases and oxygen to weld and cut metals, respectively. French engineers Edmond Fouché and Charles Picard became the first to develop oxygen-acetylene welding in 1903. Pure oxygen, instead of air, is used to increase the flame temperature to allow localized melting of the workpiece material (e.g. steel) in a room environment. A common propane/air flame burns at about 2,250 K (1,980 °C; 3,590 °F), a propane/oxygen flame burns at about 2,526 K (2,253 °C; 4,087 °F), and an acetylene/oxygen flame burns at about 3,773 K (3,500 °C; 6,332 °F).Oxy-fuel is one of the oldest welding processes, besides forge welding. Still used in industry, in recent decades it has been less widely utilized in industrial applications as other specifically devised technologies have been adopted. It is still widely used for welding pipes and tubes, as well as repair work. It is also frequently well-suited, and favored, for fabricating some types of metal-based artwork. As well, oxy-fuel has an advantage over electric welding and cutting processes in situations where accessing electricity (e.g., via an extension cord or portable generator) would present difficulties; it is more self-contained, and, hence, often more portable.In oxy-fuel welding, a welding torch is used to weld metals. Welding metal results when two pieces are heated to a temperature that produces a shared pool of molten metal. The molten pool is generally supplied with additional metal called filler. Filler material depends upon the metals to be welded.In oxy-fuel cutting, a torch is used to heat metal to its kindling temperature. A stream of oxygen is then trained on the metal, burning it into a metal oxide that flows out of the kerf as slag.Torches that do not mix fuel with oxygen (combining, instead, atmospheric air) are not considered oxy-fuel torches and can typically be identified by a single tank (Oxy-fuel cutting requires two isolated supplies, fuel and oxygen). Most metals cannot be melted with a single-tank torch. As such, single-tank torches are typically used only for soldering and brazing, rather than welding.