Potassium trans
... 0.5 g of activated charcoal was added to a solution of potassium oxalate (K2C2O4•H2O, 6.3 g, 34 mmol) in 10 mL of warm water (B). The solutions (A) and (B) were mixed together and air was bubbled through the mixture for 17 hours. The activated charcoal was removed and the red-purple filtrate was eva ...
... 0.5 g of activated charcoal was added to a solution of potassium oxalate (K2C2O4•H2O, 6.3 g, 34 mmol) in 10 mL of warm water (B). The solutions (A) and (B) were mixed together and air was bubbled through the mixture for 17 hours. The activated charcoal was removed and the red-purple filtrate was eva ...
exercise on Chapter 13 - Louisiana Tech University
... Dynamic Equilibrium: The reactants and products will interchange constantly, however maintaining same concentrations of reactants and products. This change maintains a constant concentration of reactants and products. Homogenous equilibrium: where reactants and products are in same phase. E.g. 3H2(g ...
... Dynamic Equilibrium: The reactants and products will interchange constantly, however maintaining same concentrations of reactants and products. This change maintains a constant concentration of reactants and products. Homogenous equilibrium: where reactants and products are in same phase. E.g. 3H2(g ...
MALTA
... B.Sc. (Honours) The B.Sc. (Honours) degree of the Faculty of Science is a 4 year full-time course in two science subjects. There are a number of subject combinations that a student can chose to take. Chemistry has been combined with Biology, Physics, Mathematics and Computer Science. The most popula ...
... B.Sc. (Honours) The B.Sc. (Honours) degree of the Faculty of Science is a 4 year full-time course in two science subjects. There are a number of subject combinations that a student can chose to take. Chemistry has been combined with Biology, Physics, Mathematics and Computer Science. The most popula ...
F.Y. B.Sc. - Chemistry
... 4. Integration- Rules of integration, Algebraic and exponential functions and problems. Aims and objectives: After studying this chapter, students should be able to 1. Define logarithm. 2. State all rules of logarithms. 3. Convert negative mantissa into positive (i.e. convert natural to logarithm an ...
... 4. Integration- Rules of integration, Algebraic and exponential functions and problems. Aims and objectives: After studying this chapter, students should be able to 1. Define logarithm. 2. State all rules of logarithms. 3. Convert negative mantissa into positive (i.e. convert natural to logarithm an ...
Document
... 1. The concentrations of the reacting species in the condensed phase are expressed in M. In the gaseous phase, the concentrations can be expressed in M or in atm. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium ...
... 1. The concentrations of the reacting species in the condensed phase are expressed in M. In the gaseous phase, the concentrations can be expressed in M or in atm. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium ...
Molecules, Compounds, and Chemical Equations
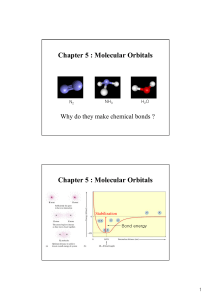
... other. Instead some electrons are shared between the two bonding atoms. The shared electrons interact with the nuclei of both atoms, lowering their potential energy through electrostatic interactions with the nuclei. The resulting bond is called a covalent bond. We can understand the stability of a ...
... other. Instead some electrons are shared between the two bonding atoms. The shared electrons interact with the nuclei of both atoms, lowering their potential energy through electrostatic interactions with the nuclei. The resulting bond is called a covalent bond. We can understand the stability of a ...
unit iv – stoichiometry 1
... VIII. Laws of Chemical Combination A) Law of Definite Proportions – different samples of the same compound will always contain the same proportion of elements by mass B) Law of Multiple Proportions – if two elements can combine to form more than one compound, the masses of one element that combine w ...
... VIII. Laws of Chemical Combination A) Law of Definite Proportions – different samples of the same compound will always contain the same proportion of elements by mass B) Law of Multiple Proportions – if two elements can combine to form more than one compound, the masses of one element that combine w ...
Unusual deprotonated alkynyl hydrogen bonding in metal
... vector a and the Cu [1̅10] direction is 20.5 ± 0.4°. The angle β specifying the azimuthal orientation of the molecules relative to the Cu[1̅1̅2] direction is obtained by superimposing a HYPERCHEM optimized ball-and-stick model (gas phase) onto the STM image, which gives βE = 10 ± 2°. STM measurement ...
... vector a and the Cu [1̅10] direction is 20.5 ± 0.4°. The angle β specifying the azimuthal orientation of the molecules relative to the Cu[1̅1̅2] direction is obtained by superimposing a HYPERCHEM optimized ball-and-stick model (gas phase) onto the STM image, which gives βE = 10 ± 2°. STM measurement ...
Improved Transparency-Nonlinearity Trade-Off with Boroxine
... geometries and electronic properties were obtained using the Gaussian 98 package65 at the density functional theory (DFT) B3LYP66 level using the 6-31G* basis set. Once we had verified that all compounds of the series lead to planar conformations, C3h symmetry was imposed for molecules B1 and B2 whe ...
... geometries and electronic properties were obtained using the Gaussian 98 package65 at the density functional theory (DFT) B3LYP66 level using the 6-31G* basis set. Once we had verified that all compounds of the series lead to planar conformations, C3h symmetry was imposed for molecules B1 and B2 whe ...
Stoichiometry and the Mole
... 1. The reactant that gave you the lowest answer is the LIMITING REACTANT. 2. The other reactant (s) are in EXCESS. 3. To find the amount of excess, subtract the amount used from the given amount. ...
... 1. The reactant that gave you the lowest answer is the LIMITING REACTANT. 2. The other reactant (s) are in EXCESS. 3. To find the amount of excess, subtract the amount used from the given amount. ...
Chemical Equilibrium
... The equilibrium constant of a reaction that has been multiplied by a number is the equilibrium constant raised to a power equal to that number The equilibrium constant for a net reaction of two or more steps is the product of the constants of the individual steps ...
... The equilibrium constant of a reaction that has been multiplied by a number is the equilibrium constant raised to a power equal to that number The equilibrium constant for a net reaction of two or more steps is the product of the constants of the individual steps ...
chemistry worksheet # 2: the mole as a unit of mass
... One important property of a mole is that it means a definite number of particles just like a dozen means a number of particles. While a dozen is only 12 particles a mole is a much larger number—6.02 x 1023 particles. Elements generally exist as the particles we call atoms. A mole of carbon contains ...
... One important property of a mole is that it means a definite number of particles just like a dozen means a number of particles. While a dozen is only 12 particles a mole is a much larger number—6.02 x 1023 particles. Elements generally exist as the particles we call atoms. A mole of carbon contains ...
Unit 1 Practice Problems
... A compound with the percent composition shown next has a molar mass of 60.10 g/mol. Determine its molecular formula. C, 39.97% H, 13.41% N, 46.62% ...
... A compound with the percent composition shown next has a molar mass of 60.10 g/mol. Determine its molecular formula. C, 39.97% H, 13.41% N, 46.62% ...
IJCA 41A(8) 1629-1633
... exchange interaction is negligible, whereas when the value of G is less than four, a considerable interaction is indicated in solid complex. The calculated G values are given in Table 2. The fraction a 2which is taken as a measure of ...
... exchange interaction is negligible, whereas when the value of G is less than four, a considerable interaction is indicated in solid complex. The calculated G values are given in Table 2. The fraction a 2which is taken as a measure of ...
M for Moles - Shop
... the moles calculations. A chemical equation shows a complete summary description of a chemical reaction. However, before the equation can be used, it must be properly balanced. Balancing a chemical equation involves two parts: (1) Balance the individual chemical formula of a compound. (2) Balance th ...
... the moles calculations. A chemical equation shows a complete summary description of a chemical reaction. However, before the equation can be used, it must be properly balanced. Balancing a chemical equation involves two parts: (1) Balance the individual chemical formula of a compound. (2) Balance th ...
Name: Period:______ Let`s make some sandwiches! Introduction: If
... Name:______________________ Period:_________ Let’s make some sandwiches! Introduction: If a sandwich shop runs out of bread, the shop closes down. No more sandwiches can be fully made without ordering more bread from a bakery. A similar thing happens in a chemical reaction. If there are fixed amount ...
... Name:______________________ Period:_________ Let’s make some sandwiches! Introduction: If a sandwich shop runs out of bread, the shop closes down. No more sandwiches can be fully made without ordering more bread from a bakery. A similar thing happens in a chemical reaction. If there are fixed amount ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Sample Exercise 3.19 Calculating the Amount of Product Formed from a Limiting Reactant Consider the following reaction that occurs in a fuel cell: 2 H2(g) + O2 (g) → 2 H2O (g) This reaction, properly done, produces energy in the form of electricity and water. Suppose a fuel cell is set up with 150 ...
... Sample Exercise 3.19 Calculating the Amount of Product Formed from a Limiting Reactant Consider the following reaction that occurs in a fuel cell: 2 H2(g) + O2 (g) → 2 H2O (g) This reaction, properly done, produces energy in the form of electricity and water. Suppose a fuel cell is set up with 150 ...
Sign of enthalpy changes Exothermic vs endothermic Acid
... Estimate the molar enthalpy of binding binding. ...
... Estimate the molar enthalpy of binding binding. ...
Empirical Formula, Molecular Formula, Percent Composition
... a) & b) As always start off with a balanced chemical equation which is given to us, so that work is done. Compare both of your reactant amounts to the same product in this case Al2(SO4)3. Then find out how much products will be produced from each individual reactant. Whichever reactant yields the le ...
... a) & b) As always start off with a balanced chemical equation which is given to us, so that work is done. Compare both of your reactant amounts to the same product in this case Al2(SO4)3. Then find out how much products will be produced from each individual reactant. Whichever reactant yields the le ...
Stoichiometry - AaronFreeman
... 1. Use Stoichiometry to find mass of C, H, O 2. Mass (g) to moles of C, H, O ...
... 1. Use Stoichiometry to find mass of C, H, O 2. Mass (g) to moles of C, H, O ...
Ribosomal crystallography: a flexible nucleotide anchoring tRNA
... derivatives that are too short to reach the upper rims of the PTC cavity; or when key constituents of the upper rims, responsible for the remote interactions (e.g., the tip of H69) are disordered, as is the case of crystal structure of H50S [5,6]. It seems, therefore, that the global localization of ...
... derivatives that are too short to reach the upper rims of the PTC cavity; or when key constituents of the upper rims, responsible for the remote interactions (e.g., the tip of H69) are disordered, as is the case of crystal structure of H50S [5,6]. It seems, therefore, that the global localization of ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.