Selected Solutions to End of Chapter 13 Problems
... 24. The mitochondrial electron transport pathway from NADH to oxygen, the overall equation is simple: NADH + H+ + ½ O2 NAD+ + H2O Here is Table 13-7 standard reduction potential of some half cells (half reactions) ...
... 24. The mitochondrial electron transport pathway from NADH to oxygen, the overall equation is simple: NADH + H+ + ½ O2 NAD+ + H2O Here is Table 13-7 standard reduction potential of some half cells (half reactions) ...
Chapter 8 "Ionic versus Covalent Bonding"
... attractive energy as 0.5 mol of gaseous LiCl? The ionic radii are Li + = 76 pm, Mg+2 = 72 pm, and Cl− = 181 pm. 3. Sketch a diagram showing the relationship between potential energy and internuclear distance (from r = ∞ to r = 0) for the interaction of a bromide ion and a potassium ion to form gaseo ...
... attractive energy as 0.5 mol of gaseous LiCl? The ionic radii are Li + = 76 pm, Mg+2 = 72 pm, and Cl− = 181 pm. 3. Sketch a diagram showing the relationship between potential energy and internuclear distance (from r = ∞ to r = 0) for the interaction of a bromide ion and a potassium ion to form gaseo ...
Thermodynamics: Entropy and Free Energy
... temperature of a substance by one degree Celsius or one kelvin. It has the same units as standard molar entropy, J/mol K. Table 19.1.1 shows standard molar entropy values for water at various temperatures. Interactive Figure 19.1.11 shows a generalized plot of the entropy of a substance at each te ...
... temperature of a substance by one degree Celsius or one kelvin. It has the same units as standard molar entropy, J/mol K. Table 19.1.1 shows standard molar entropy values for water at various temperatures. Interactive Figure 19.1.11 shows a generalized plot of the entropy of a substance at each te ...
Understanding a Millennium of Hemoglobin Evolution: Correlating
... Department of Chemistry, University of Massachusetts, Amherst, MA 01003 Hemoglobins are dioxygen transport proteins. They are universally present in higher vertebrates as a tetrameric protein, and are found in some bacteria and plants as monomers. Remarkable diversity in quaternary (4°) assemblage i ...
... Department of Chemistry, University of Massachusetts, Amherst, MA 01003 Hemoglobins are dioxygen transport proteins. They are universally present in higher vertebrates as a tetrameric protein, and are found in some bacteria and plants as monomers. Remarkable diversity in quaternary (4°) assemblage i ...
Additions to ketones and aldehydes
... ← imine (a N analog of a carbonyl compound) + H2O. i) Nucleophilic addition to give a hemiaminal (carbinolamine) is followed by E1 elimination of H2O (H+ comes from N; O is protonated before it leaves). ii) Equilibrium favors imine for R = hydroxy, alkoxy, or amino groups. (Products called oximes, o ...
... ← imine (a N analog of a carbonyl compound) + H2O. i) Nucleophilic addition to give a hemiaminal (carbinolamine) is followed by E1 elimination of H2O (H+ comes from N; O is protonated before it leaves). ii) Equilibrium favors imine for R = hydroxy, alkoxy, or amino groups. (Products called oximes, o ...
Authors Title Year Keywords Journal/Proceedings Emile Bol
... para-substituted benzylamines and their dideuterated counterparts reveal KIEs (>6) larger than those observed with native AADH (KIEs ~ unity). This is attributed to formation of the benzylamine-derived iminoquinone requiring structural rearrangement of the benzyl side chain in the active site of the ...
... para-substituted benzylamines and their dideuterated counterparts reveal KIEs (>6) larger than those observed with native AADH (KIEs ~ unity). This is attributed to formation of the benzylamine-derived iminoquinone requiring structural rearrangement of the benzyl side chain in the active site of the ...
video slide - Green River Community College
... 5. From what kind of molecules can this energy be used directly? Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings ...
... 5. From what kind of molecules can this energy be used directly? Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings ...
Oxidative ortho-C-N Fusion of Aniline by OsO4. Isolation
... N-aryl-1,2-phenylenediamides. Our interest in these reactions originated from our previous results13 on transition metal mediated oxidative ortho-C-N fusion of aromatic amines. The reactions occur because of ortho-C-H activation as a consequence of metal coordination. Furthermore, we have shown befo ...
... N-aryl-1,2-phenylenediamides. Our interest in these reactions originated from our previous results13 on transition metal mediated oxidative ortho-C-N fusion of aromatic amines. The reactions occur because of ortho-C-H activation as a consequence of metal coordination. Furthermore, we have shown befo ...
Biochemical Thermodynamics - Research Centers | Center for
... carbon but containing one or more atoms and at least one net charge. – Ions of biological significance include Cl-, Na+, K+, Mg+2, Mn+2, I-, Ca+2, PO4-3, SO4-2, NO3-, NO2-, and NH4+. – Phosphate (PO4-3) is often found in partially protonated forms HPO4-2 and H2PO4– Ammonium ions occasionally appear ...
... carbon but containing one or more atoms and at least one net charge. – Ions of biological significance include Cl-, Na+, K+, Mg+2, Mn+2, I-, Ca+2, PO4-3, SO4-2, NO3-, NO2-, and NH4+. – Phosphate (PO4-3) is often found in partially protonated forms HPO4-2 and H2PO4– Ammonium ions occasionally appear ...
AVOGADRO EXAMS 1991 - 2002 PRACTICE BOOKLET
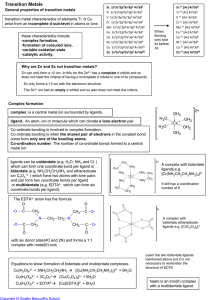
... 14. ”A valence electron in a pure metal is not held by any specific atom, but all valence electrons are used to hold together the atoms of the metal.” This statement is best classified as (a) a specific experimental fact (b) an opinion not based upon evidence (c) a correct definition of a chemical t ...
... 14. ”A valence electron in a pure metal is not held by any specific atom, but all valence electrons are used to hold together the atoms of the metal.” This statement is best classified as (a) a specific experimental fact (b) an opinion not based upon evidence (c) a correct definition of a chemical t ...
Glucose
... 2. OH group at position 2 at the right side. Both of which are present in glucose and galactose. Fructose, which does not contain -OH group to the right at position 2 is absorbed more slowly than glucose and galactose by passive diffusion (slow process). ...
... 2. OH group at position 2 at the right side. Both of which are present in glucose and galactose. Fructose, which does not contain -OH group to the right at position 2 is absorbed more slowly than glucose and galactose by passive diffusion (slow process). ...
Adenosine triphosphate Adenosine triphosphate Adenosine
... acid cycle produces two molecules of carbon dioxide, one molecule of the ATP equivalent guanosine triphosphate (GTP) through substrate-level phosphorylation catalyzed bysuccinyl-CoA synthetase, three molecules of the reduced coenzyme NADH, and one molecule of the reduced coenzyme FADH2. Both of thes ...
... acid cycle produces two molecules of carbon dioxide, one molecule of the ATP equivalent guanosine triphosphate (GTP) through substrate-level phosphorylation catalyzed bysuccinyl-CoA synthetase, three molecules of the reduced coenzyme NADH, and one molecule of the reduced coenzyme FADH2. Both of thes ...
Enzyme Power Point
... called the active site • Only certain substrates can fit the active site • Amino acid R groups in the active site help substrate bind • Enzyme-substrate complex forms • Substrate reacts to form product • Product is released ...
... called the active site • Only certain substrates can fit the active site • Amino acid R groups in the active site help substrate bind • Enzyme-substrate complex forms • Substrate reacts to form product • Product is released ...
Energy Metabolism - Georgia Institute of Technology
... – H+ actively transported out of matrix – H+ leak back as H+PO4 2- ...
... – H+ actively transported out of matrix – H+ leak back as H+PO4 2- ...
CHEM 1411 – STUDY-GUIDE-for-TEST-2
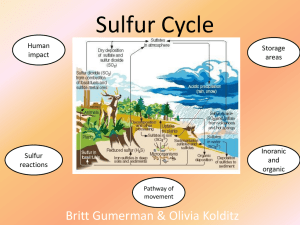
... 56. During volcanic eruptions, hydrogen sulfide gas is given off and oxidized by air according to the following chemical equation: 2H2S(g) + 3O2(g) 2SO2(g) + 2H2O(g) Calculate the standard enthalpy change for the above reaction given: 3S(s) + 2H2O(g) 2H2S(g) + SO2(g) H° = 146.9 kJ/mol S(s) + O2 ...
... 56. During volcanic eruptions, hydrogen sulfide gas is given off and oxidized by air according to the following chemical equation: 2H2S(g) + 3O2(g) 2SO2(g) + 2H2O(g) Calculate the standard enthalpy change for the above reaction given: 3S(s) + 2H2O(g) 2H2S(g) + SO2(g) H° = 146.9 kJ/mol S(s) + O2 ...
PPT - Gmu - George Mason University
... The Third Law is a statement about reaching Absolute Zero (0° K) ...
... The Third Law is a statement about reaching Absolute Zero (0° K) ...
Chapter Eleven - Wright State University
... Overview of the Citric Acid Cycle In Stage 1 of digestion the ingested foods (carbohydrates, fats, and proteins) were broken down into their smaller parts (sugars, fatty acids, etc.). In Stage 2 these compounds were further broken down to 2-carbons acetyl units bonded to CoA. These units now enter ...
... Overview of the Citric Acid Cycle In Stage 1 of digestion the ingested foods (carbohydrates, fats, and proteins) were broken down into their smaller parts (sugars, fatty acids, etc.). In Stage 2 these compounds were further broken down to 2-carbons acetyl units bonded to CoA. These units now enter ...
Quantum Tunnelling to the Origin and Evolution of Life
... photosynthesis. This type of photosynthesis fuels most ecosystems directly (phototrophy) as well as indirectly (chemotrophy based on energy from organic matter produced by oxygenic photosynthesis). It also enriches the atmosphere and hydrosphere with its byproduct O2. The enrichment with O2 forced t ...
... photosynthesis. This type of photosynthesis fuels most ecosystems directly (phototrophy) as well as indirectly (chemotrophy based on energy from organic matter produced by oxygenic photosynthesis). It also enriches the atmosphere and hydrosphere with its byproduct O2. The enrichment with O2 forced t ...
Module 1 Predictor Questions
... 15. What is the mass, in g, of a 68.2 cm3 sample of ethyl alcohol? The density of ethyl alcohol is 0.789 g/cm3. The correct answer is: 53.8 g Density converts the volume of a substance into the mass. ...
... 15. What is the mass, in g, of a 68.2 cm3 sample of ethyl alcohol? The density of ethyl alcohol is 0.789 g/cm3. The correct answer is: 53.8 g Density converts the volume of a substance into the mass. ...
Infrared spectroscopic studies: from small molecules to large.
... reactions in the cell, antibodies that bind antigens or foreign substances and target them for destruction, transporters that binds small molecules and move them from one cell location to the next, etc.5 An important group of enzymes that are central to many biological processes are the kinases. The ...
... reactions in the cell, antibodies that bind antigens or foreign substances and target them for destruction, transporters that binds small molecules and move them from one cell location to the next, etc.5 An important group of enzymes that are central to many biological processes are the kinases. The ...
11-Electrophoretic method for the separation of LDH
... pyruvate to lactic acid and this reaction is catalyzed by the enzyme lactate dehydrogenase (LDH). In skeletal muscle, where oxygen deprivation is common during exercise, the reaction is efficient and large amounts of lactate can be formed. In tissues that preferentially oxidize glucose aerobically t ...
... pyruvate to lactic acid and this reaction is catalyzed by the enzyme lactate dehydrogenase (LDH). In skeletal muscle, where oxygen deprivation is common during exercise, the reaction is efficient and large amounts of lactate can be formed. In tissues that preferentially oxidize glucose aerobically t ...
A Model For the Calculation of Solvent ... Reaction Rates for Process Design Purposes
... solvents be chosen with respect not only to their effectiveness in their respective process tasks but also for process-wide requirements such as their ease of recovery, low toxicity and environmental impact and possible applicability to other process tasks. Although there are models for the evaluati ...
... solvents be chosen with respect not only to their effectiveness in their respective process tasks but also for process-wide requirements such as their ease of recovery, low toxicity and environmental impact and possible applicability to other process tasks. Although there are models for the evaluati ...
Enzyme Specificity and Selectivity
... from the three-dimensional structure of their respective active sites. Although the overall structures of these proteases are very similar, each enzyme has an active site that is sterically and electrostatically complementary to its substrate. Both trypsin and chymotrypsin have a deep pocket at thei ...
... from the three-dimensional structure of their respective active sites. Although the overall structures of these proteases are very similar, each enzyme has an active site that is sterically and electrostatically complementary to its substrate. Both trypsin and chymotrypsin have a deep pocket at thei ...