fahad h. ahmad - Fahad`s Academy
... Where x = distance moved by the substance and; y = distance moved by the solvent Checking the Purity of Substances - Pure substances have FIXED MELTING AND BOILING POINTS. Pure water boils at 100oC and melts at 0oC. - Impure substances have NO FIXED MELTING AND BOILING POINTS. They melt and boil a ...
... Where x = distance moved by the substance and; y = distance moved by the solvent Checking the Purity of Substances - Pure substances have FIXED MELTING AND BOILING POINTS. Pure water boils at 100oC and melts at 0oC. - Impure substances have NO FIXED MELTING AND BOILING POINTS. They melt and boil a ...
Nuclear Spins in Quantum Dots
... in the theoretical physics group in Delft. In the beginning of my PhD I knew that I wanted to work on electron spins and quantum dots but I only had vague ideas about what that entailed. With time these ideas became more clear, largely with the help of my supervisors, and I found myself working on t ...
... in the theoretical physics group in Delft. In the beginning of my PhD I knew that I wanted to work on electron spins and quantum dots but I only had vague ideas about what that entailed. With time these ideas became more clear, largely with the help of my supervisors, and I found myself working on t ...
Structure and transport properties of atomic chains and molecules
... geometries, electron-phonon coupling, and polarization effects of the surrounding medium. The development of such models is a great challenge, as not only the quantum nature of the electrons, but also the atomistic details of the junction must be accounted for. This calls for combining traditional f ...
... geometries, electron-phonon coupling, and polarization effects of the surrounding medium. The development of such models is a great challenge, as not only the quantum nature of the electrons, but also the atomistic details of the junction must be accounted for. This calls for combining traditional f ...
Spin-Valley Kondo Effect in Multi
... For Si QDs, experiments on the Kondo effect have so far been able to reach the stage of few-electron dots, but not that of single-electron ones. In a few-electron Si dot6 , a Kondo resonance at zero bias voltage is split into two peaks by an applied magnetic field. The value of the extracted g facto ...
... For Si QDs, experiments on the Kondo effect have so far been able to reach the stage of few-electron dots, but not that of single-electron ones. In a few-electron Si dot6 , a Kondo resonance at zero bias voltage is split into two peaks by an applied magnetic field. The value of the extracted g facto ...
Topic 7b Redox notes
... sign followed by a number, or it is zero. Oxidation states of metals are usually written in roman numerals eg Iron (III) chloride. Atoms of elements have no overall charge and are therefore given an oxidation state of zero. When two elements combine, the atoms or ions of the more electropositive ele ...
... sign followed by a number, or it is zero. Oxidation states of metals are usually written in roman numerals eg Iron (III) chloride. Atoms of elements have no overall charge and are therefore given an oxidation state of zero. When two elements combine, the atoms or ions of the more electropositive ele ...
Print this article - Bangladesh Journals Online
... assignable for protons Hd and Ha respectively. The two doublets of doublet at δ 6.5 (JHa-Hb = JHb-Hc = J = 8.0 Hz) and 6.9 (JHb-Hc= JHc-Hd = J = 8.0 Hz) accounts for the Ha and Hd respectively, while the relatively downfield signal at δ 8.5 has been assigned for the imine (=N-H) proton of 2-mercapto ...
... assignable for protons Hd and Ha respectively. The two doublets of doublet at δ 6.5 (JHa-Hb = JHb-Hc = J = 8.0 Hz) and 6.9 (JHb-Hc= JHc-Hd = J = 8.0 Hz) accounts for the Ha and Hd respectively, while the relatively downfield signal at δ 8.5 has been assigned for the imine (=N-H) proton of 2-mercapto ...
Size-limited characteristics of semiconductor
... involve highly excited levels in the ion far away from the ground state. They are expected to set in at large distances from the surface, R, when the atomic wavefunction begins to touch the surface. This simple picture suggests the probing ion-surface interactions at large distances involving Rydber ...
... involve highly excited levels in the ion far away from the ground state. They are expected to set in at large distances from the surface, R, when the atomic wavefunction begins to touch the surface. This simple picture suggests the probing ion-surface interactions at large distances involving Rydber ...
orange review book_2014_key
... 19. One similarity between all mixtures and compounds is that both (1) are heterogeneous (2) consist of two or more substances (3) are homogeneous (4) are heterogeneous 20. A dilute, aqueous potassium nitrate solution is best classified as a (1) homogeneous compound (2) homogeneous mixture ...
... 19. One similarity between all mixtures and compounds is that both (1) are heterogeneous (2) consist of two or more substances (3) are homogeneous (4) are heterogeneous 20. A dilute, aqueous potassium nitrate solution is best classified as a (1) homogeneous compound (2) homogeneous mixture ...
Document
... A. The transmission coefficient through the barrier depends on E, V and a B. The transmission coefficient increases when a decreases for a given E and V C. The transmission coefficient increases when V decreases for a given E and a D. The transmission coefficient increases when E decreases for a giv ...
... A. The transmission coefficient through the barrier depends on E, V and a B. The transmission coefficient increases when a decreases for a given E and V C. The transmission coefficient increases when V decreases for a given E and a D. The transmission coefficient increases when E decreases for a giv ...
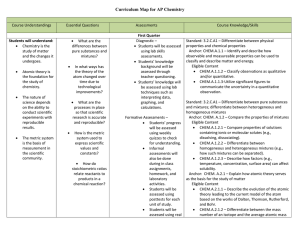
AP Chemistry Curriculum Map - Belle Vernon Area School District
... Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content CHEM.A.2.2.4 – Relate the existence of quantized energy levels to atomic emission spectra. Standard: 3.2.C.A2 – Explain how atoms combine to form compounds through both ionic and covalent bonding. Anchor: CHEM.A.1.1 ...
... Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content CHEM.A.2.2.4 – Relate the existence of quantized energy levels to atomic emission spectra. Standard: 3.2.C.A2 – Explain how atoms combine to form compounds through both ionic and covalent bonding. Anchor: CHEM.A.1.1 ...
Electronic Structure of Strained GaSb/GaAs Quantum Dot
... one without it. PL technique is employed to probe the ground-state transition energies. Using the experimental data, they calculated a band structure of these dots by using the 8-band k.p approach. The theoretical analysis accounts well in both GaAsSb/GaAs quantum dot with a capping quantum well and ...
... one without it. PL technique is employed to probe the ground-state transition energies. Using the experimental data, they calculated a band structure of these dots by using the 8-band k.p approach. The theoretical analysis accounts well in both GaAsSb/GaAs quantum dot with a capping quantum well and ...
www.fahadsacademy.com
... NUCLEON NUMBER is the number of protons and neutrons in nucleus of an atom. Therefore, to find the number of neutrons, we subtract proton number from nucleon number, i.e.: Nucleon number – Proton number = Neutrons ELECTRONS have the same number as protons to balance the charges. ...
... NUCLEON NUMBER is the number of protons and neutrons in nucleus of an atom. Therefore, to find the number of neutrons, we subtract proton number from nucleon number, i.e.: Nucleon number – Proton number = Neutrons ELECTRONS have the same number as protons to balance the charges. ...
Chapter 12
... It is important to understand that when we say that the atomic mass of carbon is 12.01 amu, we are referring to the average value. If carbon atoms could be examined individually, we would find either an atom of atomic mass 12.00000 amu or one of 13.00335 amu, but never one of 12.01 amu. Example 3.1 ...
... It is important to understand that when we say that the atomic mass of carbon is 12.01 amu, we are referring to the average value. If carbon atoms could be examined individually, we would find either an atom of atomic mass 12.00000 amu or one of 13.00335 amu, but never one of 12.01 amu. Example 3.1 ...
NOTE Mixed-Ligand Complexes of Cu2+, Ni2+, Co2+, Zn2+ with 2,2
... complexation takes place. However, curve E diverges from curve C after pH 3-4 suggesting the coordination of ABDA with primary complex, since the dissociation of primary complex does not take place in the pH range 4-8. It can be considered that ABDA combines with species [M(Bipy)]2+ just as it does ...
... complexation takes place. However, curve E diverges from curve C after pH 3-4 suggesting the coordination of ABDA with primary complex, since the dissociation of primary complex does not take place in the pH range 4-8. It can be considered that ABDA combines with species [M(Bipy)]2+ just as it does ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.