
2 The Real Scalar Field
... union of Hilbert spaces is known as “Fock space”. Later we will need to consider particles with non-zero spin, so that as well as denoting their momenta will will also need to denote the “helicity” of the particles, the component of spin in the direction of their momenta. ...
... union of Hilbert spaces is known as “Fock space”. Later we will need to consider particles with non-zero spin, so that as well as denoting their momenta will will also need to denote the “helicity” of the particles, the component of spin in the direction of their momenta. ...
SPS1: Students will investigate our current understanding of the
... Electrons are equal to the number of __PROTONS__ in a neutral atom. o The nucleus is made up of these 2 particles ___PROTONS___ and __NEUTRONS__. o The electrons are located in this region of the atom, known as the _ELECTRON CLOUD_. o The electron cloud is divided in to energy levels or energy she ...
... Electrons are equal to the number of __PROTONS__ in a neutral atom. o The nucleus is made up of these 2 particles ___PROTONS___ and __NEUTRONS__. o The electrons are located in this region of the atom, known as the _ELECTRON CLOUD_. o The electron cloud is divided in to energy levels or energy she ...
Physics-ATAR-Unit-2
... documents that are mandated. Examiners are required to address the mandated statements only. The STAWA Depth & Breadth of Content documents are a great example of teachers helping teachers for the benefit of all students. ...
... documents that are mandated. Examiners are required to address the mandated statements only. The STAWA Depth & Breadth of Content documents are a great example of teachers helping teachers for the benefit of all students. ...
No Slide Title
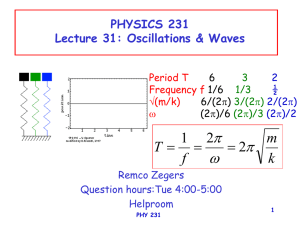
... of 2m between a maximum and the nearest minimum and vertical height of 2m. If it moves with 1m/s, what is its: a) amplitude b) period c) frequency ...
... of 2m between a maximum and the nearest minimum and vertical height of 2m. If it moves with 1m/s, what is its: a) amplitude b) period c) frequency ...
1. dia
... En ). The electrons with given n values are forming shells which are named with K, L, M, etc. letters. There can be more other states inside a shell which states are determined by the orbital quantum number. Bohr had predicted the positions of orbits with amazing accuracy but did not take count that ...
... En ). The electrons with given n values are forming shells which are named with K, L, M, etc. letters. There can be more other states inside a shell which states are determined by the orbital quantum number. Bohr had predicted the positions of orbits with amazing accuracy but did not take count that ...
Science 2nd prep 2nd term final exercise Important Laws: 1
... 8-the longest wavelength is for the ………….. color of the spectrum seven colors. 9-The German scientist …………………… proved that the energy of light waves is composed of energy quanta known as ………………………. 10-Photon energy = plank's constant × ………………………… 11-Objects can't be seen behind the ……………………. medium. ...
... 8-the longest wavelength is for the ………….. color of the spectrum seven colors. 9-The German scientist …………………… proved that the energy of light waves is composed of energy quanta known as ………………………. 10-Photon energy = plank's constant × ………………………… 11-Objects can't be seen behind the ……………………. medium. ...
Time Dilation - TuHS Physics Homepage
... Black Body Radiation -> Quantum mechanics Velocity of light With Respect to ether Maxwell’s Equations… ...
... Black Body Radiation -> Quantum mechanics Velocity of light With Respect to ether Maxwell’s Equations… ...
powerpoint - University of Illinois at Urbana
... been developed and made available online by work supported jointly by University of Illinois, the National Science Foundation under Grant CHE-1118616 (CAREER), and the Camille & Henry Dreyfus Foundation, Inc. through the Camille Dreyfus Teacher-Scholar program. Any opinions, findings, and conclusion ...
... been developed and made available online by work supported jointly by University of Illinois, the National Science Foundation under Grant CHE-1118616 (CAREER), and the Camille & Henry Dreyfus Foundation, Inc. through the Camille Dreyfus Teacher-Scholar program. Any opinions, findings, and conclusion ...
Physics 322 Final Exam Study Guide (2015) [Pages 4 Only]
... a. Know the basic characteristics of the hydrogen spectra and why it strongly suggests that the allowed energy levels of the electron in the hydrogen atom are quantized. b. Given the expression for the energy levels of the hydrogen atom, be able to compute the wavelength of a photon emitted/absorbed ...
... a. Know the basic characteristics of the hydrogen spectra and why it strongly suggests that the allowed energy levels of the electron in the hydrogen atom are quantized. b. Given the expression for the energy levels of the hydrogen atom, be able to compute the wavelength of a photon emitted/absorbed ...
Goal: To understand how light can be used to
... spectra are created by gasses of a few atoms and few molecules. • Therefore, most Absorption spectra in Astronomy consist of thin dark lines, or thin areas where most of the energy is removed at just a few wavelengths. • Any “thin” substance which is in front of a continuous light source will produc ...
... spectra are created by gasses of a few atoms and few molecules. • Therefore, most Absorption spectra in Astronomy consist of thin dark lines, or thin areas where most of the energy is removed at just a few wavelengths. • Any “thin” substance which is in front of a continuous light source will produc ...
Missing Link
... 6. Such that if the atom is in that box and if the photon passes in that arm, absorption occurs with probability 1. 7. Two Hardy atoms can be entangled ...
... 6. Such that if the atom is in that box and if the photon passes in that arm, absorption occurs with probability 1. 7. Two Hardy atoms can be entangled ...
the Bohr`s atom model - Latin-American Journal of Physics Education
... spectral lines can be expressed by means of the formula 1/λ=RH(1/221/n2), where n is an integer number and RH=109677.5810 cm-1 is the Rydberg’s constant, called so in honour to which generalized in 1889 the above expression to the whole spectrum writing it in the form 1/λ=RH(1/n´2-1/n2), with n´
... spectral lines can be expressed by means of the formula 1/λ=RH(1/221/n2), where n is an integer number and RH=109677.5810 cm-1 is the Rydberg’s constant, called so in honour to which generalized in 1889 the above expression to the whole spectrum writing it in the form 1/λ=RH(1/n´2-1/n2), with n´
Modified Einsteinian Dynamics(MOED): Discovery of a
... According to Special Relativity when a object approaches the speed of light, time slows down, length contraction (object becomes thinner) and more massive. This is a perfect analogy to the electromagnetic spectrum. Going from lower to higher wavelength the wavelength becomes shorter and the energy ...
... According to Special Relativity when a object approaches the speed of light, time slows down, length contraction (object becomes thinner) and more massive. This is a perfect analogy to the electromagnetic spectrum. Going from lower to higher wavelength the wavelength becomes shorter and the energy ...
Essentials of Modern Physics
... (b) solve simple algebraic equations. Most relevant equations are linear but some may involve inverse and inverse square relationships. Linear simultaneous equations and the use of the formula to obtain the solutions of quadratic equations are included (c) substitute physical quantities into physica ...
... (b) solve simple algebraic equations. Most relevant equations are linear but some may involve inverse and inverse square relationships. Linear simultaneous equations and the use of the formula to obtain the solutions of quadratic equations are included (c) substitute physical quantities into physica ...
Chapters 7, 8, 9 notes - SLCUSD Staff Directory
... simply cannot state how the electron is moving around the nucleus because it is a _____ and a ________ at the same time. We can give the most probabilistic region of space over which the electron can exist. We call this the _______________. The mathematics of the Heisenberg Uncertainty Principle is ...
... simply cannot state how the electron is moving around the nucleus because it is a _____ and a ________ at the same time. We can give the most probabilistic region of space over which the electron can exist. We call this the _______________. The mathematics of the Heisenberg Uncertainty Principle is ...










![Physics 322 Final Exam Study Guide (2015) [Pages 4 Only]](http://s1.studyres.com/store/data/007969504_1-e89a1630d6e27466a3e33b80f7e23b58-300x300.png)











