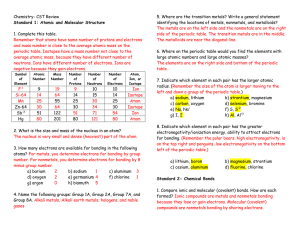
1) Basic familiarity with Atomic Labels. You will need a Periodic
... 6.3) Caffeine, a stimulant in coffee and tea and often found in OTC painkillers, has a molar mass of 194.19g/mol and mass composition 49.48% C, 5.19% H, 28.85% N and 16.48% O. What is the molecular formula of caffeine? 6.4) Ethyne and benzene both have the same empirical formula, CH. Look up (anywhe ...
... 6.3) Caffeine, a stimulant in coffee and tea and often found in OTC painkillers, has a molar mass of 194.19g/mol and mass composition 49.48% C, 5.19% H, 28.85% N and 16.48% O. What is the molecular formula of caffeine? 6.4) Ethyne and benzene both have the same empirical formula, CH. Look up (anywhe ...
First-principles calculations of long-range intermolecular dispersion forces Auayporn Jiemchooroj Link¨
... that there can be an attractive force between two electrically neutral atoms, but it is evident that the presence of long-range interactions account for many phenomena in nature; for example, the condensation of gases to their liquid or solid phases and the attraction of colloidal particles in chemi ...
... that there can be an attractive force between two electrically neutral atoms, but it is evident that the presence of long-range interactions account for many phenomena in nature; for example, the condensation of gases to their liquid or solid phases and the attraction of colloidal particles in chemi ...
Electrochemistry Lecture
... Oxidizing agent; That which is responsible to oxidize another. O2 ; Oxidizing agent; The agent itself undergoes reduction ...
... Oxidizing agent; That which is responsible to oxidize another. O2 ; Oxidizing agent; The agent itself undergoes reduction ...
EP-307 Introduction to Quantum Mechanics
... the order of /2. As we increase the wavelength and it becomes greater than the distance between the two holes. We see one big flash but cannot tell thru which hole the electron went. ...
... the order of /2. As we increase the wavelength and it becomes greater than the distance between the two holes. We see one big flash but cannot tell thru which hole the electron went. ...
chapter 4
... wave. The double slits change the propagation of the electron wave so that it is ‘processed’ to forms diffraction pattern on the screen. Such process would be impossible if electrons are particle (because no one particle can go through both slits at the same time. Such a simultaneous penetration is ...
... wave. The double slits change the propagation of the electron wave so that it is ‘processed’ to forms diffraction pattern on the screen. Such process would be impossible if electrons are particle (because no one particle can go through both slits at the same time. Such a simultaneous penetration is ...
1 CHAPTER 7 ATOMIC SPECTRA 7.1 Introduction Atomic
... century, had an interest in possible connections between the fundamental constants of physics and the natural numbers, and became almost obsessed with the notion that the reciprocal of the fine structure constant should be exactly 137, even insisting on hanging his hat on a conference hall coatpeg n ...
... century, had an interest in possible connections between the fundamental constants of physics and the natural numbers, and became almost obsessed with the notion that the reciprocal of the fine structure constant should be exactly 137, even insisting on hanging his hat on a conference hall coatpeg n ...
Excited Elements - Light Emission Spectroscopy
... corresponds to a particular type of electromagnetic radiation. For example, when electrons fall from a higher energy level down to the 2nd energy level, the wavelength and frequency of the energy produced correspond to that of visible light. Electrons of atoms can be excited in various ways includin ...
... corresponds to a particular type of electromagnetic radiation. For example, when electrons fall from a higher energy level down to the 2nd energy level, the wavelength and frequency of the energy produced correspond to that of visible light. Electrons of atoms can be excited in various ways includin ...
The Concept of Probability in Quantum Mechanics
... From about the beginning of the twentieth century experimental physics amassed an impressive array of strange phenomena which demonstrated the inadequacy of classical physics. The attempts to discover a theoretical structure for the new phenomena led at first to a confusion in which it appeared tha ...
... From about the beginning of the twentieth century experimental physics amassed an impressive array of strange phenomena which demonstrated the inadequacy of classical physics. The attempts to discover a theoretical structure for the new phenomena led at first to a confusion in which it appeared tha ...
practice exercise
... Solution Elements that are in the same group of the periodic table are most likely to exhibit similar chemical and physical properties. We therefore expect that Ca and Mg should be most alike because they are in the same group (2A, the alkaline earth metals). ...
... Solution Elements that are in the same group of the periodic table are most likely to exhibit similar chemical and physical properties. We therefore expect that Ca and Mg should be most alike because they are in the same group (2A, the alkaline earth metals). ...
CHEM 120 WEEK 11 LECTURES (INORGANIC WEEK 2) Dr. MD
... The Periodic table Alkaline earth metals ...
... The Periodic table Alkaline earth metals ...
Matter - tompkinsmath
... The formula unit is Ca(NO3)2(s) Binary ionic compound → an ionic compound composed of two monatomic ions. Two kinds of ...
... The formula unit is Ca(NO3)2(s) Binary ionic compound → an ionic compound composed of two monatomic ions. Two kinds of ...
CHAPTER 2
... __________________ to them in order to have ____________ electrons in their valence shell. (Exceptions: Li, ______, and ______ acquire the configuration of ______________ and thus follow the ___________ rule.) ...
... __________________ to them in order to have ____________ electrons in their valence shell. (Exceptions: Li, ______, and ______ acquire the configuration of ______________ and thus follow the ___________ rule.) ...
CHAPTER TWO ATOMS, MOLECULES, AND IONS
... and neutrons which can be broken down into quarks. For our purpose, electrons, neutrons, and protons are the key smaller parts of an atom. b. All atoms of hydrogen have 1 proton in the nucleus. Different isotopes of hydrogen have 0, 1, or 2 neutrons in the nucleus. Because we are talking about atoms ...
... and neutrons which can be broken down into quarks. For our purpose, electrons, neutrons, and protons are the key smaller parts of an atom. b. All atoms of hydrogen have 1 proton in the nucleus. Different isotopes of hydrogen have 0, 1, or 2 neutrons in the nucleus. Because we are talking about atoms ...
CHAPTER-4 CHEMICAL BONDING AND
... take place is called a coordinate bond. Such a bond is also known as dative bond. It is represented by an arrow (→) pointing towards the acceptor atom. E.g. H3N→BF3 Bond Length: Bond length is defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule Bond Angle: It is ...
... take place is called a coordinate bond. Such a bond is also known as dative bond. It is represented by an arrow (→) pointing towards the acceptor atom. E.g. H3N→BF3 Bond Length: Bond length is defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule Bond Angle: It is ...
chemistry I review pwrpt.
... 2. Create reactants table to compare have to need of each reactant ( units: moles or grams). 3. Identify the limiting and excess reactant. 4. Use limiting reactant to calculate theoretical yield of each product. 5. Calculate percent yield by comparing actual yield (from lab) to theoretical yield (ca ...
... 2. Create reactants table to compare have to need of each reactant ( units: moles or grams). 3. Identify the limiting and excess reactant. 4. Use limiting reactant to calculate theoretical yield of each product. 5. Calculate percent yield by comparing actual yield (from lab) to theoretical yield (ca ...
Quantum Mechanics
... Spectroscopy is an optical technique by which we can IDENTIFY a material based on its emission spectrum. It is heavily used in Astronomy and Remote Sensing. There are too many subcategories to mention here but the one you are probably the most familiar with are flame tests. When an electron gets exc ...
... Spectroscopy is an optical technique by which we can IDENTIFY a material based on its emission spectrum. It is heavily used in Astronomy and Remote Sensing. There are too many subcategories to mention here but the one you are probably the most familiar with are flame tests. When an electron gets exc ...
eBook AQA GCSE Chemistry Unit C2 Part 1
... Ammonia gas exists as molecules. A molecule is a particle made up of two or more atoms chemically bonded together. In ammonia, each molecule consists of one atom of nitrogen joined to three atoms of hydrogen. The atoms are held together by covalent bonds. A covalent bond is a shared pair of electron ...
... Ammonia gas exists as molecules. A molecule is a particle made up of two or more atoms chemically bonded together. In ammonia, each molecule consists of one atom of nitrogen joined to three atoms of hydrogen. The atoms are held together by covalent bonds. A covalent bond is a shared pair of electron ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.