BASIC CALORIMETRY SET
... When steam condenses in cool water, heat energy is released into the water in two ways. One, the latent heat of vaporization is released. With this release of heat, the steam is converted into water, but the newly converted water is still at boiling temperature, 100 °C. Second, the newly converted w ...
... When steam condenses in cool water, heat energy is released into the water in two ways. One, the latent heat of vaporization is released. With this release of heat, the steam is converted into water, but the newly converted water is still at boiling temperature, 100 °C. Second, the newly converted w ...
Coolant Filters - CARQUEST Auto Parts
... To select the right filter, you must know the type of coolant used in your vehicle. If unsure, look for an information label inside the front door. Coolant Filters without Chemicals Use coolants filters without chemicals if: • you are using an Extended Life Coolant (ELC) • you are using a Standard L ...
... To select the right filter, you must know the type of coolant used in your vehicle. If unsure, look for an information label inside the front door. Coolant Filters without Chemicals Use coolants filters without chemicals if: • you are using an Extended Life Coolant (ELC) • you are using a Standard L ...
Solid-state optical refrigeration to sub-100 Kelvin regime www.nature.com/scientificreports Seth D. Melgaard
... passes at 90 K. Consequently, provisions were made for the remaining pump light (≈30 W) to safely exit the cavity through the original entrance hole when the crystal is cold and under vacuum. Furthermore, ensuring that all of the 22 passes traverse the crystal becomes a challenge. The alignment is p ...
... passes at 90 K. Consequently, provisions were made for the remaining pump light (≈30 W) to safely exit the cavity through the original entrance hole when the crystal is cold and under vacuum. Furthermore, ensuring that all of the 22 passes traverse the crystal becomes a challenge. The alignment is p ...
Carnot`s Perfect Heat Engine: The Second Law of
... work into electrical energy. Heat exchange is used to generate steam, in part to avoid contamination of the generators with radioactivity. Two turbines are used because this is less expensive than operating a single generator that produces the same amount of electrical energy. The steam is condensed ...
... work into electrical energy. Heat exchange is used to generate steam, in part to avoid contamination of the generators with radioactivity. Two turbines are used because this is less expensive than operating a single generator that produces the same amount of electrical energy. The steam is condensed ...
Hybrid solar Desiccant Cooling System
... Hybrid solar Desiccant Cooling System Using excess summer heat from solar collectors to drive desiccant cooling systems is often proposed. A two wheel desiccant system using solar heat for desiccant regeneration is typically discussed. The two wheel system uses a desiccant wheel that is “matched” wi ...
... Hybrid solar Desiccant Cooling System Using excess summer heat from solar collectors to drive desiccant cooling systems is often proposed. A two wheel desiccant system using solar heat for desiccant regeneration is typically discussed. The two wheel system uses a desiccant wheel that is “matched” wi ...
Solid Target Engineering
... A critical heat transfer rate exists, (~1 MW/m2), at which surface boiling occurs Even below this limit there is enough cooling power to remove heat dissipated by a single beam pulse in ~2 seconds ...
... A critical heat transfer rate exists, (~1 MW/m2), at which surface boiling occurs Even below this limit there is enough cooling power to remove heat dissipated by a single beam pulse in ~2 seconds ...
Heat Engines
... Because they spin at such high speeds and because of the high operating temperatures, designing and manufacturing gas turbines is a tough problem from both the engineering and materials standpoint. Gas turbines also tend to use more fuel when they are idling, and they prefer a constant rather than a ...
... Because they spin at such high speeds and because of the high operating temperatures, designing and manufacturing gas turbines is a tough problem from both the engineering and materials standpoint. Gas turbines also tend to use more fuel when they are idling, and they prefer a constant rather than a ...
Carnot Cycle. Heat Engines. Refrigerators.
... second laws we get a very simple, explicit upper limit on the efficiency of an engine! The first law says you can not get efficiency greater than unity. The second law forbids an efficiency of unity – not all energy absorbed as heat can be converted into work. Better efficiency comes by making the r ...
... second laws we get a very simple, explicit upper limit on the efficiency of an engine! The first law says you can not get efficiency greater than unity. The second law forbids an efficiency of unity – not all energy absorbed as heat can be converted into work. Better efficiency comes by making the r ...
Engine Lubrication And Cooling
... • Occurs when engine parts that are operating at high temperatures are quickly cooled. • Idle power, high airspeed, cool or cold air. • Some parts are cooled much more rapidly then others. These parts shrink in size faster then the warmer, surrounding metals. ...
... • Occurs when engine parts that are operating at high temperatures are quickly cooled. • Idle power, high airspeed, cool or cold air. • Some parts are cooled much more rapidly then others. These parts shrink in size faster then the warmer, surrounding metals. ...
Theory
... Here, CDA ≈ 1.005 kJ/(kg dry air)·K and CW ≈ 1.88 kJ/(kg water vapor)·K are the heat capacities of dry air and water vapor. The thermodynamic properties of humid air are summarized in the chart shown in Figure 2-1. Wet bulb temperature Twb(T,h) of air at temperature T and humidity h is the temperatu ...
... Here, CDA ≈ 1.005 kJ/(kg dry air)·K and CW ≈ 1.88 kJ/(kg water vapor)·K are the heat capacities of dry air and water vapor. The thermodynamic properties of humid air are summarized in the chart shown in Figure 2-1. Wet bulb temperature Twb(T,h) of air at temperature T and humidity h is the temperatu ...
reduction of parasitic losses in heavy
... •A lower restriction means that pumping losses are lower and the coolant pump consumes less power •PID or other control strategies can be employed to help keep coolant temperature to within +/- 2% of the set temperature •Reductions in fuel consumption of 2% – 5%, 20% less CO and 10% less HC emission ...
... •A lower restriction means that pumping losses are lower and the coolant pump consumes less power •PID or other control strategies can be employed to help keep coolant temperature to within +/- 2% of the set temperature •Reductions in fuel consumption of 2% – 5%, 20% less CO and 10% less HC emission ...
cooling info sheet
... A major difference between the conventional vapor compression cycle and the absorption cycle is the refrigerant used. Chlorofluorocarbons (CFCs) have been the most popular refrigerants for mechanical refrigeration systems for decades; however, distilled water is an adequate refrigerant in most large ...
... A major difference between the conventional vapor compression cycle and the absorption cycle is the refrigerant used. Chlorofluorocarbons (CFCs) have been the most popular refrigerants for mechanical refrigeration systems for decades; however, distilled water is an adequate refrigerant in most large ...
Document
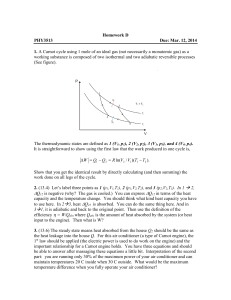
... 2. (13.4) Let’s label three points as 1 (p1,V1,T1), 2 (p1,V2,T2), and 3 (p2,V2,T3). In 1 2, ∆Q12 is negative (why? The gas is cooled.) You can express ∆Q12 in terms of the heat capacity and the temperature change. You should think what kind heat capacity you have to use here. In 23, heat ∆Q23 is ...
... 2. (13.4) Let’s label three points as 1 (p1,V1,T1), 2 (p1,V2,T2), and 3 (p2,V2,T3). In 1 2, ∆Q12 is negative (why? The gas is cooled.) You can express ∆Q12 in terms of the heat capacity and the temperature change. You should think what kind heat capacity you have to use here. In 23, heat ∆Q23 is ...
(Bio)Diesel Battery Chargers . . . DC Amps at 12/24
... refrigeration compressor, alternator output is reduced automatically to free up some power for the other load. The alternative, used by others, is to shut the alternator completely off. Typically, the DBC can produce 50–70% of it’s output Amps while powering a second load. • Remote radiator with Ene ...
... refrigeration compressor, alternator output is reduced automatically to free up some power for the other load. The alternative, used by others, is to shut the alternator completely off. Typically, the DBC can produce 50–70% of it’s output Amps while powering a second load. • Remote radiator with Ene ...
Ch 14.3 PPT - Using Heat
... because four strokes take place for each cycle of the piston. – The four strokes are: • intake • compression • power • exhaust • Internal-combustion engines always generate heat. – Friction and other forces cause much of the energy to be lost to the atmosphere as heat. ...
... because four strokes take place for each cycle of the piston. – The four strokes are: • intake • compression • power • exhaust • Internal-combustion engines always generate heat. – Friction and other forces cause much of the energy to be lost to the atmosphere as heat. ...
20 - Cabrillo College
... Due: See Website for due dates Chapter 21: Entropy and the Second Law of Thermodynamics Problems: 20, 25, 35, 37, 43 Question A A pot is half-filled with water, and a lid is placed on the pot, forming a tight seal so that no water vapor can escape. The pot is heated on a stove, forming water vapor i ...
... Due: See Website for due dates Chapter 21: Entropy and the Second Law of Thermodynamics Problems: 20, 25, 35, 37, 43 Question A A pot is half-filled with water, and a lid is placed on the pot, forming a tight seal so that no water vapor can escape. The pot is heated on a stove, forming water vapor i ...