Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... • By definition, this is the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
... • By definition, this is the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
- Malaysian Society of Plant Physiology
... Purification of MCS In the previous study, gel filtration was used as a main chromatographic method for the purification of MCS (Shioi et al. 1996; Suzuki & Shioi 2001, 2002; Suzuki et al., 2005). In the present study, we used cation-exchange chromatography (Macro-Prep High S support) and found it w ...
... Purification of MCS In the previous study, gel filtration was used as a main chromatographic method for the purification of MCS (Shioi et al. 1996; Suzuki & Shioi 2001, 2002; Suzuki et al., 2005). In the present study, we used cation-exchange chromatography (Macro-Prep High S support) and found it w ...
Topic 6 Section C
... A drying agent may not necessarily be a dehydrating agent. For example, anhydrous calcium chloride can dry gases. However, it cannot remove chemically combined water from other compounds. ...
... A drying agent may not necessarily be a dehydrating agent. For example, anhydrous calcium chloride can dry gases. However, it cannot remove chemically combined water from other compounds. ...
Regulation of Phenylalanine and Tyrosine
... Extraction and purification procedures. All procedures were carried out at 4 "C using buffer A and buffer B (0.15 M-Tris/HC], pH 7.7, containing 0.1 mM-dithioerythritoland 0.1 m-EDTA). When necessary, buffers and protein solutions were supplemented with the protease inhibitor phenylmethanesulphonyl ...
... Extraction and purification procedures. All procedures were carried out at 4 "C using buffer A and buffer B (0.15 M-Tris/HC], pH 7.7, containing 0.1 mM-dithioerythritoland 0.1 m-EDTA). When necessary, buffers and protein solutions were supplemented with the protease inhibitor phenylmethanesulphonyl ...
CHAPTER 5 GASES
... Recall that Avogadro’s Law states that the volume of a gas is directly proportional to the number of moles of gas at constant temperature and pressure. The ammonia and nitric oxide coefficients in the balanced equation are the same, so one volume of nitric oxide must be obtained from one volume of a ...
... Recall that Avogadro’s Law states that the volume of a gas is directly proportional to the number of moles of gas at constant temperature and pressure. The ammonia and nitric oxide coefficients in the balanced equation are the same, so one volume of nitric oxide must be obtained from one volume of a ...
Part 2-ICHO-26-30
... more atoms of the elements F, Cl, Br, and I, was examined. A small amount of the substance reacted with water. It was completely hydrolyzed without any oxidation or reduction, and all reaction products dissolved. 0.1 M solutions of a series of test reagents were added to separate small portions of a ...
... more atoms of the elements F, Cl, Br, and I, was examined. A small amount of the substance reacted with water. It was completely hydrolyzed without any oxidation or reduction, and all reaction products dissolved. 0.1 M solutions of a series of test reagents were added to separate small portions of a ...
Chapter 1 Introduction: Matter and Measurement
... Uncertainty in Measurements Different measuring devices have different uses and different degrees of accuracy. ...
... Uncertainty in Measurements Different measuring devices have different uses and different degrees of accuracy. ...
Chapter 03 - KFUPM Faculty List
... So when 10 g SO3 are produced, then all O2 is used up, the reaction stops and some S is left over: O2 is the limiting reactant, S the excess reactant. The other possible method (to calculate how much oxygen is needed to react with all the sulfur) gives the same result: ...
... So when 10 g SO3 are produced, then all O2 is used up, the reaction stops and some S is left over: O2 is the limiting reactant, S the excess reactant. The other possible method (to calculate how much oxygen is needed to react with all the sulfur) gives the same result: ...
preview as pdf
... Types of Electrolytes Electrolytes can be further classified as strong electrolytes or weak electrolytes. For a strong electrolyte, such as sodium chloride 1NaCl2, there is 100% dissociation of the solute into ions. When the electrodes from the light bulb apparatus are placed in the NaCl solution, t ...
... Types of Electrolytes Electrolytes can be further classified as strong electrolytes or weak electrolytes. For a strong electrolyte, such as sodium chloride 1NaCl2, there is 100% dissociation of the solute into ions. When the electrodes from the light bulb apparatus are placed in the NaCl solution, t ...
ARIUS MACULATUS EAST COAST OF INDIA
... Stargazers and some species of Shark, Ratfish, Catfish, Surgeonfish and Blenny are known or suspected to be venomous [1]. The production of toxins by aquatic animals is an important strategy that guarantees its survival in a highly competitive ecosystem. In this way, these animals defend themselves ...
... Stargazers and some species of Shark, Ratfish, Catfish, Surgeonfish and Blenny are known or suspected to be venomous [1]. The production of toxins by aquatic animals is an important strategy that guarantees its survival in a highly competitive ecosystem. In this way, these animals defend themselves ...
Part 3-ICHO-31-35
... bomb calorimeter, which initially contains 710.0 g of water at 25.000 °C. After the reaction is completed, the temperature is observed to be 27.250 °C, and 1.5144 g of CO 2 (g) and 0.2656 g of H2O(l) are produced. 1.1 Determine the molecular formula and write a balanced equation with correct state o ...
... bomb calorimeter, which initially contains 710.0 g of water at 25.000 °C. After the reaction is completed, the temperature is observed to be 27.250 °C, and 1.5144 g of CO 2 (g) and 0.2656 g of H2O(l) are produced. 1.1 Determine the molecular formula and write a balanced equation with correct state o ...
Changing Matter
... • Mass is conserved amount of atoms cannot change – Nothing is created or destroyed ...
... • Mass is conserved amount of atoms cannot change – Nothing is created or destroyed ...
Under Choice Based Credit System Proposed syllabus and Scheme of Examination
... Kc and Kx for reactions involving ideal gases. (8 Lectures) Ionic Equilibria: Strong, moderate and weak electrolytes, degree of ionization, factors affecting degree of ionization, ionization constant and ionic product of water. Ionization of weak acids and bases, pH scale, common ion effect. Salt hy ...
... Kc and Kx for reactions involving ideal gases. (8 Lectures) Ionic Equilibria: Strong, moderate and weak electrolytes, degree of ionization, factors affecting degree of ionization, ionization constant and ionic product of water. Ionization of weak acids and bases, pH scale, common ion effect. Salt hy ...
Quantitative Chemical Analysis
... One of our most pressing problems is the need for sources of energy to replace oil. The chart at the right shows that world production of oil per capita has probably already peaked. Oil will play a decreasing role as an energy source and should be more valuable as a raw material than as a fuel. Ther ...
... One of our most pressing problems is the need for sources of energy to replace oil. The chart at the right shows that world production of oil per capita has probably already peaked. Oil will play a decreasing role as an energy source and should be more valuable as a raw material than as a fuel. Ther ...
Complete Set
... • The ocean contains a variety of forms of CO32– and CO2 with a variety of acid-base and solubility equilibria determining their concentrations. There is concern that increasing levels of CO2 will lead to increased dissolution of CaCO3 and critically affect the survival of life forms that rely on a ...
... • The ocean contains a variety of forms of CO32– and CO2 with a variety of acid-base and solubility equilibria determining their concentrations. There is concern that increasing levels of CO2 will lead to increased dissolution of CaCO3 and critically affect the survival of life forms that rely on a ...
(1) Giycophorin was incorporated into large
... was found that in glycophorin/DOPE vesicles much larger particles (about 100 A) were present at the fracture face than in the case of glycop h o r i n / D O P C and glycophorin/DOPS vesicles. Earlier studies [1] showing differences in barrier function upon incorporation of glycophorin into a mixture ...
... was found that in glycophorin/DOPE vesicles much larger particles (about 100 A) were present at the fracture face than in the case of glycop h o r i n / D O P C and glycophorin/DOPS vesicles. Earlier studies [1] showing differences in barrier function upon incorporation of glycophorin into a mixture ...
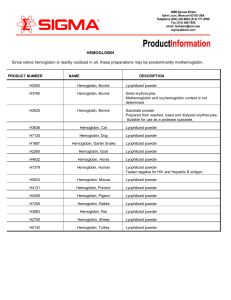
HEMOGLOBIN Since native hemoglobin is readily oxidized in air
... Hemoglobin is usually prepared by separating red blood corpuscles from the lighter plasma components by centrifugation. The plasma is siphoned off and ether is added to the corpuscle paste, causing the cells to burst. Another centrifugation removes the ruptured cell envelopes, and leaves a clear red ...
... Hemoglobin is usually prepared by separating red blood corpuscles from the lighter plasma components by centrifugation. The plasma is siphoned off and ether is added to the corpuscle paste, causing the cells to burst. Another centrifugation removes the ruptured cell envelopes, and leaves a clear red ...
PDF Links - Journal of the Korean Ceramic Society
... The properties of biological bone tissue16) are essentially based on the organic-inorganic interaction between calcium phosphate [CaP] and the protein matrix, such as each type of COL, glucosamine or other proteins. In this report the GEL matrix was modified with Aspartic acid [Asp], Asparagine [Asn ...
... The properties of biological bone tissue16) are essentially based on the organic-inorganic interaction between calcium phosphate [CaP] and the protein matrix, such as each type of COL, glucosamine or other proteins. In this report the GEL matrix was modified with Aspartic acid [Asp], Asparagine [Asn ...
- Academy Test Bank
... -(CH2CHCN)n-, where n is typically greater than 10,000. Given that a sample of monodisperse polyacrilonitrile weighs 197.4 g and contains 1.046 10 20 molecules of -(CH2CHCN)n-, calculate n. A) 2.141 10 4 B) 6.026 10 7 C) 6.018 10 39 D) 6.022 10 23 E) 1.136 10 6 ANS: A PTS: 1 DIF: difficu ...
... -(CH2CHCN)n-, where n is typically greater than 10,000. Given that a sample of monodisperse polyacrilonitrile weighs 197.4 g and contains 1.046 10 20 molecules of -(CH2CHCN)n-, calculate n. A) 2.141 10 4 B) 6.026 10 7 C) 6.018 10 39 D) 6.022 10 23 E) 1.136 10 6 ANS: A PTS: 1 DIF: difficu ...
General and Inorganic Chemistry
... 3. III Basic laboratory procedures and methods ............................................................................... 20 1. III.1 Basic guidelines for working with hazardous materials .............................................. 20 1.1. III.1.1 Laboratory safety .......................... ...
... 3. III Basic laboratory procedures and methods ............................................................................... 20 1. III.1 Basic guidelines for working with hazardous materials .............................................. 20 1.1. III.1.1 Laboratory safety .......................... ...
PART 3-ICHO 11-15
... substances: barium chloride, sodium sulphate, potassium chloride, magnesium nitrate, sodium orthophosphate, barium hydroxide, lead nitrate, potassium hydroxide, aluminium sulphate, sodium carbonate. Using only these solutions as reagents, determine in which of the numbered test tubes each of the abo ...
... substances: barium chloride, sodium sulphate, potassium chloride, magnesium nitrate, sodium orthophosphate, barium hydroxide, lead nitrate, potassium hydroxide, aluminium sulphate, sodium carbonate. Using only these solutions as reagents, determine in which of the numbered test tubes each of the abo ...
Ch. 12 Stoichiometry
... Molecules: Indicates amt of molecules reacting/ being produced Moles: Coefficients of balanced equation indicate relative # of moles of reactant and products / Most important info from equation Mass: Obeys law of conservation of mass ...
... Molecules: Indicates amt of molecules reacting/ being produced Moles: Coefficients of balanced equation indicate relative # of moles of reactant and products / Most important info from equation Mass: Obeys law of conservation of mass ...
Chemical polymerization of aniline in phenylphosphinic
... step. The inherent viscosities of 0.01 % solutions of the PANI samples doped with APP in sulfuric acid were determined using an Ubbelohde viscosimeter. The densities of the PANI samples were determined by the picnometer method, in decaline. The electrical conductivities were measured on press pellet ...
... step. The inherent viscosities of 0.01 % solutions of the PANI samples doped with APP in sulfuric acid were determined using an Ubbelohde viscosimeter. The densities of the PANI samples were determined by the picnometer method, in decaline. The electrical conductivities were measured on press pellet ...
Solution Structure of the Tandem Acyl Carrier Protein Domains from
... The polyunsaturated fatty acid (PUFA) synthases from deep-sea bacteria invariably contain multiple acyl carrier protein (ACP) domains in tandem. This conserved tandem arrangement has been implicated in both amplification of fatty acid production (additive effect) and in structural stabilization of t ...
... The polyunsaturated fatty acid (PUFA) synthases from deep-sea bacteria invariably contain multiple acyl carrier protein (ACP) domains in tandem. This conserved tandem arrangement has been implicated in both amplification of fatty acid production (additive effect) and in structural stabilization of t ...
IIT-JEE - Brilliant Public School Sitamarhi
... Find the Schottky defect in g cm–3. Q.30 A strong current of trivalent gaseous boron passed through a germanium crystal decreases the density of the crystal due to part replacement of germanium by boron and due to interstitial vacancies created by missing Ge atoms. In one such experiment, one gram o ...
... Find the Schottky defect in g cm–3. Q.30 A strong current of trivalent gaseous boron passed through a germanium crystal decreases the density of the crystal due to part replacement of germanium by boron and due to interstitial vacancies created by missing Ge atoms. In one such experiment, one gram o ...
Size-exclusion chromatography

Size-exclusion chromatography (SEC) is a chromatographic method in which molecules in solution are separated by their size, and in some cases molecular weight. It is usually applied to large molecules or macromolecular complexes such as proteins and industrial polymers. Typically, when an aqueous solution is used to transport the sample through the column, the technique is known as gel-filtration chromatography, versus the name gel permeation chromatography, which is used when an organic solvent is used as a mobile phase. SEC is a widely used polymer characterization method because of its ability to provide good molar mass distribution (Mw) results for polymers.