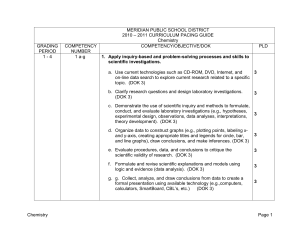
MERIDIAN PUBLIC SCHOOL DISTRICT
... 2. Demonstrate an understanding of the atomic model of matter by explaining atomic structure and chemical bonding. b. Research and explain crucial contributions and critical experiments of Dalton, Thomson, Rutherford, Bohr, de Broglie, and Schrődinger and describe how each discovery contributed to t ...
... 2. Demonstrate an understanding of the atomic model of matter by explaining atomic structure and chemical bonding. b. Research and explain crucial contributions and critical experiments of Dalton, Thomson, Rutherford, Bohr, de Broglie, and Schrődinger and describe how each discovery contributed to t ...
L14alternative - Particle Physics and Particle Astrophysics
... For bound states the wavefunction penetrates the classically forbidden region. Thus, the particle exists in a region where its kinetic energy is negative. To find energies of these states we’ll solve the time independent Schrödinger equation: ...
... For bound states the wavefunction penetrates the classically forbidden region. Thus, the particle exists in a region where its kinetic energy is negative. To find energies of these states we’ll solve the time independent Schrödinger equation: ...
LN_atoms_etc
... Matter consists of individual atoms. All atoms of a given chemical element are identical. Different chemical elements have differing atoms of different mass. Atoms are indestructible – they retain their identity in reactions. Compounds are formed from a combination of elements in small whole number ...
... Matter consists of individual atoms. All atoms of a given chemical element are identical. Different chemical elements have differing atoms of different mass. Atoms are indestructible – they retain their identity in reactions. Compounds are formed from a combination of elements in small whole number ...
PowerPoint - Balancing Equations
... • Molecules may also have brackets to indicate numbers of atoms. E.g. Ca(OH)2 • Notice that the OH is a group O Ca O H • The 2 refers to both H and O H • How many of each atom are in the following? a) NaOH Na = 1, O = 1, H = 1 b) Ca(OH)2 Ca = 1, O = 2, H = 2 c) 3Ca(OH)2 Ca = 3, O = 6, H = 6 ...
... • Molecules may also have brackets to indicate numbers of atoms. E.g. Ca(OH)2 • Notice that the OH is a group O Ca O H • The 2 refers to both H and O H • How many of each atom are in the following? a) NaOH Na = 1, O = 1, H = 1 b) Ca(OH)2 Ca = 1, O = 2, H = 2 c) 3Ca(OH)2 Ca = 3, O = 6, H = 6 ...
shp_05 - Columbia University
... spheres of charge, why should their spins be quantized in magnitude and direction? Classically, there is no way to explain this behavior. In 1925, S. Goudsmidt and G. Uhlenbeck realized that the classical model just cannot apply. Electrons do not spin like tops; their magnetic behavior must be expla ...
... spheres of charge, why should their spins be quantized in magnitude and direction? Classically, there is no way to explain this behavior. In 1925, S. Goudsmidt and G. Uhlenbeck realized that the classical model just cannot apply. Electrons do not spin like tops; their magnetic behavior must be expla ...
Molecules and formulae - Deans Community High School
... change the elements that are in it. For example carbon tetrachloride is the same as carbon chloride so it will still contain carbon and chlorine; carbon dioxide will still contain carbon and oxygen. Write a heading and try the work below. 1. How do you know that a tricycle has three wheels? 2. What ...
... change the elements that are in it. For example carbon tetrachloride is the same as carbon chloride so it will still contain carbon and chlorine; carbon dioxide will still contain carbon and oxygen. Write a heading and try the work below. 1. How do you know that a tricycle has three wheels? 2. What ...
CH2 Student Revision Guides pdf
... Van der Waals forces are the weak intermolecular forces that exist between all atoms and molecules and include induced-dipole - induced-dipole interactions and dipole-dipole interactions. . The electrons within an atom or molecule are in motion and at a given instant they may be so displaced that th ...
... Van der Waals forces are the weak intermolecular forces that exist between all atoms and molecules and include induced-dipole - induced-dipole interactions and dipole-dipole interactions. . The electrons within an atom or molecule are in motion and at a given instant they may be so displaced that th ...
Chapter 2: Atoms, Molecules and Ions
... • Understand atomic mass, isotopes and be able to calculate average atomic mass. • Become more familiar with the Periodic table: Families (columns) • Understand compound formulas: molecular, empirical, structural • Understand the difference between molecular and ionic compounds • Become proficient i ...
... • Understand atomic mass, isotopes and be able to calculate average atomic mass. • Become more familiar with the Periodic table: Families (columns) • Understand compound formulas: molecular, empirical, structural • Understand the difference between molecular and ionic compounds • Become proficient i ...
Selective field ionization in Li and Rb: Theory and experiment
... start to cross, since there are a very large number of paths that lead to ionization at field F, with nearly randomly varying phases on the different paths; the differing phases essentially guarantee that the interference between different paths will average to zero. In the model of Ref. 关13兴, the p ...
... start to cross, since there are a very large number of paths that lead to ionization at field F, with nearly randomly varying phases on the different paths; the differing phases essentially guarantee that the interference between different paths will average to zero. In the model of Ref. 关13兴, the p ...
chemistry-2nd-edition-julia-burdge-solution
... oxoanions: the anions that remain when oxoacids lose H + ions; hydrates: ionic solids that have water molecules in their formulas. ...
... oxoanions: the anions that remain when oxoacids lose H + ions; hydrates: ionic solids that have water molecules in their formulas. ...
Openstax - Chemistry - Answer Key
... spheres and two purple spheres. This violates Dalton’s postulate that that atoms are not created during a chemical change, but are merely redistributed. 3. This statement violates Dalton’s fourth postulate: In a given compound, the numbers of atoms of each type (and thus also the percentage) always ...
... spheres and two purple spheres. This violates Dalton’s postulate that that atoms are not created during a chemical change, but are merely redistributed. 3. This statement violates Dalton’s fourth postulate: In a given compound, the numbers of atoms of each type (and thus also the percentage) always ...
Solid-state quantum computing using spectral holes M. S. Shahriar, P. R. Hemmer,
... discuss a concrete model of quantum computing via spectral hole burning. A wide variety of SHB materials exists including quantum dots, and it may be possible to design media that are optimized for quantum computing. During the adiabatic transfer step, there are two primary sources of decoherence: d ...
... discuss a concrete model of quantum computing via spectral hole burning. A wide variety of SHB materials exists including quantum dots, and it may be possible to design media that are optimized for quantum computing. During the adiabatic transfer step, there are two primary sources of decoherence: d ...
Chemistry Activity: Determining the “weighted average” atomic mass
... 4. In the real world, two naturally-occurring samples of an element may not give exactly the same %’s of the isotopes (although they’re usually fairly close.) Think of 2 reasons why this may be so. (2 pts) ...
... 4. In the real world, two naturally-occurring samples of an element may not give exactly the same %’s of the isotopes (although they’re usually fairly close.) Think of 2 reasons why this may be so. (2 pts) ...
IGCSE Revision Question Booklet Mark Scheme
... it would place them in incorrect groups (owtte) idea of not being placed with elements which have similar properties or converse argument accept would not have same number of electrons in outer shell allow it would put them in wrong period if linked to argon and potassium / calcium do not accept rea ...
... it would place them in incorrect groups (owtte) idea of not being placed with elements which have similar properties or converse argument accept would not have same number of electrons in outer shell allow it would put them in wrong period if linked to argon and potassium / calcium do not accept rea ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.