Lecture 13: Heisenberg and Uncertainty
... For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. ...
... For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. ...
Teacher text
... work- is one of the most powerful tools in teaching and learning, particularly if it is not graded (no marks given). Indeed, interaction and feedback are the consistent ingredients of the interactive-engagement methods promoted by PER researchers (Hake, 1998; Mazur, 1997; Meltzer & Manivannan, 2002) ...
... work- is one of the most powerful tools in teaching and learning, particularly if it is not graded (no marks given). Indeed, interaction and feedback are the consistent ingredients of the interactive-engagement methods promoted by PER researchers (Hake, 1998; Mazur, 1997; Meltzer & Manivannan, 2002) ...
Coulomb blockade in the fractional quantum Hall effect regime *
... Despite enormous theoretical and experimental effort during the past decade, the nature of transport in the fractional quantum Hall effect 共FQHE兲 regime of the two-dimensional electron gas1 remains uncertain. Although chiral Luttinger liquid 共CLL兲 theory2,3 has successfully predicted transport and s ...
... Despite enormous theoretical and experimental effort during the past decade, the nature of transport in the fractional quantum Hall effect 共FQHE兲 regime of the two-dimensional electron gas1 remains uncertain. Although chiral Luttinger liquid 共CLL兲 theory2,3 has successfully predicted transport and s ...
Microwave background radiation of hydrogen atoms 1 Introduction
... We will show below, as simple and clear as possible, the derivation of the both aforementioned spectra. For this aim, we will lay stress mainly on the wave motion of the electron along the orbit taking into account that one half-wave of the fundamental tone of the electron is placed on the Bohr firs ...
... We will show below, as simple and clear as possible, the derivation of the both aforementioned spectra. For this aim, we will lay stress mainly on the wave motion of the electron along the orbit taking into account that one half-wave of the fundamental tone of the electron is placed on the Bohr firs ...
Periodicity - Teach-n-Learn-Chem
... The Modern periodic table is based on the periodic law. Periodic law - properties of elements tend to change with increasing atomic number in a periodic way ...
... The Modern periodic table is based on the periodic law. Periodic law - properties of elements tend to change with increasing atomic number in a periodic way ...
... The Royal Swedish Academy of Sciences has decided to award the Nobel Prize in Chemistry for 2000 to three scientists who have revolutionised the development of electrically conductive polymers. Professor Alan J. Heeger at the University of California at Santa Barbara, USA Professor Alan G. MacDiarmi ...
Topic 2
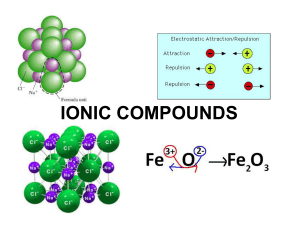
... negatively charged ion, called an anion (more electrons than protons). i.e, Cl– An atom that loses electrons becomes a positively charged ion, called a cation (more protons than electrons). i.e., Na+ – An ionic compound is a compound composed of cations and anions. Answer the following questions for ...
... negatively charged ion, called an anion (more electrons than protons). i.e, Cl– An atom that loses electrons becomes a positively charged ion, called a cation (more protons than electrons). i.e., Na+ – An ionic compound is a compound composed of cations and anions. Answer the following questions for ...
Balancing Chemical Equations – A Primer
... need a process or methodology to help us do it correctly. Let’s start with a reaction that bonds two chemicals together... Sodium and Fluorine Na + F ...
... need a process or methodology to help us do it correctly. Let’s start with a reaction that bonds two chemicals together... Sodium and Fluorine Na + F ...
Production of Materials by Jimmy Huang
... The oxidation reaction at the anode is: Zn(s) + 2OH–(aq) → Zn(OH)2(s) + 2e– The reduction reaction that occurs at the cathode is: Ag2O(s) + H2O(l) + 2e- 2Ag(s) + 2OH-(aq) The button cell is very practical due to its small size and tendency to be light weighted. However, it may be relatively expens ...
... The oxidation reaction at the anode is: Zn(s) + 2OH–(aq) → Zn(OH)2(s) + 2e– The reduction reaction that occurs at the cathode is: Ag2O(s) + H2O(l) + 2e- 2Ag(s) + 2OH-(aq) The button cell is very practical due to its small size and tendency to be light weighted. However, it may be relatively expens ...
Chapter 8 - Power Point Presentation
... • Explains how molecules obtain their shapes. • Coulomb’s Law allows us to predict that regions of High Electron Density (bonds or lone pairs) arrange themselves around a central atom as far away from each other as possible so as to minimize repulsive forces. ...
... • Explains how molecules obtain their shapes. • Coulomb’s Law allows us to predict that regions of High Electron Density (bonds or lone pairs) arrange themselves around a central atom as far away from each other as possible so as to minimize repulsive forces. ...
Topological Quantum Matter
... there is a well-know mapping from classical statistical mechanics in two spatial dimensions to quantum mechanics “(1+1) ...
... there is a well-know mapping from classical statistical mechanics in two spatial dimensions to quantum mechanics “(1+1) ...
Chapter 2
... • Atoms of the various elements differ in number of subatomic particles • An element’s atomic number is the number of protons in its nucleus • An element’s mass number is the sum of protons plus neutrons in the nucleus • Atomic mass, the atom’s total mass, can be approximated by the mass number Copy ...
... • Atoms of the various elements differ in number of subatomic particles • An element’s atomic number is the number of protons in its nucleus • An element’s mass number is the sum of protons plus neutrons in the nucleus • Atomic mass, the atom’s total mass, can be approximated by the mass number Copy ...
The Transactional Interpretation
... • Theory needed to predict behavior of very small particles such as atoms, electrons, photons, and other subatomic particles. • QM works very well but what it actually tells us about reality is very unclear • An interpretation is intended to make clear what the theory tells us about reality ...
... • Theory needed to predict behavior of very small particles such as atoms, electrons, photons, and other subatomic particles. • QM works very well but what it actually tells us about reality is very unclear • An interpretation is intended to make clear what the theory tells us about reality ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.