Chemistry CPA Mid-Term Exam Study Guide January 2012
... Describe the trends in first ionization energy within groups and across periods in the periodic table. Provide examples. Positive ions are smaller than the atoms from which they are formed, but negative ions are larger than the atoms from which they are formed. Explain why this is so. Describe ...
... Describe the trends in first ionization energy within groups and across periods in the periodic table. Provide examples. Positive ions are smaller than the atoms from which they are formed, but negative ions are larger than the atoms from which they are formed. Explain why this is so. Describe ...
cavity design - lombarda.web.cern.ch
... in a periodic structure of period l the solution of the wave equation is such that --- for a given frequency and mode of oscillation, and in absence of losses, the solution may differ from a period to the next only by a factor like jk z l ...
... in a periodic structure of period l the solution of the wave equation is such that --- for a given frequency and mode of oscillation, and in absence of losses, the solution may differ from a period to the next only by a factor like jk z l ...
week 1
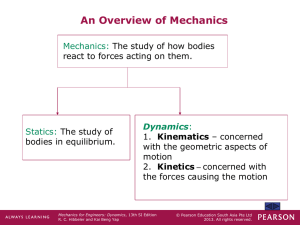
... Velocity is a measure of the rate of change in the position of a particle. It is a vector quantity (it has both magnitude and direction). The magnitude of the velocity is called speed, with units of m/s or ft/s. The average velocity of a particle during a time interval t is vavg = r / t The insta ...
... Velocity is a measure of the rate of change in the position of a particle. It is a vector quantity (it has both magnitude and direction). The magnitude of the velocity is called speed, with units of m/s or ft/s. The average velocity of a particle during a time interval t is vavg = r / t The insta ...
Momentum
... Impulse helps us when dealing with forces that occur over a short period of time. A golf ball being hit A water balloon catching contest ? It should be noted that Impulse I = F x t Can be represented by the area under a graph of force vs time. ...
... Impulse helps us when dealing with forces that occur over a short period of time. A golf ball being hit A water balloon catching contest ? It should be noted that Impulse I = F x t Can be represented by the area under a graph of force vs time. ...
V. Angular momentum
... between the sphere and the surface is at rest the frictional force is the static frictional force. 3. Work done by frictional force = 0 the point of contact is at rest (static friction). ...
... between the sphere and the surface is at rest the frictional force is the static frictional force. 3. Work done by frictional force = 0 the point of contact is at rest (static friction). ...
Chapter 9 Problems - University of Colorado Colorado Springs
... makes an elastic head-on collision with the nucleus of a carbon atom initially at rest. (a) What fraction of the neutron's kinetic energy is transferred to the carbon nucleus? (b) If the initial kinetic energy of the neutron is 1.60 10–13 J, find its final kinetic energy and the kinetic energy of ...
... makes an elastic head-on collision with the nucleus of a carbon atom initially at rest. (a) What fraction of the neutron's kinetic energy is transferred to the carbon nucleus? (b) If the initial kinetic energy of the neutron is 1.60 10–13 J, find its final kinetic energy and the kinetic energy of ...
Quantum Mechanics of Many-Electrons Systems and the Theories of
... Independent particle models are extremely useful for interpreting and rationalizing the results of quantum mechanical calculations. In fact, ...
... Independent particle models are extremely useful for interpreting and rationalizing the results of quantum mechanical calculations. In fact, ...
Arrangement of Electrons in Atoms (Chapter 4) Notes
... Electrons cannot have just any energy; only orbits of certain radii having CERTAIN energies are permitted. Thus, when an electron absorbs quanta of energy, it will cause them to jump away from the nucleus to a higher orbit (energy level or n) and when the electron falls from a high orbit to a lower ...
... Electrons cannot have just any energy; only orbits of certain radii having CERTAIN energies are permitted. Thus, when an electron absorbs quanta of energy, it will cause them to jump away from the nucleus to a higher orbit (energy level or n) and when the electron falls from a high orbit to a lower ...